Abstract
Fine-needle aspiration (FNA) with ultrasonography is considered a minimally invasive and safe procedure. Complications of it are infrequent and occur immediately or within a few days after FNA. Such complications may occur mainly in patients with underlying problems. We here report a rare case of thyroid cystic nodule infection occurring 1 month after FNA in an immunocompetent patient and serial sonographic findings in this patient. A 33-year-old woman with a cystic nodule including partially isoechoic solid areas on the right thyroid gland complained of difficulty swallowing and painful sensations in the right neck 1 month after FNA. On follow-up examination, the cystic nodule and perithyroidal soft tissue were suspicious of infection. The possibility of infection after FNA should be considered even if the patient is immunocompetent in order to prompt evaluation and immediate management with empirical antibiotic therapy to avoid life-threatening complications.
Thyroid nodules are very common and can be classified as solid, mixed, or cystic according to their compositions by ultrasonography (US). Up to 50% of solitary thyroid nodules are cystic.12) The clinical importance of thyroid nodules rests with the need to exclude thyroid cancer. The vast majority (82–91%) of thyroid cancer are solid.3) Partially or purely cystic nodules have a low risk of malignancy. Appropriate evaluation and management of patients with cystic thyroid nodules are areas of controversy.
Currently, fine-needle aspiration (FNA) with US has become the diagnostic intervention to evaluate whether a thyroid nodule is malignant or benign. Especially, in purely cystic nodules, the risk of malignancy is very low and FNA is not recommended for diagnostic purposes. Aspiration with or without ethanol ablation may be considered as a therapeutic intervention if a cyst is large and symptomatic; cytology should be performed if aspiration is performed.3) US-guided FNA is considered a minimally invasive and safe procedure and complications are infrequent. The most common complications are local pain, and mild hematoma in this procedure. Acute or delayed transient swelling of the thyroid gland can occurs rarely and is usually resolved with conservative treatments. Serious complications include massive hematoma, abscess, and acute suppurative thyroiditis. Such complications mainly occur immediately or within a few days after FNA in patients with underlying problem. Thyroid gland infections are rare. The remarkable resistance of the thyroid gland to infection depends on protective mechanisms such as a rich blood supply to lymphatic drainage from the thyroid, high glandular iodine content, and a protective capsule surrounding the gland.4) We here report a rare case of the infection of thyroid cystic nodule occurring 1 month after FNA in an immunocompetent patient and serial sonographic findings in this patient.
A 33-year-old woman came to our hospital for follow-up regarding a right thyroid nodule on January 14, 2015 (Fig. 1A, B). She had been incidentally diagnosed with a 4.12×2.42×4.15 cm thyroid nodule 6 months earlier at another hospital (Fig. 1C, D). US-guided FNA cytology was performed at that time, and the result was benign. She had no history of medical disease. On physical examination, there was a palpable nodule and no focal tenderness of the neck. The US findings of the thyroid nodule showed a cystic nodule with partially isoechoic solid areas. Chlorhexidine gluconate 0.5% solution was applied to the skin surface for disinfection. After FNA, the patient did not have any complaints. The cytologic examination revealed benign follicular cells (Fig. 2A). However, 1 month later, the patient visited the hospital and complained of difficulty swallowing and swelling in the right neck for several days. She had no fever. On physical examination, her right neck exhibited focal tenderness without erythematous change. She had a blood pressure of 120/80 mmHg, pulse rate of 88 beats per minute, body temperature of 36.8℃, and respiratory rate of 20 breaths per minute. US of neck revealed slightly increased the nodule size and soft tissue thickening involving the right perithyroidal area and strap muscle (Fig. 3A, B). Among the laboratory findings, a peripheral blood test indicated a white blood cell count of 10,700/mm3 (neutrophil 73.9%), hemoglobin of 14.2 g/dL, and platelet count of 274,000/mm3. Erythrocyte sedimentation rate (ESR, 51 mm, 0–20 mm) and C-reactive protein (CRP, 93 mg/L, <5 mg/L) were elevated. The thyroid function test showed normal levels of thyroid stimulating hormone (TSH, 0.8 uIU/mL, 0.4–4.7 uIU/mL) with free thyroxine levels of 1.17 ng/dL (0.8–1.9 ng/dL), and triiodothyronine levels of 0.69 ng/dL (0.6–1.7 ng/dL). FNA was performed for decompression and 9 cc of dark brown