Abstract
We investigated the effect of transforming growth factor beta 1 (TGF-β1) on equine hyaluronan synthase 2 (HAS2) gene expression and hyaluronan (HA) synthesis in culture models of articular chondrocytes. Equine chondrocytes were treated with TGF-β1 at different concentrations and times in monolayer cultures. In three-dimensional cultures, chondrocyte-seeded gelatin scaffolds were cultured in chondrogenic media containing 10 ng/mL of TGF-β1. The amounts of HA in conditioned media and in scaffolds were determined by enzyme-linked immunosorbent assays. HAS2 mRNA expression was analyzed by semi-quantitative reverse transcription polymerase chain reaction. The uronic acid content and DNA content of the scaffolds were measured by using colorimetric and Hoechst 33258 assays, respectively. Cell proliferation was evaluated by using the alamarBlue assay. Scanning electron microscopy (SEM), histology, and immunohistochemistry were used for microscopic analysis of the samples. The upregulation of HAS2 mRNA levels by TGF-β1 stimulation was dose and time dependent. TGF-β1 was shown to enhance HA and uronic acid content in the scaffolds. Cell proliferation and DNA content were significantly lower in TGF-β1 treatments. SEM and histological results revealed the formation of a cartilaginous-like extracellular matrix in the TGF-β1-treated scaffolds. Together, our results suggest that TGF-β1 has a stimulatory effect on equine chondrocytes, enhancing HA synthesis and promoting cartilage matrix generation.
Osteoarthritis (OA), a degenerative joint disease, results in the erosion of articular cartilage and can cause loss of joint function and chronic pain. Chondrocytes produce and maintain the cartilage matrix, which consists primarily of collagens, proteoglycans, and hyaluronan (HA). HA is a non-sulfated glycosaminoglycan composed of disaccharide units containing N-acetyl glucosamine and glucuronic acid. Together with proteoglycans and collagens, HA is responsible for the resilience of cartilage. When joint cartilage experiences excessive mechanical compressive loads, the components of the extracellular matrix (ECM) are degraded, resulting in cartilage degeneration and loss of function. Subsequently, healing of the cartilage occurs as the ECM is remodeled via interactions between chondrocytes and their environment, as stimulated by growth factors.
Transforming growth factor beta (TGF-β), an anabolic factor consisting of three isoforms (TGF-β1, TGF-β2, and TGF-β3), is involved in numerous biological processes [12]. In cartilage metabolism, TGF-β is involved in all stages of the genesis and proliferation of chondrocytes [26], including deposition of the ECM [22]. Numerous prior studies have reported an association between TGF-β and enhanced ECM production. In horses, TGF-β1 increased the synthesis and deposition of proteoglycan in cartilage explant, monolayer, and three-dimensional (3D) fibrin matrix cultures of foal chondrocytes [91112]. In equine chondrocyte cultures, TGF-β1 stimulated an increase in collagen type II mRNA [34]. However, documentation of TGF-β1–induced HA synthesis in equine chondrocytes is limited.
Three distinct hyaluronan synthase (HAS) genes (HAS1, HAS2, and HAS3) associated with HA production have been cloned from humans and mice [28]. Each isoform of the HAS gene has a different role in HA synthesis in vivo [2830]. There is abundant evidence of HA synthesis in response to TGF-β1 stimulation across several cell types and various species [163032]. In horses, an increase in the expression of HAS2 was accompanied by enhanced HA production (after human chorionic gonadotropin treatment) in the mural granulosa cells of preovulatory follicles [29]. To date, a report of HAS2 in HA synthesis in equine chondrocytes is rare.
In this study, we investigate the effects of TGF-β1 by examining HAS2 expression and HA synthesis in equine chondrocytes using both monolayer and 3D cultures. Furthermore, we evaluate whether the two culture systems provide an adequate model for the study of TGF-β1 and its effect on chondrocyte culture in horses. The results of this study contribute to our basic understanding of articular cartilage metabolism and can be applied to the development of tissue engineering as a treatment for equine OA.
All procedures and animal use in this study were approved by the Animal Care and Use Committee, Faculty of Veterinary Medicine, Chiang Mai University (FVM–ACUC; approval No. R9/2560).
Equine articular cartilage was obtained from the normal metacarpophalangeal joint of cadaver horse legs within 6 to 8 h after death. Equine chondrocytes were prepared for primary cell culture as previously described [13] with a modification of using fetal calf serum (FCS).
