Abstract
Backgrounds/Aims
Despite hepatolithiasis being a risk factor for biliary neoplasm including cholangiocarcinoma, the incidence of underlying biliary neoplasm is unknown in patients with preoperative benign hepatolithiasis. The aim of this study was to evaluate the incidence of underlying biliary neoplasm in patients who underwent major hepatectomy for preoperative benign hepatolithiasis.
Methods
Between March 2005 and December 2015, 73 patients who underwent major hepatectomy for preoperative benign hepatolithiasis were enrolled in this study. The incidence and pathological differentiation of concomitant biliary neoplasm were retrospectively determined by review of medical records. Postoperative complications after major hepatectomy were evaluated.
Results
Concomitant biliary neoplasm was pathologically confirmed in 20 patients (27.4%). Biliary intraepithelial neoplasia (BIN) was detected in 12 patients (16.4%), and 1 patient (1.4%) had intraductal papillary mucinous neoplasm (IPMN), as the premalignant lesion. Cholangiocarcinoma was pathologically confirmed in 7 patients (9.6%). Preoperative imaging of the 73 patients revealed biliary stricture at the first branch of bile duct in 31 patients (42.5%), and at the second branch of bile duct in 39 patients (53.4%). Postoperative complications developed in 14 patients (19.1%). Almost all patients recovered from complications, including intra-abdominal abscess (9.6%), bile leakage (4.1%), pleural effusion (2.7%), and wound infection (1.4%). Only 1 patient (1.4%) died from aspiration pneumonia.
Go to : 
Hepatolithiasis is defined as the presence of stones within the intrahepatic bile ducts, proximal to the right and left hepatic ducts. Hepatolithiasis has a poor prognosis due to other associated complications such as recurrent cholangitis, biliary strictures, liver abscess, liver atrophy, or cirrhosis.12 Additionally, hepatolithiasis is an important leading cause of intrahepatic cholangiocarcinoma.34
Hepatolithiasis treatment includes both non-surgical and surgical approaches. Non-surgical procedures, such as percutaneous transhepatic cholangioscopic lithotripsy, show a high clearance rate of intrahepatic stones; however, these treatments are not effective for preventing recurrence of hepatolithiasis and removal of benign biliary strictures.567 Surgical treatment can be divided into minor and major hepatectomies, depending on the resection range. The range of minor hepatectomy is generally limited to atrophic segments and intrahepatic stones. In this context, a residual intrahepatic biliary stricture may remain, which is a risk factor for hepatolithiasis. Unlike minor hepatectomy, the range of major hepatectomy includes all intrahepatic duct stones and a wider segment, including the intrahepatic biliary strictures. Thus, major hepatectomy can be the fundamental treatment for hepatolithiasis by eliminating not only the stones but also the benign biliary stricture, which is a crucial cause of the stones.