fluid was aspirated. Biochemical test of the fluid showed a white blood cell count of 57,800/mm3 (neutrophil 94.6%) and hemoglobin of 1.5 g/dL. Cytology showed the presence of many neutrophils and degenerative colloidal materials (Fig. 2B). Unfortunately, the culture of aspirated fluid was not performed. Thyroid dynamic computed tomography (CT) revealed a 4.2×3.6×4.8 cm cystic mass involving the right thyroid gland and compressing the trachea (Fig. 4). Soft tissue infiltration and multilayering hypodensity involving right perithyroidal area, strap muscle, and retropharyngeal area were observed. The cystic nodule and perithyroidal soft tissue were suspicious of infection. The patient was given an empirical antibiotic therapy of oral cefixime (100 mg q 12 hours for 6 weeks) and acetaminophen. The decision and schedules of antibiotic use were based on the possibility of complicated infection and the US monitoring of the response to antibiotic therapy. Three days after the initiation of antibiotic therapy, the US characteristics of the cystic nodule changed to spongiform-like lesion (Fig. 3C, D). The patient responded to the treatment well. During two months of follow-up, CT showed a 2.3-cm-sized, thick-walled cystic mass interval improvement of inflammatory change (Fig. 4C, D). The infiltration of retropharyngeal space and strap muscle was disappeared, and CRP (0.8 mg/L) was decreased. Thyroid function tests were within the normal range. During 6 months (Fig. 5A, B, 1.43×0.71×1.3 cm, volume 0.69 mL) and 2 years (Fig. 5C, D, 0.83×0.64×1.20 cm, volume 0.37 mL) of follow-up, thyroid US showed a marked decrease in the size of the nodule.
Thyroid infection regardless of the FNA is extremely rare and usually occurs in immunocompromised patients. Conditions including chemotherapy, diabetes mellitus, hemodialysis, and human immunodeficiency virus infection appear to increase the risk for developing of acute suppurative thyroiditis or thyroid abscess.5678910) However, it has also been reported in immunocompetent patients.1112) In our patient who had no anatomical deformities and underlying disease, the infection of a cystic thyroid nodule occurred 1 month after FNA cytology. Although we perform up to 1000 FNAs per year in our department, this is the only case in which an infection of the thyroid cystic nodule developed both before and after FNA. The possibility of such complications should be kept in mind even if a patient is immunocompetent. To our knowledge, this is the first report of the infection of thyroid cyst occurring 1 month after FNA in a healthy individual in South Korea.
The management of partially cystic nodules of the thyroid is still a matter of debate. However, FNA is generally preferred to decompress the volume rather than to exclude malignancy in cases of large cystic nodules. This treatment can lead to size decrease or resolution of the thyroid cyst. As mentioned earlier, thyroid gland is resistant to bacterial infection due to protective mechanisms, including a rich blood supply to and lymphatic drainage form the thyroid, the high glandular content of iodine, and a protective capsule surrounding the gland.813) The route of infection is rarely documented.
Acute suppurative thyroiditis regardless FNA is known to occur in the patients with pyriform sinus fistulae, which are considered the most common source of thyroid infection.13) Acute suppurative thyroiditis can arise from extensions from infection in the neck and spread via hematogenous or lymphatic pathways from distance sources. Rarely, infections result from direct trauma or inoculation during invasive procedures, such as FNA of the thyroid gland or central venous line placement.8) Thyroid infection is mostly caused by bacteria, whereas fungi, parasitic organisms, and mycobacteria are rarely isolated. Direct seeding of pathogenic organisms has been recognized as a possible, albeit rare, complication of FNA.814) In Korea, several cases, which did not undergo FNA, of acute suppurative thyroiditis have been reported in adults.15161718) Most patients had anatomical deformities such as pyriform sinus fistula or underlying condition such as leukemia, diabetes mellitus or elderly.
