초록
Anti-programmed cell death-1 (PD-1) humanized monoclonal antibody inhibits PD-1 activity by binding to the PD-1 receptor on T-cells and blocking PD-1 ligands and induces immune tolerance of cancer cells. It has been widely used for various kinds of cancer treatment. However, many immune-related adverse events (irAEs) have been reported because it modulates our immune system. In this case study, we reported a case of 42-year-old woman with Hashimoto's thyroiditis who showed rapid aggravation of thyroid goiter and acute hyperventilation syndrome after treatment with PD-1 inhibitor as a neoadjuvant chemotherapy for breast cancer.
Go to : 
REFERENCES
1). Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med. 2015; 372(21):2018–28.

2). Herbst RS, Baas P, Kim DW, Felip E, Perez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD- L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016; 387(10027):1540–50.
3). Langer CJ, Gadgeel SM, Borghaei H, Papadimitrakopoulou VA, Patnaik A, Powell SF, et al. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol. 2016; 17(11):1497–508.

4). Chen R, Zinzani PL, Fanale MA, Armand P, Johnson NA, Brice P, et al. Phase II study of the efficacy and safety of pembrolizumab for relapsed/refractory classic Hodgkin lymphoma. J Clin Oncol. 2017; 35(19):2125–32.

5). Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012; 366(26):2443–54.

6). Robert C, Schachter J, Long GV, Arance A, Grob JJ, Mortier L, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015; 372(26):2521–32.

7). de Filette J, Jansen Y, Schreuer M, Everaert H, Velkeniers B, Neyns B, et al. Incidence of thyroid-related adverse events in melanoma patients treated with pembrolizumab. J Clin Endocrinol Metab. 2016; 101(11):4431–9.

8). Delivanis DA, Gustafson MP, Bornschlegl S, Merten MM, Kottschade L, Withers S, et al. Pembrolizumab-induced thyroiditis: comprehensive clinical review and insights into underlying involved mechanisms. J Clin Endocrinol Metab. 2017; 102(8):2770–80.

9). Orlov S, Salari F, Kashat L, Walfish PG. Induction of painless thyroiditis in patients receiving programmed death 1 receptor immunotherapy for metastatic malignancies. J Clin Endocrinol Metab. 2015; 100(5):1738–41.

10). Hashimoto H. Zur Kenntnis der lymphomatösen Veränderung der Schilddrüse (Struma lymphomatosa). Arch Klin Chir. 1912; 97:219–48.
11). Rapoport B. Pathophysiology of Hashimoto's thyroiditis and hypothyroidism. Annu Rev Med. 1991; 42:91–6.

12). Huber G, Staub JJ, Meier C, Mitrache C, Guglielmetti M, Huber P, et al. Prospective study of the spontaneous course of subclinical hypothyroidism: prognostic value of thyrotropin, thyroid reserve, and thyroid antibodies. J Clin Endocrinol Metab. 2002; 87(7):3221–6.

13). Rieu M, Bekka S, Sambor B, Berrod JL, Fombeur JP. Prevalence of subclinical hyperthyroidism and relationship between thyroid hormonal status and thyroid ultrasonographic parameters in patients with non-toxic nodular goitre. Clin Endocrinol (Oxf). 1993; 39(1):67–71.

14). Ribas A, Puzanov I, Dummer R, Schadendorf D, Hamid O, Robert C, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015; 16(8):908–18.
15). Guaraldi F, La Selva R, Sama MT, D'Angelo V, Gori D, Fava P, et al. Characterization and implications of thyroid dysfunction induced by immune checkpoint inhibitors in real-life clinical practice: a long-term prospective study from a referral institution. J Endocrinol Invest. 2018; 41(5):549–56.

Go to : 
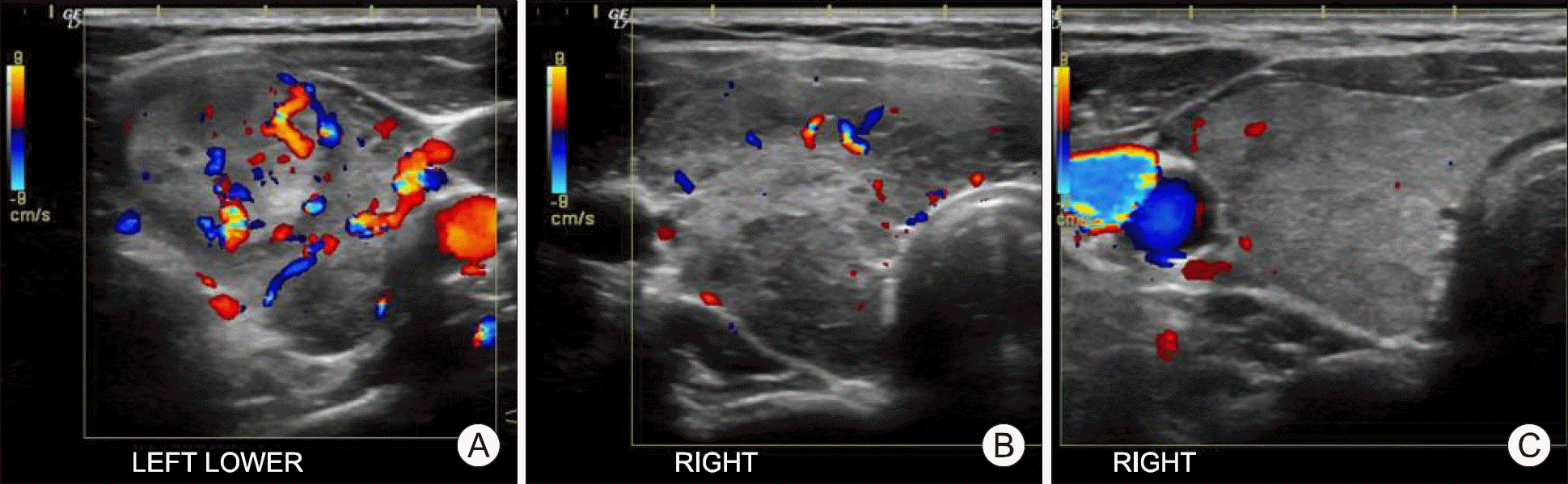 | Fig. 1.Representative images of thyroid ultrasonography. (A) Diffuse enlargement of goiter and increased blood flow in left thyroid gland at the time of respiratory distress. (B) The size of right middle lobe was 25.9×28.2×45.7 mm at the time of respiratory distress. (C) Twenty days after treatment with prednisolone, the size of right thyroid middle lobe was 22.9×15.0×43.8 mm and the volume decreased by 54%. |
Table 1.
The changes in thyroid function and thyroid autoantibodies with treatment of steroid and levothyroxine




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download