Previous studies have reported the rate of unidentified underlying cholangiocarcinoma before hepatectomy in patients with hepatolithiasis.28 In these reports, it is uncertain whether the resection range includes all intrahepatic biliary strictures and their segments. To confirm the incidence of underlying biliary neoplasm in patients with hepatolithiasis, we need to investigate patients who underwent major hepatectomy for both hepatolithiasis and intrahepatic biliary strictures.
Here, we evaluated the incidence of underlying biliary neoplasm in patients who underwent major hepatectomy for benign hepatolithiasis, based on preoperative diagnosis.
Go to : 
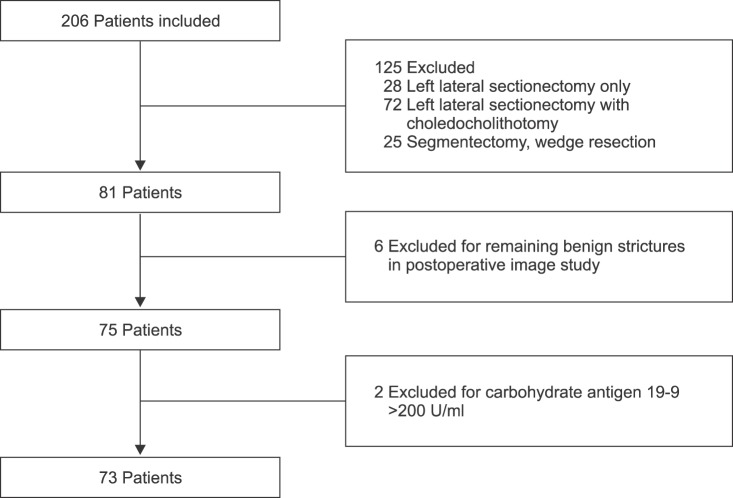
The medical records of Chonnam National University Hospital were examined between March 2005 and December 2015. We identified 206 patients with possible benign hepatolithiasis detected on preoperative computed tomography (CT) or magnetic resonance (MR) imaging, who subsequently underwent hepatic resection. Of the 206 patients, 125 patients underwent minor hepatectomy, including left lateral sectionectomy for hepatolithiasis. These were excluded because of possible residual stricture of the bile ducts related to hepatolithiasis after resection. A total of 81 patients underwent major hepatectomy for hepatolithiasis. Of the 81 patients, 6 patients were excluded because of residual intrahepatic biliary stricture identified on the postoperative CT imaging after the procedure.
For the diagnosis of cholangiocarcinoma, the value of tumor markers remains controversial. Various cut-off values of carbohydrate antigen 19-9 (CA 19-9) have been proposed.9 One study proposed 253 U/ml for CA 19-9 as the mean serum level for hilar cholangiocarcinoma in cases of International Union against Cancer tumor stage I.10 It is generally considered that CA 19-9 levels over 200 U/ml indicate a possibility of cholangiocarcinoma. In our study, 2 patients with serum CA 19-9 values above 200 U/ml on preoperative laboratory tests were excluded due to suspected underlying biliary malignancy. Finally, 73 patients who underwent major hepatectomy for benign hepatolithiasis as diagnosed on preoperative imaging and laboratory tests, were enrolled in this study (Fig. 1).
At our hospital, endoscopic retrograde cholangiopancreatography (ERCP) is performed in patients having stones or strictures localized in the first branch of bile ducts. Patients with definite stones or strictures in other peripheral branches of intrahepatic bile ducts undergo major hepatectomy, regardless of the symptoms and irrespective of the presence of atrophy.
The locations of hepatolithiasis and intrahepatic biliary strictures were evaluated on preoperative CT or MR cholangiography in all patients. Since multiple strictures can be present in the intrahepatic ducts, we designated the biliary stricture located farthest distal in the direction of the bile flow as the main stricture site.
The incidence and the pathological differentiation of a concomitant biliary neoplasm were retrospectively evaluated through review of the medical records for all patients. The postoperative complications after major hepatectomy were also evaluated.
