1. Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009; 361(21):2066–2078.

2. Kang B, Choe YH. Early biologic treatment in pediatric Crohn's disease: catching the therapeutic window of opportunity in early disease by treat-to-target. Pediatr Gastroenterol Hepatol Nutr. 2018; 21(1):1–11.

3. Jung D, Lee S, Jeong I, Oh SH, Kim KM. Short-term outcome of infliximab therapy in pediatric Crohn's disease: a single-center experience. Pediatr Gastroenterol Hepatol Nutr. 2017; 20(4):236–243.

4. Shen X, Du J, Guan W, Zhao Y. The balance of intestinal Foxp3+ regulatory T cells and Th17 cells and its biological significance. Expert Rev Clin Immunol. 2014; 10(3):353–362.
5. Ueno A, Jeffery L, Kobayashi T, Hibi T, Ghosh S, Jijon H. Th17 plasticity and its relevance to inflammatory bowel disease. J Autoimmun. 2018; 87:38–49.

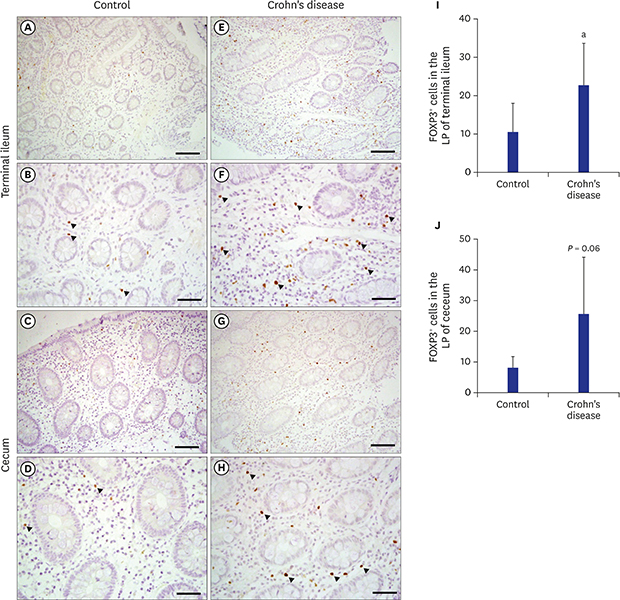
6. Ban H, Andoh A, Shioya M, Nishida A, Tsujikawa T, Fujiyama Y. Increased number of FoxP3+CD4+ regulatory T cells in inflammatory bowel disease. Mol Med Rep. 2008; 1(5):647–650.
7. Lord JD, Valliant-Saunders K, Hahn H, Thirlby RC, Ziegler SF. Paradoxically increased FOXP3+ T cells in IBD do not preferentially express the isoform of FOXP3 lacking exon 2. Dig Dis Sci. 2012; 57(11):2846–2855.
8. Gil JH, Oh JE, Seo JW, Cho MS, Cho KY, Yoo ES. FOXP3+ T cells and TGF-β1 in colonic mucosa of children with Crohn's disease. Korean J Pediatr Gastroenterol Nutr. 2011; 14(3):258–268.
9. Lord JD. Promises and paradoxes of regulatory T cells in inflammatory bowel disease. World J Gastroenterol. 2015; 21(40):11236–11245.

10. Eastaff-Leung N, Mabarrack N, Barbour A, Cummins A, Barry S. Foxp3+ regulatory T cells, Th17 effector cells, and cytokine environment in inflammatory bowel disease. J Clin Immunol. 2010; 30(1):80–89.

11. Neurath MF. Cytokines in inflammatory bowel disease. Nat Rev Immunol. 2014; 14(5):329–342.

12. Strober W, Fuss IJ. Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology. 2011; 140(6):1756–1767.

13. Guidi L, Felice C, Procoli A, Bonanno G, Martinelli E, Marzo M, et al. FOXP3+ T regulatory cell modifications in inflammatory bowel disease patients treated with anti-TNFα agents. BioMed Res Int. 2013; 2013:286368.
14. Ueno A, Jijon H, Chan R, Ford K, Hirota C, Kaplan GG, et al. Increased prevalence of circulating novel IL-17 secreting Foxp3 expressing CD4+ T cells and defective suppressive function of circulating Foxp3+ regulatory cells support plasticity between Th17 and regulatory T cells in inflammatory bowel disease patients. Inflamm Bowel Dis. 2013; 19(12):2522–2534.