Passaged cells (2 × 105 per well) were seeded into 6-well plates and incubated with 10% FCS-Dulbecco's modified Eagle's medium (DMEM; Gibco, USA) for 48 h. The medium was then replaced by 10% FCS-DMEM containing the growth factor TGF-β1 (PeproTech Asia, Israel) at concentrations of 0, 2.5, 5, and 10 ng/mL. The 10% FCS-DMEM was used as a control. At day 3, the culture media were collected, and an enzyme-linked immunosorbent assay (ELISA) was used to determine the HA level.
Twenty-four hours prior to treatment, at 80% confluence, the cultured cells were starved and then treated with 2.5 ng/mL TGF-β1 at the indicated times (0, 3, 6, 9, 15, 24, 36, and 48 h) to evaluate the time-effect of TGF-β1 on HAS2 gene expression. Three-hour treatments with different concentrations of TGF-β1 (0, 2.5, 5, and 10 ng/mL) for 3 h were conducted to evaluate the dose-effect of TGF-β1 on HAS2 gene expression as assessed by using semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) [31].
Cells (1 × 104 per well) were seeded into 24-well plates and incubated with 10% FCS-DMEM containing TGF-β1 at concentrations of 0, 2.5, 5, and 10 ng/mL. Evaluations of cell proliferation were performed at days 0, 7, and 14 by using alamarBlue assays.
Gelatin-based scaffolds (Spongostan Standard; Johnson & Johnson, Germany) were cut into 5 mm × 2.5 mm circles, sterilized, and then presoaked with serum-free DMEM. Equine chondrocytes (1 × 106 cells) were resuspended in a final medium volume of 25 µL and then seeded onto the upper surface of the scaffold. The cellular scaffolds were incubated for 4 h to allow cell diffusion and attachment to the scaffolds, and then 1 mL of medium was added. After 24 h, each cellular scaffold was transferred to a blank 24-well plate to begin a 21 day culture. Each scaffold was replenished with 1.0 mL of chondrogenic media, 10% FCS-DMEM supplemented with insulin–transferrin–selenium (10 µg/mL insulin, 5.5 mg/mL transferrin, and 5 ng/mL selenium; PAA Laboratories, Austria), 25 µg/mL ascorbic acid, and 10-7 M dexamethasone [18]. Scaffolds were treated with or without TGF-β1. Chondrogenic media were changed every 2 to 3 days. Cell proliferation in the scaffold was measured at days 7, 14, and 21 by using alamarBlue assays. At day 21, the scaffolds were harvested for the determination of HA, uronic acid, and DNA contents and for scanning electron microscopy (SEM), histological, and HA immunohistochemical analyses.
Harvested scaffolds were digested with 2U papain at 60℃ prior to evaluating the amounts of HA, uronic acid, and DNA. The monolayer culture medium and the supernatant of papain-digested scaffolds were measured for HA by using ELISA as previously described [15]. The uronic acid and DNA contents in the supernatant of papain-digested scaffolds were assessed utilizing the carbazole method and the Hoechst 33258 dye assay, respectively, as described previously [720].
Expression of the HAS2 gene in monolayer culture cells was investigated by using the RT-PCR as previously described [35]. RNA was extracted from the cells using a NucleoSpin RNA II kit (Machere-Nagel, Germany). Total RNA was reverse transcribed into complementary DNA (cDNA) using the RevertAid First Strand cDNA synthesis kit (MBI Fermentas, Germany). Semi-quantitative PCR was performed by using Taq DNA polymerase (MBI Fermentas) with primers for equine glyceraldehydes-3-phosphate dehydrogenase (GAPDH). The upstream primer (5′-TGGTATCGTGGAAGGACTCAT-3′) and the downstream primer (5′-GTGGGTGTCGCTGTTGAAGTC-3′) were annealed at 55℃ for 1.15 min. For the equine HAS2, the upstream primer (5′-AGAGAAGTCATGTACACGG-CCTTC-3′) and the downstream primer (5′-GGTCTGCTGGTTTAACCATCTGAG-3′) were annealed at 57.8℃ for 1.15 min.
Cell viability and proliferation were evaluated from monolayer culture cells and cellular scaffolds by using the alamarBlue assay (Thermo Fisher Scientific, USA) according to the manufacturer's instructions.
The conditioned scaffolds were harvested from 21-day cultures, washed twice with phosphate-buffered saline (PBS), and immersed three times in PBS. The scaffolds were fixed overnight in 2.5% glutaraldehyde at pH 7.4 and 4℃ [37]. They were dehydrated, dried, and then sputter coated with gold particles at 40 mA prior to observing under SEM.
The tissue sections of harvested scaffolds at 21 days of 3D cultures were cut and stained with H&E. For immunohistochemical analysis of HA localization, endogenous peroxidase in the tissue sections was blocked with 3% H2O2, and non-specific sites were blocked with 3% bovine serum albumin (BSA). The sections were incubated with biotinylated-HA binding proteins (B-HABPs; a kind gifts from Prof. Dr. Prachya Kongtawelert, Chiang Mai University, Thailand) for 24 h and then incubated for 1 h using anti-biotin antibody conjugated to anti-biotin horseradish peroxidase (HRP; Sigma-Aldrich, USA). After PBS washing, 3, 3-diaminobenzidine (DAB; Sigma-Aldrich) solutions were gradually added to the section and the staining reaction was observed until the sections changed to a brown color. The slide was then mounted and covered with a coverslip.