Benign hepatolithiasis was defined as hepatolithiasis with a concomitant stricture of the intrahepatic bile duct, with no clinical evidence of malignancy on CT or MRI, or on preoperative laboratory tests.
Major hepatectomy was defined as the resection of 3 or more Couinaud's liver segments, while segmentectomy of 1 or 2 segments and non-anatomical wedge resection were classified as minor hepatectomy.11
The first branch of the intrahepatic bile duct was defined from the confluence of the right and left hepatic ducts to the bifurcation of the right anterior sectoral duct and right posterior sectoral duct, or to the bifurcation of the segment IV duct and the left lateral sectoral duct. The second branch of the intrahepatic bile duct was defined from the proximal end of the first branch with bile flow to the confluence of the segment V duct and VIII duct, or to the confluence of the segment VI duct and VII duct, or to the confluence of the segment II duct and III duct. The third branch was defined from the proximal end of the second branch to the proximal end of each segmental duct, according to the direction of bile flow.
Patient demographics including sex, age, preoperative symptoms (including abdominal pain, fever, jaundice, or weight loss), preoperative serum CA 19-9, presence of atrophy in preoperative image, and serum total bilirubin before the operation, were compared between the benign stricture group and pathologically proven biliary neoplasm group. The locations of the main intrahepatic biliary strictures and the type of surgical procedure applied were further evaluated between the two groups.
Continuous variables were analyzed using the Mann-Whitney analysis. Categorical variables were compared using the Chi-squared test or Fisher's exact test. Age, preoperative serum CA 19-9, and preoperative total bilirubin were included as continuous variables, while sex, preoperative symptoms, locations of the main biliary stricture, type of operative procedure, presence of atrophy in preoperative image were considered as categorical variables. A p-value of <0.05 was considered statistically significant. The Statistical Package for Social Science version 21.0 was used for the analysis.
Go to : 
Of the 73 patients with preoperative benign hepatolithiasis, 20 patients (27.4%) were confirmed as pathological biliary neoplasm, and 53 patients as chronic inflammation with bile duct stones. Of the 20 patients, 12 (16.4%) presented with biliary intraepithelial neoplasia (BIN) and 1 (1.4%) with intraductal papillary mucinous neoplasm (IPMN) as the premalignant lesions. Cholangiocarcinoma was pathologically confirmed in 7 patients (9.6%) (Table 1).
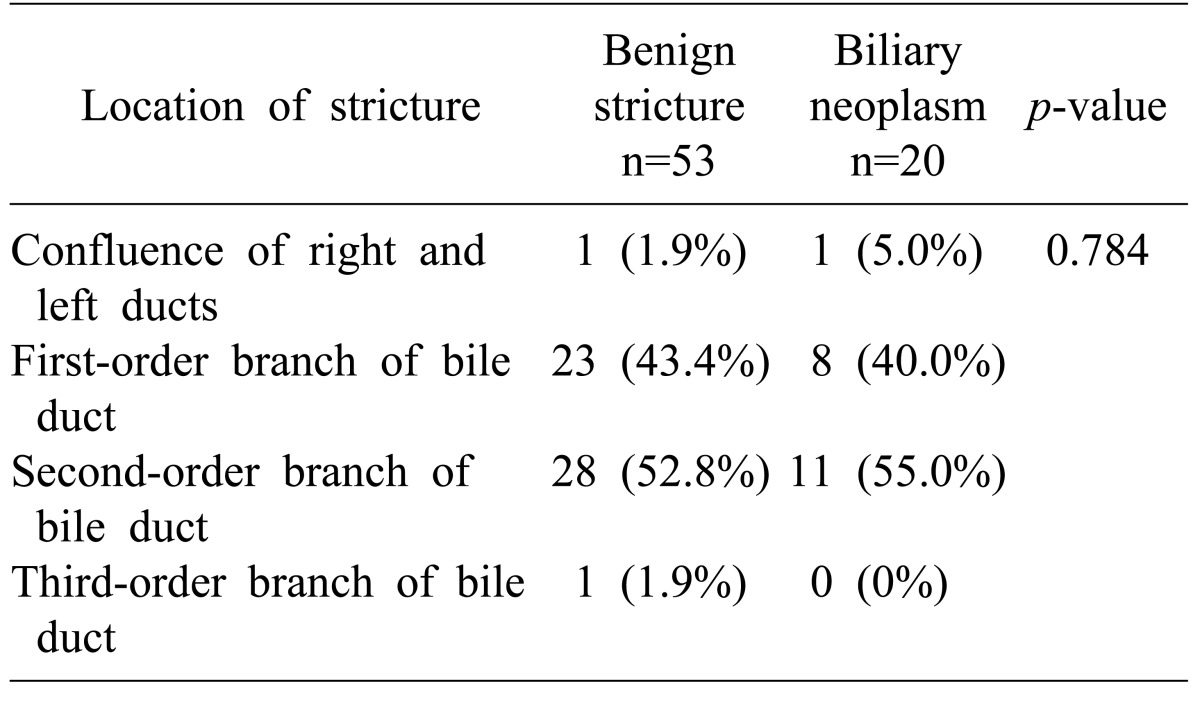
