INTRODUCTION
MATERIALS AND METHODS
Study population and HCCEP
Echocardiography
Statistical analyses of data
RESULTS
Study population
Table 1
Baseline Characteristics and 1-Year HCCEP of the Study Population
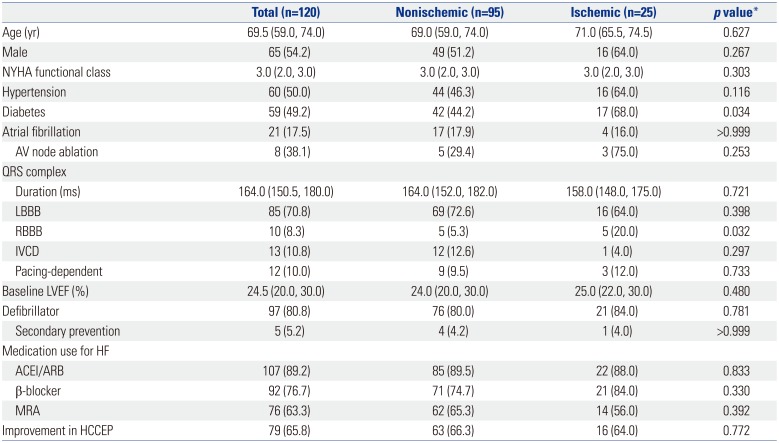
ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; AV, atrioventricular; HCCEP, hierarchical clinical composite end point; HF, heart failure; IVCD, intraventricular conduction delay; LBBB, left bundle branch block; LVEF, left ventricular ejection fraction; MRA, mineralocorticoid receptor antagonist; NYHA, New York Heart Association; RBBB, right bundle branch block.
Data are expressed as median (interquartile range) or number (percent).
*Comparison between nonischemic and ischemic groups.
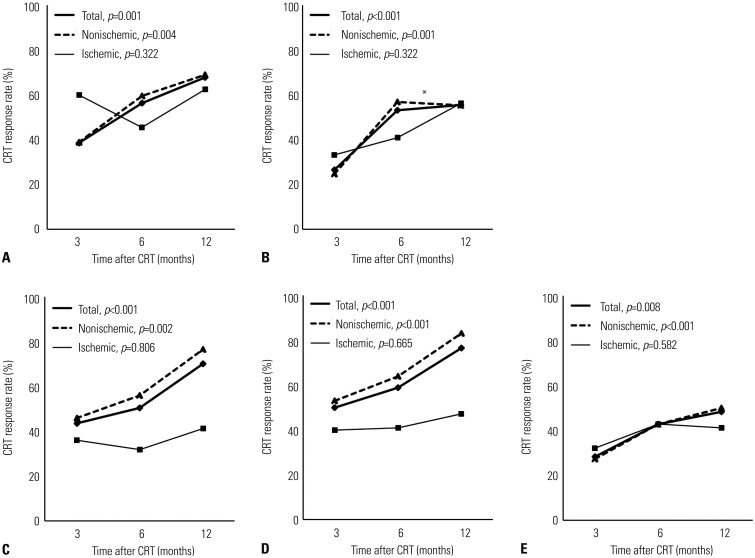
Temporal changes in CRT response
Fig. 1
Temporal changes in CRT response rates according to (A) ΔLVESV, (B) ΔLVEDV, (C) absolute ΔLVEF, (D) relative ΔLVEF, and (E) ΔMR in total, nonischemic, and ischemic groups. p values indicate the statistical significance of temporal changes in CRT response rates according to each echocardiographic CRT response criterion in each group from 3 to 12 months. *The temporal change in CRT response, based on ΔLVEDV from 6 to 12 months, in nonischemic group is insignificant in post-hoc analysis. CRT, cardiac resynchronization therapy; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; MR, mitral regurgitation.
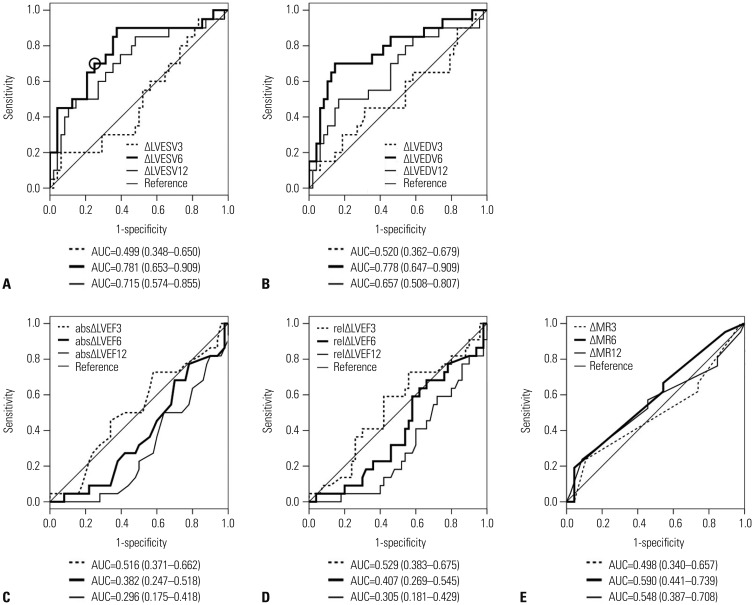
Accuracy and optimal cutoff value of echocardiographic CRT response criteria for the discrimination of 1-year HCCEP
Fig. 2
Receiver operating characteristic curve and area under the curve of (A) ΔLVESV, (B) ΔLVEDV, (C) absolute ΔLVEF, (D) relative ΔLVEF, and (E) ΔMR for the discrimination of improvement in 1-year hierarchical clinical composite end point in total group. When the optimal cutoff value of ΔLVESV at 6 months is set as 13.5% (circle), sensitivity and specificity are 0.719 and 0.719, respectively. abs, absolute; AUC, area under the curve; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; MR, mitral regurgitation; rel, relative.
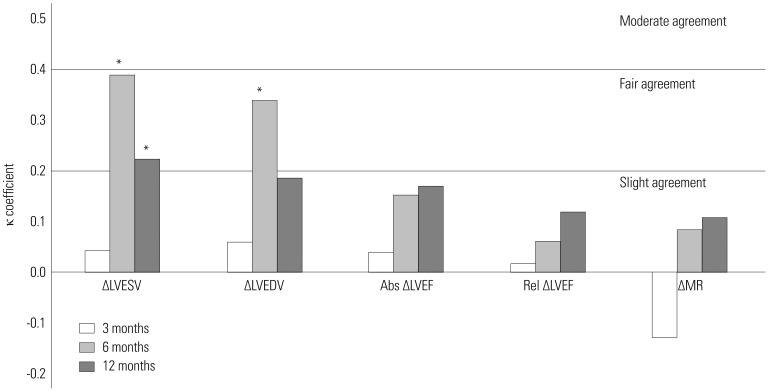
Agreement of echocardiographic CRT response criteria with 1-year HCCEP
Fig. 3
Agreements of echocardiographic CRT response criteria at each time with improvement in 1-year hierarchical clinical composite end point in total group. *p<05. Abs, absolute; CRT, cardiac resynchronization therapy; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; MR, mitral regurgitation; Rel, relative.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download