All results are expressed as mean ± SEM from three to five independent experiments unless otherwise stated. The differences in means between TGF-β1 treatments and controls were analyzed using Student's t-test. Differences in means between control and experimental groups treated with various concentrations of TGF-β1 were analyzed by using ANOVA with Bonferroni adjustments as included in the R software (R Foundation for Statistical Computing, Austria). The level of significance was set at 0.05.
The TGF-β1 treatment resulted in a significant, concentration-dependent increase in the amount of HA in the conditioned media (panel A in Fig. 1). The effect of TGF-β1 was time dependent. Upregulation of HAS2 mRNA expression in equine chondrocytes treated with TGF-β1 reached the maximum level at 3 h, followed by the second significant difference in expression level at 9 h, and then a gradual decrease in expression after 15 h of stimulation (panel C in Fig. 1). HAS2 mRNA levels were upregulated in a dose-dependent manner. Either 5 or 10 ng/mL of TGF-β1 treatment resulted in significantly higher HAS2 mRNA levels than those of the control treatment (panel B in Fig. 1). Cell proliferation in the monolayer cultures gradually increased over 7 days. TGF-β1 treatments tended to activate cell proliferation at day 7 in a dose-dependent manner (Fig. 2).
The TGF-β1–treated scaffold had a watery pink appearance with a moister, glossier, and smoother surface than that of the untreated control (panel A in Fig. 3). At day 21, the accumulated levels of HA in the cultured media of TGF-β1–treated groups were significantly higher than those of the controls (panel B in Fig. 3). Compared with the control groups, TGF-β1–treated groups had significantly higher HA and uronic acid contents within the scaffold (panels C and D in Fig. 3). The equine chondrocytes gradually proliferated until day 14, with no further proliferation occurring through day 21 (panel A in Fig. 4). The numbers of chondrocytes on days 14 and 21 in TGF-β1–treated scaffolds were significantly lower than those in untreated control groups; the DNA content was also lower (Fig. 4).
Scanning electron micrographs of the free-cell and embedded scaffolds from the control and TGF-β1–treated groups are shown in Fig. 5. The untreated acellular scaffolds were highly porous and lacked ECM components. The microscopic consistency of the cellular scaffold treated with TGF-β1 was more compact with a dense continuous layer of hyaline cartilaginous-like tissue throughout the surface of the scaffold when compared with both the acellular and cellular scaffolds from the untreated control groups. The H&E staining of the TGF-β1–treated scaffolds showed the formation of a cartilaginous-like ECM containing chondrocytes at the scaffold surface (panels A–D in Fig. 6). The positive immunohistochemical staining of HA was more prominent in the TGF-β1–treated groups than in the control groups (panels E and F in Fig. 6), with strong immunolabeling of HA at the surface of the TGF-β1–treated scaffolds (panels G and H in Fig. 6).
TGF-β1 is known to stimulate ECM synthesis including type II collagen, aggrecan, and proteoglycan in chondrocytes [9]. In addition, TGF-β1 has a strong stimulatory effect on chondrogenic proliferation and differentiation in rabbits, horses, and humans [91922]. TGF-β1 has also been shown to stimulate HA synthesis [32]. In cartilage tissue, chondrocytes produce HA, a chief component of the ECM that is essential to the biological function of cartilage [24]. HAS, the membrane-bound enzyme responsible for the synthesis of HA, has been identified as three distinct isoforms: HAS1, HAS2, and HAS3 [27]. Each isoform is encoded by a separate gene, regulated by unique growth factors, and has a different role in HA production in vivo [2830]. HAS2 is the most abundant isoform that is expressed constitutively in articular chondrocytes in humans and animals. The HAS2 enzyme produces high molecular weight HA associated with maintenance of the cartilage matrix [1727].
This study focused on the influence of TGF-β1 on the expression of HAS2. Previous studies demonstrated that TGF-β1 upregulated HAS2 mRNA associated with HA production in mouse and human skin fibroblasts [30] and in human corneal cells [14]. In articular cartilage, HA synthesis by induction of the HAS2 gene in rabbit synovial fibroblasts was enhanced by TGF-β1 [32]. Similar to the present study, TGF-β1 upregulated HAS2 mRNA levels in a dose-dependent manner in equine chondrocytes, consistent with the increase in the amount of HA in both monolayer and 3D cultures. These results suggest that the HAS2 gene has a crucial role in the maintenance of HA homeostasis and in cartilage tissue formation.