The main intrahepatic biliary strictures were categorized according to their location at the confluence of the right hepatic duct and the left hepatic duct, at the first branch of the intrahepatic bile duct, at the second branch of the bile duct, or at the third branch of bile duct. Biliary stricture at the first branch of bile duct was observed in 23 patients (43.4%) of the benign stricture group and 8 patients (40.0%) of the biliary neoplasm group. A biliary stricture was identified at the second branch of the bile duct in 28 patients (52.8%) in the benign stricture group and in 11 patients (55%) in the biliary neoplasm group. One patient each in the benign stricture group and in the biliary neoplasm group had a biliary stricture at the confluence of the right and left hepatic ducts. A stricture at the third branch of bile duct was seen in 1 patient in the biliary stricture group. Postoperative pathological examination confirmed that the location of the intrahepatic strictures was consistent with that of concomitant biliary neoplasms (Table 2).
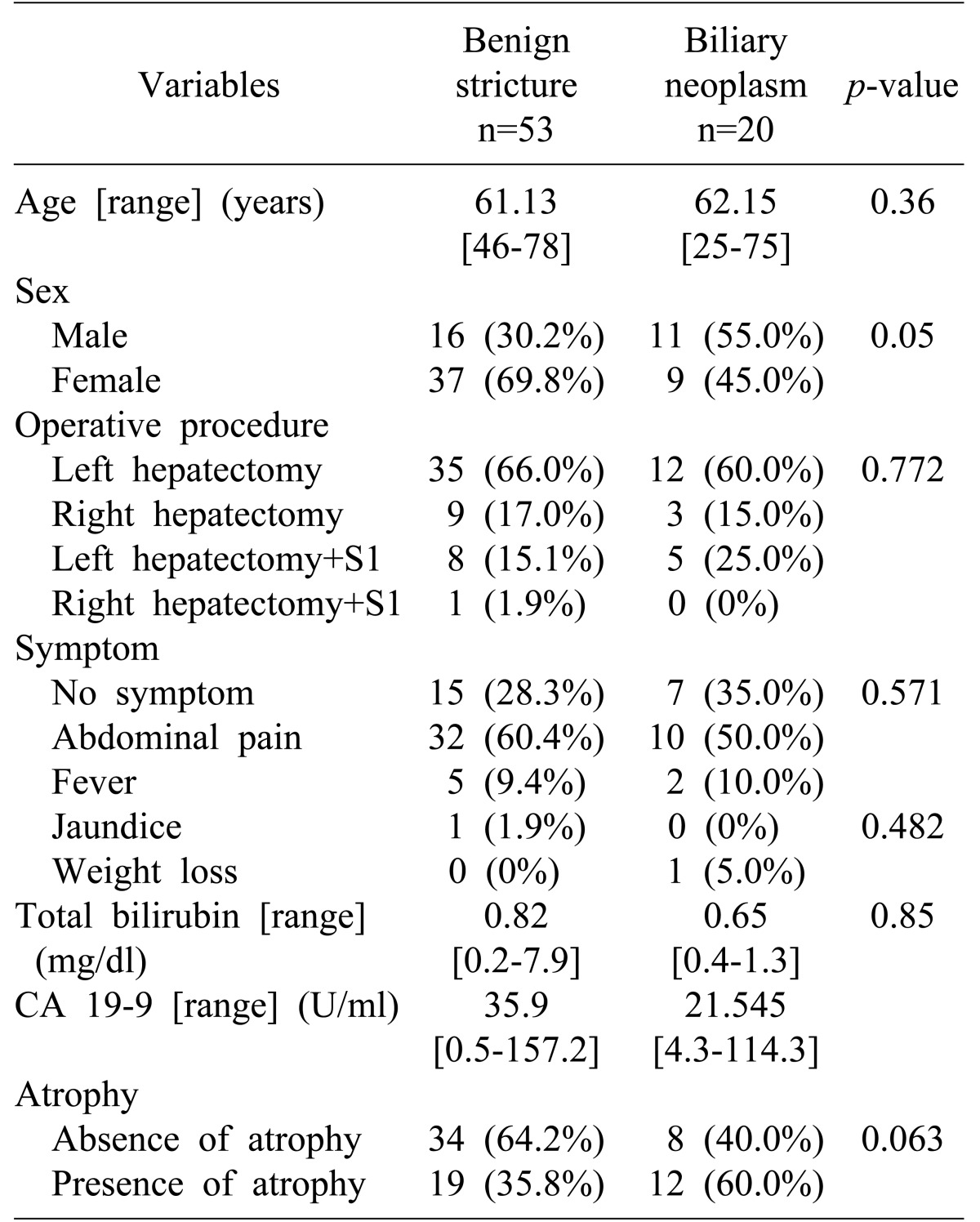
The variables considered were sex, age, the type of surgical procedure, location of the biliary stricture, preoperative symptoms, preoperative serum CA 19-9, presence of atrophy in preoperative image, and preoperative serum total bilirubin level. The main symptoms of patients with hepatolithiasis were abdominal pain, fever, jaundice, and weight loss. Results from the statistical analysis showed no significant difference when comparing the benign stricture group and the pathologically proven biliary neoplasm group. Although sex appeared to be significant, it was not a definitive factor (Table 3).
According to the Clavien-Dindo classification, postoperative complications above grade IIIa developed in 14 patients (19.1%). Of these, 10 patients (13.7%) were treated by procedures such as pig-tail catheter insertion, for intra-abdominal abscess (9.6%), bile leakage (1.4%), or pleural effusion (2.7%). Three patients (4.1%) were re-operated for wound infection, bile duct injury, and common bile duct primary repair site dehiscence. Only 1 patient (1.4%) expired during the hospital stay as a result of aspiration pneumonia, and not by hepatic failure or surgical complications (Table 4).
Go to : 
Epidemiological, pathological, and genetic studies demonstrate a relationship between hepatolithiasis and cholangiocarcinoma.121314 The overall incidence of hepatolithiasis-related cholangiocarcinoma has been reported as 5% to 13%.151617 In a cohort study, 65 years or older (hazard ratio, 3.029; p-value, 0.017) and having stone removal only as the initial treatment (hazard ratio, 2.873; p-value, 0.012) were found to be significant risk factors for the development of cholangiocarcinoma.18 The authors reported that hepatectomy could significantly reduce the risk of developing cholangiocarcinoma (hazard ratio, 0.066; p-value, 0.010). In another cohort study, it was reported that hepatectomy significantly reduced the risk of developing cholangiocarcinoma.19 In general, hepatic resection might offer an advantage in eliminating the risk of developing cholangiocarcinoma because of complete removal of both the intrahepatic stones and the bile ducts involved, which are likely to have a hidden malignancy. In this regard, hepatic resection could be considered as a primary treatment in hepatolithiasis.