A literature review of cases of acute suppurative thyroiditis and thyroid abscess after FNA is summarized in Table 1.5681112) Three out of five cases were in immunocompromised patients with impaired glucose tolerance, diabetes mellitus, leukopenia due to chemotherapy for cancer, and hypertension. One had severe atopic dermatitis, and another was immunocompetent. Interestingly, except for one woman aged 39, all four cases occurred in men between the ages of 50 and 65 years. The sizes of cyst and techniques were various. All patients had thyroid function tests that were normal at the time of FNA. One case showed mild thyrotoxicosis after infection.8) Two cases occurred immediately, and two cases occurred within one week after FNA. Only one case, in a patient with leukopenia due to chemotherapy for colon cancer and who was highly immunosuppressed, had delayed infection at 3 months. The pathogens identified were Escherichia coli (E. coli), Coagulase-negative staphylococcus (CoNS), Propionibacterium acnes (P. acnes), and Staphylococcus aureus (S. aureus). Staphylococci and group A streptococci are among the most common inhabitants of the skin and mucosa in humans. P. acnes is a normal part of the skin flora present on healthy adult humans skin. The case of E. coli was occurred the patient with cancer and leukopenia. E. coli and other Enterobacteriaceae tend to colonize the skin of debilitated patient and those with defects in host resistance due to diabetes mellitus, cirrhosis, recent irradiation, cytotoxic drugs, or steroids.519) In previous reports, the direct spread of infection occurring a relatively short time after FNA and considering the pathogens was suspected.812) In our patient, who was a healthy individual, the infection of thyroid cyst involving soft tissue at 1 month after FNA was resolved after empirical antibiotic therapy, although the pathogen itself was not confirmed. This case had no yellowish pus. The result of cytology showed many neutrophil with degenerative colloidal materials. There was no suspicious necrotic debris of acute suppurative thyroiditis or thyroid abscess. The patient didn't have skin disease. During FNA procedure, we implemented appropriate disinfection protocols to prevent the infection. Considering the time of infection after the procedure, the possibility of direct infection from skin is low. It might have occurred independently of FNA. However, it is difficult to exclude a postoperative infection with a long incubation period although the cause of infection and the relation between procedure and infection are not clear.
Complications of thyroid abscesses include tracheal or esophageal perforation, descending necrotizing mediastinitis, extension into the deep spaces of the neck, and death.20) Therefore, the prompt evaluation and immediate management of infections with empirical antibiotic therapy are important to avoid life-threatening complications. Physicians should be aware of the possibility of infection, especially in immunocompromised patients. Even though there are no recommendations for the use of prophylactic antibiotics after FNA and further studies should be conducted to investigate the utility it, such prophylaxis may be needed for highly selective individual.
In conclusion, FNA-related thyroid infection is very rare. The possibility of delayed infection should be considered in immunocompetent patient. Immediate treatment with empirical antibiotic therapy is recommended in case of infection. If possible, the characteristic of the fluid from thyroid cyst is required to be identified via FNA and the pathogen should be cultures because it can differ in clinical course and management according to pathogens.
Figures and Tables
 | Fig. 1Thyroid US (transverse view) and Doppler flow showed a 4.12×2.42×4.15 cm partially cystic nodule (arrows) with solid area in the right thyroid gland during the first visit our hospital (A, B) and a local hospital visit (C, D). |
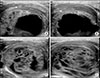
 | Fig. 2(A) FNA cytology showed normal thyroid follicular cells without inflammatory cells at the first visit to our hospital. (B) After 1 month, cytology showed many neutrophils and degenerative colloidal m aterials (Papanicolaou's stain, ×400). |
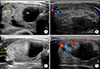
 | Fig. 3After 1 month, thyroid US in transverse view (A) and longitudinal view (B) showed soft tissue thickening involving the right perithyroidal area and strap muscle (arrows). Three days later after antibiotic therapy, thyroid US showed the changed (C, D). |
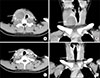 | Fig. 4Thyroid dynamic CT showed a 4.2×3.6×4.8 cm cystic mass (arrows) involving right thyroid gland and compressing the trachea in axial view (A) and in coronal view (B). It showed an ill-defined right thyroid capsule and soft tissue infiltration involving right perithyroidal area, strap muscle and retropharyngeal area. After 6 months, a cystic mass was decreased (C, D). |
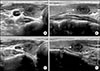 | Fig. 5Thyroid US showed a significant decreases in the size of nodule (arrows) during 6 months (A, B) and 2 year of follow-up (C, D) in transverse and longitudinal views. |
References
1. Frates MC, Benson CB, Doubilet PM, Kunreuther E, Contreras M, Cibas ES, et al. Prevalence and distribution of carcinoma in patients with solitary and multiple thyroid nodules on sonography. J Clin Endocrinol Metab. 2006; 91(9):3411–3417.