In our study of equine chondrocyte cultures, HAS2 mRNA levels had increased after 3 h of TGF-β1 stimulation, with a second increase at 9 h and then a gradual decrease after 15 h. In human skin fibroblasts, HAS2 mRNA was detected as early as 2 h, reached maximum levels at 6 h, and then slightly decreased at 18 h after stimulation [30]. Similarly, another study reported that maximum levels of HAS2 mRNA occurred after 5 to 6 h of TGF-β1 exposure [3]. In rabbits, the level of HAS2 mRNA in synovial fibroblasts was enhanced and reached optimum levels at 6 h after TGF-β1 stimulation [32]. These findings suggest that the responsiveness of the HAS2 gene to TGF-β1 may differ by cell type. In our study, HAS2 mRNA expression was early detectable at 3 h after stimulation with TGF-β1 whereas measurements of the product (HA amount) should be taken at 24 h, similar to the results of a prior study of SW982 human synovial cells [35]. However, in our experiments, differences in the cumulative amount of HA between treatment and control groups were manifest over the course of 3 days. We found an increase in HAS2 mRNA expression within the first 9 h, indicating that the HAS2 gene was stimulated within a short time, whereas the expressed protein, HAS2 enzyme, was active around the plasma membrane, producing HA continuously over a longer period of time. Our findings are supported by a previous study that demonstrated that the HAS2 enzyme is localized at the plasma membrane and is expressed as a multipass transmembrane protein to export the HA polymers through the cell membrane into the extracellular space via ABC-transporters [25]. In our study, significant gene expression results could not be obtained if the gene expression analysis was performed later than 9 h after treatment, suggesting that studies of HAS2 regulation must include measurements at times throughout the experimental period. Thus, our findings suggest a fundamental experiment model that can be applied to future studies of gene expression and signaling mechanisms.
Our measurements of cell proliferation showed a time-dependent increase in the proliferation of chondrocytes in monolayer cultures over a period of 7 days. At day 7, the proliferation tended to be activated; however, there was no significant difference among various doses of TGF-β1. In 3D cultures, cell proliferation in both the control and TGF-β1–treated groups only increased in the second week and proliferation was then constant until the third week of culture. These results may suggest that the effect of TGF-β1 on cell proliferation depends on culture condition and duration. Interestingly, the proliferation of TGF-β1–treated groups in the last two weeks of the 3D cultures was significantly lower than proliferation in the control groups, consistent with the measurement results for DNA content. The decline of cell proliferation associated with TGF-β1 treatment may have resulted from the influence of this growth factor on redifferentiation rather than on proliferation when cultured in a 3D system. This interpretation is supported by a previous study that demonstrated that the 3D system facilitated the reversion of dedifferentiated cells to the chondrocyte phenotype [5]. Similarly, another study reported that human osteoarthritic fibroblast-like chondrocytes were able to redifferentiate when supplemented with TGF-β in 3D culture [1].
In the 3D cultures, the TGF-β1–treated groups showed increases in the amount of HA accumulated in media and the content of HA and uronic acid in the scaffold, as well as improved gross and microscopic appearances when compared with the controls; these results are consistent with a decrease in cell proliferation. Similarly, a previous study reported that human fibroblast-like chondrocytes redifferentiated when stimulated by TGF-β, as evidenced by enhanced expression of chondrogenic genes and elevated retention of newly synthesized proteoglycans and collagen [1]. Another study demonstrated that highly proliferated chondrocytes had lower production of glycosaminoglycans when compared with the lowly proliferated chondrocytes [10]. Our results, in conjunction with those of previous studies, indicate that TGF-β1 may promote differentiation of passaged equine chondrocytes to return to specialized form performing their functions in the synthesis of ECM. However, the gelatin-based scaffold used in 3D cultures in this study probably had a limited capacity for determining collagen synthesis.
In our study, the agreement between the significant immunolocalization of HA and the integrity of the gross appearance of TGF-β1–treated scaffolds suggests that TGF-β1 likely influences the synthesis of HA and other molecules simultaneously. However, there have been reports that gelatin scaffolds treated with exogenous HA promote the production of a cartilage matrix more than that by non-HA-treated scaffolds [23]. Our results, in conjunction with those of previous studies, indicate that HA is not only a major component of the cartilage matrix but also modulates the metabolism of other biomolecules.