Despite preoperative evaluations, it is still difficult to detect underlying biliary neoplasms or chlangiocarcinoma in hepatolithiasis. Catena et al.20 reported that the rate of unrecognized cholangiocarcinoma was quite high at 11.7%, and that it might be underestimated. Although serum CA 19-9 is known as a tumor biomarker for cholangiocarcinoma, its levels could be normal or increased in benign diseases, such as in bacterial cholangitis or choledocholithiasis.2122 In preoperative images or even during the surgery, a diagnosis of cholangiocarcinoma associated with hepatolithiasis is difficult, because the affected liver segment is often fibrotic and scarred.2 Chen et al.2 reported 2 patients (2.0%) having the tumor diagnosed during the operation, and 4 patients (3.9%) having cholangiocarcinoma diagnosed only after pathological examination. They also reported that in 1 patient (1.0%), a cholangiocarcinoma was diagnosed 7 months after the hepatic resection for hepatolithiasis, and the tumor was likely present during the surgery, but was unidentified, and the undetected cholangiocarcinoma was present in the remnant liver even after hepatectomy.
Hepatic resection is regarded as an established treatment, recommended for its ability to resolve not only the stones but also the strictures.2324 Biliary strictures, found in 42% to 96% of patients with hepatolithiasis,252627 may be related to biliary carcinogenesis as an aspect of recurrent cholangitis, bile stasis, and bacterial infections. Biliary stricture is associated with recurrent and chronic inflammations causing prolonged inflammation of the bile duct epithelium, leading to the subsequent development of cholangiocarcinoma.2829 Up to 50% of patients with malignant biliary strictures may have coexistent hepatolithiasis.30 To prevent the development of cholangiocarcinogenesis, hepatic resection, including the biliary stricture, would be considered in cases of hepatolithiasis.
Minor hepatectomy, which involves segmentectomy of 1 or 2 segments and non-anatomical wedge resection, does not include either the first branch or the second branch of the bile duct in the range of resection. In left lateral sectionectomy, where hepatic resection including segments II and III, the resection range includes only the third branch of the bile duct in segments II and III, and does not include the confluence of the segments II duct and III duct, or the second branch of the bile duct. In our study, biliary strictures in hepatolithiasis were mostly located in the first and second branches of the intrahepatic bile duct, and the location of the concomitant biliary neoplasms was consistent with the location of the biliary strictures. Among all the patients who underwent major hepatectomy in benign hepatolithiasis, 31 patients (42.5%) had a biliary stricture at the first branch of bile duct, and 39 patients (53.4%) had a biliary stricture at the second branch of bile duct. Only 1 patient (1.4%) had a biliary stricture at the third branch of the bile duct. In order to completely remove biliary strictures and thus eliminate the hidden biliary neoplasms, major hepatectomy, including the first and second branches of bile duct, would be an appropriate approach in the treatment of hepatolithiasis.