2. Lee MJ, Kim EK, Kwak JY, Kim MJ. Partially cystic thyroid nodules on ultrasound: probability of malignancy and sonographic differentiation. Thyroid. 2009; 19(4):341–346.

3. Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association Management Guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016; 26(1):1–133.

4. Berger SA, Zonszein J, Villamena P, Mittman N. Infectious diseases of the thyroid gland. Rev Infect Dis. 1983; 5(1):108–122.

5. Wang YC, Yeh TS, Lin JD. Gram-negative thyroid abscess resulting from fine-needle aspiration in an immunosuppressed patient. Clin Infect Dis. 1997; 25(3):745–746.

6. Isenberg SF. Thyroid abscess resulting from fine-needle aspiration. Otolaryngol Head Neck Surg. 1994; 111(6):832–833.

7. Igarashi H, Yoshino H, Hijikata M, Kumashiro N, Ando Y, Uchino H, et al. Acute suppurative thyroiditis in infected thyroid cyst in an adult patient under hemodialysis. Clin Case Rep. 2017; 5(5):570–573.

8. Nishihara E, Miyauchi A, Matsuzuka F, Sasaki I, Ohye H, Kubota S, et al. Acute suppurative thyroiditis after fine-needle aspiration causing thyrotoxicosis. Thyroid. 2005; 15(10):1183–1187.

9. Jeng LB, Lin JD, Chen MF. Acute suppurative thyroiditis: a ten-year review in a Taiwanese hospital. Scand J Infect Dis. 1994; 26(3):297–300.

10. Lecuit M, Caumes E, Bricaire F, Gatineau M, Menegaux F, Gentilini M. Acute suppurative Salmonella enteritidis thyroiditis associated with thyrotoxicosis in a patient infected with the human immunodeficiency virus. Clin Infect Dis. 1995; 20(1):196.

11. Ünlütürk U, Ceyhan K, Corapcioglu D. Acute suppurative thyroiditis following fine-needle aspiration biopsy in an immunocompetent patient. J Clin Ultrasound. 2014; 42(4):215–218.

12. Sun JH, Chang HY, Chen KW, Lin KD, Lin JD, Hsueh C. Anaerobic thyroid abscess from a thyroid cyst after fine-needle aspiration. Head Neck. 2002; 24(1):84–86.

13. Werner SC, Ingbar SH, Braverman LE, Utiger RD. Werner and Ingbar's the thyroid a fundamental and clinical text. 9th ed. Philadelphia: Lippincott Williams & Wilkins;2005. p. 541–547.
14. Farwell AP, Braverman LE. Inflammatory thyroid disorders. Otolaryngol Clin North Am. 1996; 29(4):541–556.
15. Kim BS, Nam KW, Kim JE, Park JH, Yoon JS, Park JH, et al. A case of acute suppurative thyroiditis with thyrotoxicosis in an elderly patient. Endocrinol Metab (Seoul). 2013; 28(1):50–54.

16. Choi HS, Ahn HY, Lee JS, Kim H, Lim JA, Kim TH, et al. A case of acute suppurative thyroiditis in a patient with leukemia who was treated with chemotherapy. J Korean Endocr Soc. 2009; 24(1):38–41.

17. Kim KW, Park YJ, Kim TY, Moon MK, Han SW, Eun CJ, et al. A case of acute suppurative thyroiditis caused by pyriform sinus fistula with thyrotoxicosis. J Korean Soc Endocrinol. 2004; 19(1):69–75.
18. Yoo CH, Kim YN, Shon CB, Park EH, Choi YS, Park YH, et al. A case of acute suppurative thyroiditis with thyrotoxicosis. Korean J Med. 2008; 75(2):242–245.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download