The TGF-β1 applied in the present study was a recombinant human TGF-β1 protein that has been used in studies of equine tenocytes [2]. Our findings demonstrate that the human TGF-β1 is practical for the analysis of equine HAS2 gene expression and HA synthesis. In fact, the equine TGF-β1 amino acid sequence differs from that of other mammalian species by only two amino acids [36]. Surprisingly, the structure of equine TGF-β1 receptors TGF-βRI and TGF-βRII are 98% and 100% homologous, respectively, with human proteins [36]. The homology between the receptors of the two species suggests that equine TGF-β receptors could recognize human TGF-β1 [36]. Previous studies have reported that the TGF-β signaling pathway involves binding to TGF-β type I and type II receptors and the R-Smads/co-Smad complex in the regulation of target gene expression [33]. We postulate that a similar signaling pathway is involved in TGF-β1–stimulated HA synthesis in equine articular chondrocytes.
The main advantage of two-dimensional (2D) monolayer cultures is it provides a simpler model for cell-based studies. However, its limitations such as the inability to describe traits exhibited by in vivo systems (e.g., altered gene expression) have been increasingly recognized. Since almost all cells in the in vivo environment are surrounded by other cells and the ECM in a 3D fashion, 2D cell culture does not adequately take into account the natural 3D environment of cells [8]. Therefore, a 3D culture model using a scaffold provides an appropriate chondrocytes' microenvironment [8] facilitating the chondrocytes to maintain chondrogenic properties. Our observation of differences in cell proliferation in response to TGF-β1 between monolayer and 3D cultures agrees with a previous report that TGF-β1 enhanced the proliferation of bovine articular chondrocyte in monolayer cultures [6]. In contrast, the stimulatory effect of TGF-β1 decreased during chondrocyte subculture, especially in the late passages [4]. In our study of equine chondrocytes, TGF-β1 enhanced HA synthesis. This result parallels the increase in HA production in human synovial cell stimulated by TGF-β1 [16]. In contrast, an inhibitory effect of TGF-β1 on the synthesis of HA has been shown for human dermal fibroblasts [21]. TGF-β1 has been shown to activate DNA and proteoglycan synthesis in monolayer chondrocyte cultures for 7 days, whereas, on day 1, DNA and proteoglycan synthesis was inhibited in freshly isolated chondrocyte cultures [11]. These results suggest that the effect of TGF-β1 on chondrocyte biological activity depends on various parameters including culture type, culture condition and duration, presence or absence of serum in the medium, type of growth factor or cytokine, type and differentiated stage of cells, and age and species of the study animal.
In conclusion, we have clarified that TGF-β1 is capable of enhancing HA synthesis in both monolayer and 3D cultures of equine chondrocytes via upregulation of HAS2 mRNA expression. The present study demonstrates that monolayer cultures may not reflect the real physiological conditions of cartilage and chondrocytes as well as that obtained using 3D cultures. The findings of this study are applicable to future investigations of gene expression in other cell types and to studies of HA metabolism in other species. Our results are also relevant to future studies of tissue engineering as it is applied to the clinical treatment of equine OA and other joint diseases.
Figures and Tables
Fig. 1
Transforming growth factor beta 1 (TGF-β1) upregulated hyaluronan synthase 2 (HAS2) mRNA levels and enhanced hyaluronan (HA) synthesis in monolayer cultures of equine articular chondrocytes. Chondrocytes treated with different concentrations of TGF-β1 for 3 days (A) and 3 h (B). HA levels in the conditioned media and HAS2 mRNA levels were determined by enzyme-linked immunosorbent assay and semi-quantitative reverse transcription polymerase chain reaction (RT-PCR), respectively. (C) HAS2 mRNA levels determined by semi-quantitative RT-PCR, measured from chondrocytes cultured in the absence or presence of 2.5 ng/mL of TGF-β1 for the times shown. GAPDH, glyceraldehydes-3-phosphate dehydrogenase. Asterisks denote significant differences at *p < 0.05 and **p < 0.01.
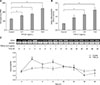
Fig. 2
Transforming growth factor beta 1 (TGF-β1) tended to increase cell proliferation in monolayer cultures of equine articular chondrocytes. Chondrocytes were treated with TGF-β1 in different concentrations. Cell proliferation was analyzed on days 0, 4, and 7 using the alamarBlue assay.