Immunohistochemical studies have identified 2 types of neoplastic lesions preceding invasive intrahepatic cholangiocarcinoma in hepatolithiasis: BIN and IPNM.3132 Ohta et al.33 observed hyperplasia in the epithelium of the bile ducts associated with chronic cholangitis related to hepatolithiasis, and suggested that mucosal dysplasia may be a precursor to the cholangiocarcinoma. It seems that elimination of BIN and IPNM, potential precursors of cholangiocarcinoma, may be required to prevent the development of cholangiocarcinoma in hepatolithiasis.
There also exist concerns regarding surgical complications after major hepatectomy. The surgical techniques of hepatectomy and the perioperative management of patients have sufficiently developed in the past decade, resulting in a marked decrease in the morbidity and mortality following liver surgery.343536 In one study, the cumulative survival rates, excluding unrelated deaths, did not differ significantly between the hepatic resection group and the cholangioscopic lithotomy group.25 In another study evaluating the outcome of hepatectomy for hepatolithiasis, 7 patients (6.8%) underwent re-operation and 2 patients (1.9%) died during hospital stay.2 In our study, 3 patients (4.1%) underwent re-operation and only 1 patient (1.4%) died during hospital stay. The rate of postoperative complication in our study was not significantly different compared to that of other studies.
Our study presents some limitations, including its retrospective nature and the relatively small number of patients involved. Also, there was no comparison of the postoperative complications in major hepatectomy and minor hepatectomy in patients with hepatolithiasis. Finally, no long-term follow-up was performed to confirm that there would be a significant difference in the development of cholangiocarcinoma between major hepatectomy and minor hepatectomy in benign hepatolithiasis.
In our study, the incidence of biliary neoplasm (including cholangiocarcinoma) not detected before hepatectomy was 27.4% in benign hepatolithiasis. Although preoperative evaluations were performed in patients with hepatolithiasis, in order to exclude underlying biliary neoplasm, the incidence of biliary neoplasm was not negligible. Major hepatectomy as the definite treatment was safely performed for these patients. The presence of cholangiocarcinoma was a significant factor influencing the long-term survival in hepatolithiasis patients, as demonstrated by multivariate analysis.214 Major hepatectomy, including not only hepatolithiasis but also concomitant strictures of intrahepatic bile duct, would be one of the appropriate treatment options for preoperative benign hepatolithiasis. However, further research is required to compare major hepatectomy with other treatments, for a prolonged period.
Go to : 
References
1. Lee TY, Chen YL, Chang HC, Chan CP, Kuo SJ. Outcomes of hepatectomy for hepatolithiasis. World J Surg. 2007; 31:479–482. PMID: 17334864.

