INTRODUCTION
CASE DESCRIPTION
Case 1
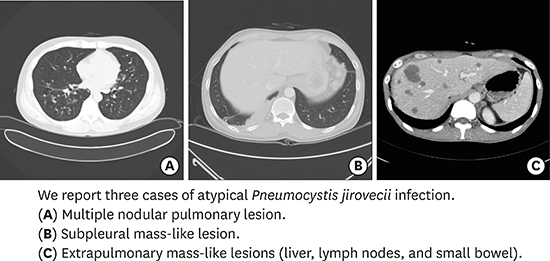
 | Fig. 1Findings of the 32-year-old man with multiple pulmonary nodules (case 1). (A, B) Simple chest X-ray and chest CT on admission day show disseminated multiple nodules with some cavitation (arrow) on whole lung field. (C) Chest CT on hospital day 29 shows decreased size of multiple nodules with disappearance of cavitary lesions (arrow). (D) Simple chest X-ray after 16 months from discharge shows previous multiple nodules disappeared. (E-G) Histopathologic feature of lung specimen shows necrotizing granulomatous lesions (arrows) and pink, foamy exudate (E, F: H & E stain; × 100, × 200, respectively) containing oval shaped fungal cysts (arrows) within alveolar space (G: GMS stain; × 400).
CT = computed tomography, H & E = hematoxylin and eosin, GMS = gomori methenamine-silver.
|
Case 2
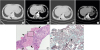 | Fig. 2Findings of the 43-year-old man with subpleural mass-like lesion (case 2). (A, B) Chest CT on admission day shows a 13.35 cm2 lobulated homogeneously enhancing mass-like lesion (arrows) on right lower lobe. (C, D) Chest CT shows the mass size decreased to 5.87 cm2 (arrows) after one month from discharge. (E, F) Histopathologic feature of lung specimen shows fibrotic granulomatous lesions with necrotic amorphous bubbly area (arrows) (E: H & E stain; × 200) which contained oval shaped fungal cysts (arrows) (F: GMS stain; × 400).
CT = computed tomography, H & E = hematoxylin and eosin, GMS = gomori methenamine-silver.
|
Case 3
 | Fig. 3Findings of the 39-year-old man with extrapulmonary mass-like lesions (case 3). (A, B) Abdominal CT on admission day shows multiple hepatic nodules (arrows), multiple small lymph nodes in upper abdominal region (arrows), and diffuse jejunal wall thickening (arrows). (C, D) PET-CT on hospital day 10 shows no increased metabolic uptake on mentioned lesions, and diffuse jejunal wall thickening. (E, F) Abdominal CT on hospital day 17 shows significant improvement in mentioned areas (arrows). (G, H) Histopathologic feature of liver specimen shows granuloma with ill-formed necrosis (arrows) (H & E stain; × 200, × 400, respectively).
CT = computed tomography, PET-CT = positron emission tomography-computed tomography, H & E = hematoxylin and eosin.
|




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download