Fig. 3
Transforming growth factor beta 1 (TGF-β1) enhanced hyaluronan (HA) synthesis in three-dimensional cultures of equine articular chondrocytes. Chondrocytes embedded in scaffold were cultured for 21 days in the absence or presence of 10 ng/mL of TGF-β1. (A) Gross appearance of cultured scaffold at day 21. Scale bars = 5 mm. (B) Cumulative amount of HA in conditioned media was measured by enzyme-linked immunosorbent assay (ELISA) at indicated time intervals. The contents of HA (C) and uronic acid (D) in scaffold structures were determined at day 21 using ELISA and a colorimetric assay, respectively. Asterisks denote significant differences at *p < 0.05.
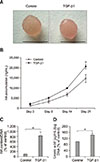
Fig. 4
Transforming growth factor beta 1 (TGF-β1) decreased cell proliferation and DNA content in three-dimensional cultures of equine articular chondrocytes. (A) Proliferation of equine chondrocytes was determined using the alamarBlue assay. (B) DNA content in scaffolds was analyzed based on two independent experiments using Hoechst dye 33258 assays. Cultures were maintained in the absence (control) or presence of 10 ng/mL TGF-β1. Asterisks denote significant differences at *p < 0.05.

Fig. 5
Transforming growth factor beta 1 (TGF-β1) enhanced extracellular matrix synthesis as visible in scanning electron micrographs of scaffolds from three-dimensional cultures of equine articular chondrocytes. The acellular scaffold has a highly porous appearance. Cellular scaffolds were cultured in the absence (control) or presence of 10 ng/mL of TGF-β1 for 21 days. 35× (upper), 1,000× (bottom).
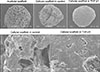
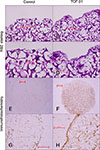
Fig. 6
Transforming growth factor beta 1 (TGF-β1) enhanced extracellular matrix and hyaluronan (HA) synthesis as shown by histological and immunohistochemical staining of scaffolds from three-dimensional cultures of equine articular chondrocytes. H&E staining and immunohistochemical localization of HA in scaffolds embedded with equine chondrocytes in the absence (A, C, E, and G) or presence (B, D, F, and H) of 10 ng/mL of TGF-β1 for 21 days. 200× (A and B), 400× (C, D, G, and H), 40× (E and F).
Acknowledgments
This research work was partially supported by CMU
Research Group Grant (2015) Chiang Mai University and
PERCH-CIC, Thailand. We are grateful to the Thailand
Excellence Center for Tissue Engineering and Stem Cells,
Department of Biochemistry, Faculty of Medicine, CMU for
general support throughout the research process.
References
1. Andrejevic K, Backstein D, Kandel R. TGF-beta 3 signaling in redifferentiating passaged human articular chondrocytes. Osteoarthritis Cartilage. 2017; 25:S168–S169.