2. Chen DW, Tung-Ping Poon R, Liu CL, Fan ST, Wong J. Immediate and long-term outcomes of hepatectomy for hepatolithiasis. Surgery. 2004; 135:386–393. PMID: 15041962.

3. Blechacz B, Komuta M, Roskams T, Gores GJ. Clinical diagnosis and staging of cholangiocarcinoma. Nat Rev Gastroenterol Hepatol. 2011; 8:512–522. PMID: 21808282.

4. Tyson GL, El-Serag HB. Risk factors for cholangiocarcinoma. Hepatology. 2011; 54:173–184. PMID: 21488076.

5. Lee SK, Seo DW, Myung SJ, Park ET, Lim BC, Kim HJ, et al. Percutaneous transhepatic cholangioscopic treatment for hepatolithiasis: an evaluation of long-term results and risk factors for recurrence. Gastrointest Endosc. 2001; 53:318–323. PMID: 11231390.

6. Jeng KS, Sheen IS, Yang FS. Percutaneous transhepatic cholangioscopy in the treatment of complicated intrahepatic biliary strictures and hepatolithiasis with internal metallic stent. Surg Laparosc Endosc Percutan Tech. 2000; 10:278–283. PMID: 11083209.

7. Yeh YH, Huang MH, Yang JC, Mo LR, Lin J, Yueh SK. Percutaneous trans-hepatic cholangioscopy and lithotripsy in the treatment of intrahepatic stones: a study with 5 year follow-up. Gastrointest Endosc. 1995; 42:13–18. PMID: 7557170.

8. Kim HJ, Kim JS, Suh SJ, Lee BJ, Park JJ, Lee HS, et al. Cholangiocarcinoma risk as long-term outcome after hepatic resection in the hepatolithiasis patients. World J Surg. 2015; 39:1537–1542. PMID: 25648078.

10. Juntermanns B, Radunz S, Heuer M, Hertel S, Reis H, Neuhaus JP, et al. Tumor markers as a diagnostic key for hilar cholangiocarcinoma. Eur J Med Res. 2010; 15:357–361. PMID: 20947473.

11. Fan ST. Hepatocellular carcinoma--resection or transplant? Nat Rev Gastroenterol Hepatol. 2012; 9:732–737. PMID: 22965432.

12. Zhou Q, Gong Y, Huang F, Lin Q, Zeng B, Li Z, et al. Expression levels and significance of nuclear factor-κB and epidermal growth factor receptor in hepatolithiasis associated with intrahepatic cholangiocarcinoma. Dig Surg. 2013; 30:309–316. PMID: 24008372.

13. Lee KT, Chang WT, Wang SN, Chuang SC, Chai CY, Hu SW. Expression of DPC4/Smad4 gene in stone-containing intrahepatic bile duct. J Surg Oncol. 2006; 94:338–343. PMID: 16917866.

14. Kuroki T, Tajima Y, Kanematsu T. Hepatolithiasis and intrahepatic cholangiocarcinoma: carcinogenesis based on molecular mechanisms. J Hepatobiliary Pancreat Surg. 2005; 12:463–466. PMID: 16365819.

15. Uenishi T, Hamba H, Takemura S, Oba K, Ogawa M, Yamamoto T, et al. Outcomes of hepatic resection for hepatolithiasis. Am J Surg. 2009; 198:199–202. PMID: 19249730.

16. Cheon YK, Cho YD, Moon JH, Lee JS, Shim CS. Evaluation of long-term results and recurrent factors after operative and nonoperative treatment for hepatolithiasis. Surgery. 2009; 146:843–853. PMID: 19744434.

17. Lin CC, Lin PY, Chen YL. Comparison of concomitant and subsequent cholangiocarcinomas associated with hepatolithiasis: clinical implications. World J Gastroenterol. 2013; 19:375–380. PMID: 23372360.

18. Suzuki Y, Mori T, Abe N, Sugiyama M, Atomi Y. Predictive factors for cholangiocarcinoma associated with hepatolithiasis determined on the basis of Japanese Multicenter study. Hepatol Res. 2012; 42:166–170. PMID: 22151748.

19. Suzuki Y, Mori T, Yokoyama M, Nakazato T, Abe N, Nakanuma Y, et al. Hepatolithiasis: analysis of Japanese nationwide surveys over a period of 40 years. J Hepatobiliary Pancreat Sci. 2014; 21:617–622. PMID: 24824191.

20. Catena M, Aldrighetti L, Finazzi R, Arzu G, Arru M, Pulitanò C, et al. Treatment of non-endemic hepatolithiasis in a Western country. The role of hepatic resection. Ann R Coll Surg Engl. 2006; 88:383–389. PMID: 16834860.

21. Khan SA, Davidson BR, Goldin R, Pereira SP, Rosenberg WM, Taylor-Robinson SD, et al. Guidelines for the diagnosis and treatment of cholangiocarcinoma: consensus document. Gut. 2002; 51(Suppl 6):VI1–VI9. PMID: 12376491.

