A 6-year-old boy visited emergency room of Seoul National University Children's Hospital during winter, complaining of respiratory difficulty. When he was an infant, he developed macrocytic hypochromic anemia. At that time, it was suspected that the anemia was due to a drug allergy. We tried to determine the cause of the patient's anemia but found no apparent cause, and the anemia spontaneously recovered. He subsequently suffered from atopic dermatitis and egg allergy, but he was lost to follow-up.
Three days before visiting the emergency room, the patient had a cough with sputum and a body temperature of 37.8°C. Within a day, his symptoms aggravated and he had difficulty in breathing. He was unable to sleep because of the cough. His parents also recently had a respiratory infection. When he arrived at the emergency room, the pulse oximetry was 68%. His blood pressure was 108/67 mmHg, pulse rate was 154 beats/min, respiratory rate was 52 breaths/min, and the body temperature was 37.4°C. Crackles and wheezing were audible in both lung fields. The lung sound of the right lung was weaker than that of the left lung.
Oxygen (15 L/min) was supplied via a facial mask with a reservoir bag, but the oxygen saturation did not rise above 70%. The white blood cell count was 32,280 cells/μL, of which 89.1% were segmented neutrophils and 4.5% were lymphocytes. Hemoglobin level was 12.4 g/dL. The platelet count was 526,000/μL. The level of C-reactive protein in blood was 3.16 mg/dL. His arterial blood gas analysis revealed that blood pH was 7.14; pCO
2 was 63 mmHg, pO
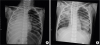
2 was 46 mmHg; and oxygen index was 35. Endotracheal intubation was performed immediately, and he was transferred to the pediatric emergency intensive care unit. The initial chest radiograph showed that the patient's right lung was totally collapsed (
Fig. 1A). Endotracheal tube adjustment and massive pulmonary physiotherapy were performed. The mechanical ventilator was adjusted to a higher setting which included an FiO
2 of 1.0, and empirical antibiotics (piperacillin/tazobactam, vancomycin) were administered. Oseltamivir was also administered because the patient fell ill during the flu epidemic season. Inhalation of nitric oxide was started. However, respiratory acidosis, hypercapnia, and hypoxemia remained persistent, with the arterial blood gas analysis showing that the blood pH was 7.09, pCO
2 was 86 mmHg, pO
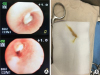
2 was 45 mmHg, and oxygenation index was 40. Thus, we performed veno-venous (from the right femoral to the right internal jugular vein) extracorporeal membrane oxygenation (VV ECMO) within 9 hours of the patient's emergency room arrival. Three hours after ECMO insertion, we performed flexible bronchoscopy. We found that thick mucus plugs were obstructing the right bronchus intermedius and upper lobe orifice (
Fig. 2). After removal of the thick mucus plugs, the right lung could be ventilated, and the tracheal deviation decreased, as observed using a chest radiograph (
Fig. 1B). The endotracheal aspiration samples tested negative on the flu rapid antigen test and the respiratory syncytial virus rapid antigen test. However, real-time multiplex polymerase chain reaction (PCR) detected the presence of influenza A virus in the endotracheal aspiration. There was no bacterial growth in blood, endotracheal and bronchoalveolar lavage cultures. The
Mycoplasma pneumonia antibody titer was 1:640, but PCR did not detect the presence of
M. pneumonia in endotracheal aspiration and bronchoalveolar lavage fluid. After 4 days, we could decrease the FiO
2 to 0.3 on the ventilator settings, and the oxygenation index was lower than 10 (
Fig. 3). We performed washing via bronchoscopy 2 more times due to persistent right upper lobe atelectasis. We stopped the VV ECMO on hospital day 6. The patient could breathe without a ventilator on hospital day 11, and was discharged without any sequelae on hospital day 19. After discharge, he was able to breathe normally and is currently undergoing regular follow-up.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download