2. Arai K, Kasashima Y, Kobayashi A, Kuwano A, Yoshihara T. TGF-β alters collagen XII and XIV mRNA levels in cultured equine tenocytes. Matrix Biol. 2002; 21:243–250.

3. Balazs EA, Watson D, Duff IF, Roseman S. Hyaluronic acid in synovial fluid. I. Molecular parameters of hyaluronic acid in normal and arthritis human fluids. Arthritis Rheum. 1967; 10:357–376.

4. Blanco FJ, Geng Y, Lotz M. Differentiation-dependent effects of IL-1 and TGF-beta on human articular chondrocyte proliferation are related to inducible nitric oxide synthase expression. J Immunol. 1995; 154:4018–4026.
5. Chaipinyo K, Oakes BW, van Damme MP. Effects of growth factors on cell proliferation and matrix synthesis of low-density, primary bovine chondrocytes cultured in collagen I gels. J Orthop Res. 2002; 20:1070–1078.

6. de Haart M, Marijnissen WJ, van Osch GJ, Verhaar JA. Optimization of chondrocyte expansion in culture. Effect of TGF beta-2, bFGF and L-ascorbic acid on bovine articular chondrocytes. Acta Orthop Scand. 1999; 70:55–61.

8. Edmondson R, Broglie JJ, Adcock AF, Yang L. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. Assay Drug Dev Technol. 2014; 12:207–218.

9. Fortier LA, Nixon AJ, Mohammed HO, Lust G. Altered biological activity of equine chondrocytes cultured in a three-dimensional fibrin matrix and supplemented with transforming growth factor beta-1. Am J Vet Res. 1997; 58:66–70.
10. Fukuhira Y, Kaneko H, Yamaga M, Tanaka M, Yamamoto S, Shimomura M. Effect of honeycomb-patterned structure on chondrocyte behavior in vitro. Colloids Surf A Physicochem Eng Asp. 2008; 313-314:520–525.

11. Glansbeek HL, van der Kraan PM, Vitters EL, van den Berg WB. Correlation of the size of type II transforming growth factor beta (TGF-beta) receptor with TGF-beta responses of isolated bovine articular chondrocytes. Ann Rheum Dis. 1993; 52:812–816.

12. Gordon KJ, Blobe GC. Role of transforming growth factor-β superfamily signaling pathways in human disease. Biochim Biophys Acta. 2008; 1782:197–228.

13. Gründer T, Gaissmaier C, Fritz J, Stoop R, Hortschansky P, Mollenhauer J, Aicher WK. Bone morphogenetic protein (BMP)-2 enhances the expression of type II collagen and aggrecan in chondrocytes embedded in alginate beads. Osteoarthritis Cartilage. 2004; 12:559–567.

14. Guo N, Li X, Mann MM, Funderburgh ML, Du Y, Funderburgh JL. Hyaluronan synthesis mediates the fibrotic response of keratocytes to transforming growth factor β. J Biol Chem. 2010; 285:32012–32019.

15. Hanprasertpong N, Teekachunhatean S, Chaiwongsa R, Ongchai S, Kunanusorn P, Sangdee C, Panthong A, Bunteang S, Nathasaen N, Reutrakul V. Analgesic, anti-inflammatory, and chondroprotective activities of Cryptolepis buchanani extract: in vitro and in vivo studies. Biomed Res Int. 2014; 2014:978582.
16. Haubeck HD, Kock R, Fischer DC, Van de Leur E, Hoffmeister K, Greiling H. Transforming growth factor β1, a major stimulator of hyaluronan synthesis in human synovial lining cells. Arthritis Rheum. 1995; 38:669–677.

17. Hiscock DRR, Caterson B, Flannery CR. Expression of hyaluronan synthases in articular cartilage. Osteoarthritis Cartilage. 2000; 8:120–126.

