INTRODUCTION
CASE DESCRIPTION
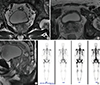 | Fig. 1Preoperative prostate MRI and bone scan images.
(A) Sagittal view; (B) horizontal view; (C) axial view of MRI of the prostate showing left side-dominant PC with extracapsular extension, seminal vesicle invasion, and enlarged left external and internal iliac lymph nodes; and (D) Disseminated bone metastasis in the whole axial and proximal appendicular bones.
MRI = magnetic resonance imaging, PC = prostate cancer.
|
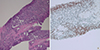 | Fig. 2Representative microphotographs of the prostate tumor.
(A) Characteristic features of Gleason patterns 4 and 5, including fused glands and an almost complete loss of glandular lumina (H & E staining, [× 100]). (B) Immunohistochemical staining of BRCA2 (× 200).
BRCA2 = breast cancer predisposition gene 2, H & E = hematoxylin and eosin.
|
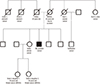 | Fig. 3Pedigree.
The patient's mother died from a car accident at 45 years of age. His first aunt died from ovarian cancer, and her son from biliary cancer. His second aunt died from colorectal cancer. The 2 daughters of his elder sister were diagnosed with breast cancer at 39 and 36 years of age, and their gene sequencing analyses revealed that both had BRCA2 mutation NM_000059.3:c.3744_3747delTGAG (p.Ser1248Argfs*10).
BRCA2 = breast cancer predisposition gene 2.
|




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download