15. Makita S, Kanai T, Oshima S, Uraushihara K, Totsuka T, Sawada T, et al. CD4+CD25bright T cells in human intestinal lamina propria as regulatory cells. J Immunol. 2004; 173(5):3119–3130.
16. Yu QT, Saruta M, Avanesyan A, Fleshner PR, Banham AH, Papadakis KA. Expression and functional characterization of FOXP3+ CD4+ regulatory T cells in ulcerative colitis. Inflamm Bowel Dis. 2007; 13(2):191–199.
17. Saruta M, Yu QT, Fleshner PR, Mantel PY, Schmidt-Weber CB, Banham AH, et al. Characterization of FOXP3+CD4+ regulatory T cells in Crohn’s disease. Clin Immunol. 2007; 125(3):281–290.
18. Fantini MC, Monteleone G. Update on the therapeutic efficacy of tregs in IBD: thumbs up or thumbs down? Inflamm Bowel Dis. 2017; 23(10):1682–1688.
19. Desreumaux P, Foussat A, Allez M, Beaugerie L, Hébuterne X, Bouhnik Y, et al. Safety and efficacy of antigen-specific regulatory T-cell therapy for patients with refractory Crohn's disease. Gastroenterology. 2012; 143(5):1207–1217.e2.

20. Choi P, Reiser H. IL-4: role in disease and regulation of production. Clin Exp Immunol. 1998; 113(3):317–319.

21. Park SH, Kim MS, Lim HX, Cho D, Kim TS. IL-33-matured dendritic cells promote Th17 cell responses via IL-1β and IL-6. Cytokine. 2017; 99:106–113.

22. Matsuura T, West GA, Levine AD, Fiocchi C. Selective resistance of mucosal T-cell activation to immunosuppression in Crohn's disease. Dig Liver Dis. 2000; 32(6):484–494.

23. West GA, Matsuura T, Levine AD, Klein JS, Fiocchi C. Interleukin 4 in inflammatory bowel disease and mucosal immune reactivity. Gastroenterology. 1996; 110(6):1683–1695.

24. Mikhalkevich N, Becknell B, Caligiuri MA, Bates MD, Harvey R, Zheng WP. Responsiveness of naive CD4 T cells to polarizing cytokine determines the ratio of Th1 and Th2 cell differentiation. J Immunol. 2006; 176(3):1553–1560.

25. Fuss IJ, Neurath M, Boirivant M, Klein JS, de la Motte C, Strong SA, et al. Disparate CD4+ lamina propria (LP) lymphokine secretion profiles in inflammatory bowel disease. Crohn's disease LP cells manifest increased secretion of IFN-gamma, whereas ulcerative colitis LP cells manifest increased secretion of IL-5. J Immunol. 1996; 157(3):1261–1270.
26. Reinisch W, Hommes DW, Van Assche G, Colombel JF, Gendre JP, Oldenburg B, et al. A dose escalating, placebo controlled, double blind, single dose and multidose, safety and tolerability study of fontolizumab, a humanised anti-interferon gamma antibody, in patients with moderate to severe Crohn's disease. Gut. 2006; 55(8):1138–1144.
27. Leonard WJ, Wan CK. IL-21 signaling in immunity. F1000 Res. 2016; 5:5.

28. Tangye SG. Advances in IL-21 biology - enhancing our understanding of human disease. Curr Opin Immunol. 2015; 34:107–115.
29. Pastorelli L, Garg RR, Hoang SB, Spina L, Mattioli B, Scarpa M, et al. Epithelial-derived IL-33 and its receptor ST2 are dysregulated in ulcerative colitis and in experimental Th1/Th2 driven enteritis. Proc Natl Acad Sci U S A. 2010; 107(17):8017–8022.

30. Sakuraba A, Sato T, Kamada N, Kitazume M, Sugita A, Hibi T. Th1/Th17 immune response is induced by mesenteric lymph node dendritic cells in Crohn's disease. Gastroenterology. 2009; 137(5):1736–1745.

31. Hueber W, Sands BE, Lewitzky S, Vandemeulebroecke M, Reinisch W, Higgins PD, et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn's disease: unexpected results of a randomised, double-blind placebo-controlled trial. Gut. 2012; 61(12):1693–1700.







 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download