18. Hwang NS, Kim MS, Sampattavanich S, Baek JH, Zhang Z, Elisseeff J. Effects of three-dimensional culture and growth factors on the chondrogenic differentiation of murine embryonic stem cells. Stem Cells. 2006; 24:284–291.

19. Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998; 238:265–272.

20. Kim YJ, Sah RL, Doong JY, Grodzinsky AJ. Fluorometric assay of DNA in cartilage explants using Hoechst 33258. Anal Biochem. 1988; 174:168–176.

21. Li Y, Toole BP, Dealy CN, Kosher RA. Hyaluronan in limb morphogenesis. Dev Biol. 2007; 305:411–420.

22. Mackay AM, Beck SC, Murphy JM, Barry FP, Chichester CO, Pittenger MF. Chondrogenic differentiation of cultured human mesenchymal stem cells from marrow. Tissue Eng. 1998; 4:415–428.

23. Pruksakorn D, Khamwaen N, Pothacharoen P, Arpornchayanon O, Rojanasthien S, Kongtawelert P. Chondrogenic properties of primary human chondrocytes culture in hyaluronic acid treated gelatin scaffold. J Med Assoc Thai. 2009; 92:483–490.
24. Recklies AD, White C, Melching L, Roughley PJ. Differential regulation and expression of hyaluronan synthases in human articular chondrocytes, synovial cells and osteosarcoma cells. Biochem J. 2001; 354:17–24.

25. Schulz T, Schumacher U, Prehm P. Hyaluronan export by the ABC transporter MRP5 and its modulation by intracellular cGMP. J Biol Chem. 2007; 282:20999–21004.

26. Song JJ, Aswad R, Kanaan RA, Rico MC, Owen TA, Barbe MF, Safadi FF, Popoff SN. Connective tissue growth factor (CTGF) acts as a downstream mediator of TGF-β1 to induce mesenchymal cell condensation. J Cell Physiol. 2007; 210:398–410.

27. Spicer AP, McDonald JA. Characterization and molecular evolution of a vertebrate hyaluronan synthase gene family. J Biol Chem. 1998; 273:1923–1932.

28. Spicer AP, Olson JS, McDonald JA. Molecular cloning and characterization of a cDNA encoding the third putative mammalian hyaluronan synthase. J Biol Chem. 1997; 272:8957–8961.

29. Stock AE, Bouchard N, Brown K, Spicer AP, Underhill CB, Doré M, Sirois J. Induction of hyaluronan synthase 2 by human chorionic gonadotropin in mural granulosa cells of equine preovulatory follicles. Endocrinology. 2002; 143:4375–4384.

30. Sugiyama Y, Shimada A, Sayo T, Sakai S, Inoue S. Putative hyaluronan synthase mRNA are expressed in mouse skin and TGF-β upregulates their expression in cultured human skin cells. J Invest Dermatol. 1998; 110:116–121.

31. Tangyuenyong S, Viriyakhasem N, Peansukmanee S, Kongtawelert P, Ongchai S. Andrographolide exerts chondroprotective activity in equine cartilage explant and suppresses interleukin-1β-induced MMP-2 expression in equine chondrocyte culture. Int Sch Res Notices. 2014; 2014:464136.

32. Tanimoto K, Suzuki A, Ohno S, Honda K, Tanaka N, Doi T, Yoneno K, Ohno-Nakahara M, Nakatani Y, Ueki M, Tanne K. Effects of TGF-β on hyaluronan anabolism in fibroblasts derived from the synovial membrane of the rabbit temporomandibular joint. J Dent Res. 2004; 83:40–44.

33. ten Dijke P, Hill CS. New insights into TGF-β-Smad signalling. Trends Biochem Sci. 2004; 29:265–273.

34. Thompson CC, Clegg PD, Carter SD. Differential regulation of gelatinases by transforming growth factor beta-1 in normal equine chondrocytes. Osteoarthritis Cartilage. 2001; 9:325–331.

35. Viriyakhasem N, Khuajan S, Kongtawelert P, Panthong A, Ongchai S, Reutrakul V. In vitro model of hyaluronan synthase gene expression associated with lipopolysaccharide-induced inflammation in SW982 cell line. In Vitro Cell Dev Biol Anim. 2014; 50:787–791.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download