1. Katz J, Cooper EM, Walther RR, Sweeney EW, Dworkin RH. Acute pain in herpes zoster and its impact on health-related quality of life. Clin Infect Dis. 2004; 39:342–348.
2. Cohen JI. Clinical practice: Herpes zoster. N Engl J Med. 2013; 369:255–263.
3. Reid JS, Ah Wong B. Herpes zoster (shingles) at a large New Zealand general practice: incidence over 5 years. N Z Med J. 2014; 127:56–60.
4. Kawai K, Gebremeskel BG, Acosta CJ. Systematic review of incidence and complications of herpes zoster: towards a global perspective. BMJ Open. 2014; 4:e004833.
5. Hillebrand K, Bricout H, Schulze-Rath R, Schink T, Garbe E. Incidence of herpes zoster and its complications in Germany, 2005-2009. J Infect. 2015; 70:178–186.
6. Pinchinat S, Cebrián-Cuenca AM, Bricout H, Johnson RW. Similar herpes zoster incidence across Europe: results from a systematic literature review. BMC Infect Dis. 2013; 13:170.
7. Mullooly JP, Riedlinger K, Chun C, Weinmann S, Houston H. Incidence of herpes zoster, 1997-2002. Epidemiol Infect. 2005; 133:245–253.
8. Choi WS, Noh JY, Huh JY, Jo YM, Lee J, Song JY, Kim WJ, Cheong HJ. Disease burden of herpes zoster in Korea. J Clin Virol. 2010; 47:325–329.
9. Kim YJ, Lee CN, Lim CY, Jeon WS, Park YM. Population-based study of the epidemiology of herpes zoster in Korea. J Korean Med Sci. 2014; 29:1706–1710.
10. Mick G, Gallais JL, Simon F, Pinchinat S, Bloch K, Beillat M, Serradell L, Derrough T. Burden of herpes zoster and postherpetic neuralgia: Incidence, proportion, and associated costs in the French population aged 50 or over. Rev Epidemiol Sante Publique. 2010; 58:393–401.
11. Lin YH, Huang LM, Chang IS, Tsai FY, Lu CY, Shao PL, Chang LY. Varicella-Zoster Working Group. Advisory Committee on Immunization Practices, Taiwan. Disease burden and epidemiology of herpes zoster in pre-vaccine Taiwan. Vaccine. 2010; 28:1217–1220.
12. Levi M, Bellini I, Capecchi L, Pieri L, Bechini A, Boccalini S, Callaioli S, Gasparini R, Panatto D, Tiscione E, et al. The burden of disease of Herpes Zoster in Tuscany. Hum Vaccin Immunother. 2015; 11:185–191.
13. Song H, Lee J, Lee M, Choi WS, Choi JH, Lee MS, Hashemi M, Rampakakis E, Kawai K, White R, et al. Burden of illness, quality of life, and healthcare utilization among patients with herpes zoster in South Korea: a prospective clinical-epidemiological study. Int J Infect Dis. 2014; 20:23–30.
14. Choi WS, Kwon SS, Lee J, Choi SM, Lee JS, Eom JS, Sohn JW, Choeng HJ. Immunity and the burden of herpes zoster. J Med Virol. 2014; 86:525–530.
15. Oxman MN, Levin MJ, Johnson GR, Schmader KE, Straus SE, Gelb LD, Arbeit RD, Simberkoff MS, Gershon AA, Davis LE, et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N Engl J Med. 2005; 352:2271–2284.
16. Schmader KE, Levin MJ, Gnann JW Jr, McNeil SA, Vesikari T, Betts RF, Keay S, Stek JE, Bundick ND, Su SC, et al. Efficacy, safety, and tolerability of herpes zoster vaccine in persons aged 50-59 years. Clin Infect Dis. 2012; 54:922–928.
17. Hammond O, Wang Y, Green T, Antonello J, Kuhn R, Motley C, Stump P, Rich B, Chirmule N, Marchese RD. The optimization and validation of the glycoprotein ELISA assay for quantitative varicella-zoster virus (VZV) antibody detection. J Med Virol. 2006; 78:1679–1687.
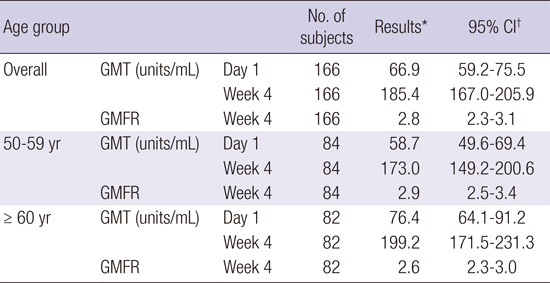









 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download