22. Dodson RM, Weiss MJ, Cosgrove D, Herman JM, Kamel I, Anders R, et al. Intrahepatic cholangiocarcinoma: management options and emerging therapies. J Am Coll Surg. 2013; 217:736–750.e4. PMID: 23890842.

23. Jan YY, Chen MF, Wang CS, Jeng LB, Hwang TL, Chen SC. Surgical treatment of hepatolithiasis: long-term results. Surgery. 1996; 120:509–514. PMID: 8784405.

24. Sato M, Watanabe Y, Horiuchi S, Nakata Y, Sato N, Kashu Y, et al. Long-term results of hepatic resection for hepatolithiasis. HPB Surg. 1995; 9:37–41. PMID: 8857452.

25. Otani K, Shimizu S, Chijiiwa K, Ogawa T, Morisaki T, Sugitani A, et al. Comparison of treatments for hepatolithiasis: hepatic resection versus cholangioscopic lithotomy. J Am Coll Surg. 1999; 189:177–182. PMID: 10437840.
26. Jeng KS, Yang FS, Ohta I, Chiang HJ. Dilatation of intrahepatic biliary strictures in patients with hepatolithiasis. World J Surg. 1990; 14:587–592. PMID: 2238657.

27. Jeng KS, Yang FS, Chiang HJ, Ohta I. Bile duct stents in the management of hepatolithiasis with long-segment intrahepatic biliary strictures. Br J Surg. 1992; 79:663–666. PMID: 1643480.

28. Nakanuma Y, Terada T, Tanaka Y, Ohta G. Are hepatolithiasis and cholangiocarcinoma aetiologically related? A morphological study of 12 cases of hepatolithiasis associated with cholangiocarcinoma. Virchows Arch A Pathol Anat Histopathol. 1985; 406:45–58. PMID: 2986349.
29. Rizvi S, Gores GJ. Pathogenesis, diagnosis, and management of cholangiocarcinoma. Gastroenterology. 2013; 145:1215–1229. PMID: 24140396.

30. Wakabayashi H, Akamoto S, Yachida S, Okano K, Izuishi K, Nishiyama Y, et al. Significance of fluorodeoxyglucose PET imaging in the diagnosis of malignancies in patients with biliary stricture. Eur J Surg Oncol. 2005; 31:1175–1179. PMID: 16019182.

31. Itatsu K, Zen Y, Ohira S, Ishikawa A, Sato Y, Harada K, et al. Immunohistochemical analysis of the progression of flat and papillary preneoplastic lesions in intrahepatic cholangiocarcinogenesis in hepatolithiasis. Liver Int. 2007; 27:1174–1184. PMID: 17919228.

32. Zen Y, Sasaki M, Fujii T, Chen TC, Chen MF, Yeh TS, et al. Different expression patterns of mucin core proteins and cytokeratins during intrahepatic cholangiocarcinogenesis from biliary intraepithelial neoplasia and intraductal papillary neoplasm of the bile duct--an immunohistochemical study of 110 cases of hepatolithiasis. J Hepatol. 2006; 44:350–358. PMID: 16360234.

33. Ohta T, Nagakawa T, Ueda N, Nakamura T, Akiyama T, Ueno K, et al. Mucosal dysplasia of the liver and the intraductal variant of peripheral cholangiocarcinoma in hepatolithiasis. Cancer. 1991; 68:2217–2223. PMID: 1655206.
34. Sun WB, Han BL, Cai JX. The surgical treatment of isolated left-sided hepatolithiasis: a 22-year experience. Surgery. 2000; 127:493–497. PMID: 10819056.

35. Midorikawa Y, Kubota K, Takayama T, Toyoda H, Ijichi M, Torzilli G, et al. A comparative study of postoperative complications after hepatectomy in patients with and without chronic liver disease. Surgery. 1999; 126:484–491. PMID: 10486600.

36. Fan ST, Lo CM, Liu CL, Lam CM, Yuen WK, Yeung C, et al. Hepatectomy for hepatocellular carcinoma: toward zero hospital deaths. Ann Surg. 1999; 229:322–330. PMID: 10077043.

Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download