Abstract
Emerging evidence demonstrates that the microbiota plays an essential role in shaping the development and function of host immune responses. A variety of environmental stimuli, including foods and commensals, are recognized by the host through the epithelium, acting as a physical barrier. Two allergic diseases, atopic dermatitis and food allergy, are closely linked to the microbiota, because inflammatory responses occur on the epidermal border. The microbiota generates metabolites such as short-chain fatty acids and poly-γ-glutamic acid (γ PGA), which can modulate host immune responses. Here, we review how microbial metabolites can regulate allergic immune responses. Furthermore, we focus on the effect of γ PGA on allergic T helper (Th) 2 responses and its therapeutic application.
Go to : 
References
1. Marenholz I, Grosche S, Kalb B, Ruschendorf F, Blumchen K, Schlags R, Harandi N, Price M, Hansen G, Seidenberg J, et al. Genome-wide association study identifies the SERPINB gene cluster as a susceptibility locus for food allergy. Nat Commun. 2017; 8:1056.

2. van Ginkel CD, Flokstra-de Blok BM, Kollen BJ, Kukler J, Koppelman GH, Dubois AE. Loss-of-function variants of the filaggrin gene are associated with clinical reactivity to foods. Allergy. 2015; 70:461–464.

4. Imai Y, Yasuda K, Sakaguchi Y, Haneda T, Mizutani H, Yoshimoto T, Nakanishi K, Yamanishi K. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc Natl Acad Sci U S A. 2013; 110:13921–13926.

5. Seneschal J, Clark RA, Gehad A, Baecher-Allan CM, Kupper TS. Human epidermal Langerhans cells maintain immune homeostasis in skin by activating skin resident regulatory T cells. Immunity. 2012; 36:873–884.

6. Otsuka A, Nakajima S, Kubo M, Egawa G, Honda T, Kitoh A, Nomura T, Hanakawa S, Sagita Moniaga C, Kim B, et al. Basophils are required for the induction of Th2 immunity to haptens and peptide antigens. Nat Commun. 2013; 4:1739.

7. Kim BS, Wang K, Siracusa MC, Saenz SA, Brestoff JR, Monticelli LA, Noti M, Tait Wojno ED, Fung TC, Kubo M, et al. Basophils promote innate lymphoid cell responses in inflamed skin. J Immunol. 2014; 193:3717–3725.

8. Roediger B, Kyle R, Yip KH, Sumaria N, Guy TV, Kim BS, Mitchell AJ, Tay SS, Jain R, Forbes-Blom E, et al. Cutaneous immunosurveillance and regulation of inflammation by group 2 innate lymphoid cells. Nat Immunol. 2013; 14:564–573.

9. La Cava A, Van Kaer L, Shi FD. CD4+CD25+ Tregs and NKT cells: regulators regulating regulators. Trends Immunol. 2006; 27:322–327.

10. Lee YJ, Holzapfel KL, Zhu J, Jameson SC, Hogquist KA. Steady-state production of IL-4 modulates immunity in mouse strains and is determined by lineage diversity of iNKT cells. Nat Immunol. 2013; 14:1146–1154.

11. Kwon DI, Lee YJ. Lineage differentiation program of invariant natural killer t cells. Immune Netw. 2017; 17:365–377.

12. Wu WH, Park CO, Oh SH, Kim HJ, Kwon YS, Bae BG, Noh JY, Lee KH. Thymic stromal lymphopoietin-activated invariant natural killer T cells trigger an innate allergic immune response in atopic dermatitis. J Allergy Clin Immunol. 2010; 126:290–299. 299.e1–4.

13. Frossard CP, Zimmerli SC, Rincon Garriz JM, Eigenmann PA. Food allergy in mice is modulated through the thymic stromal lymphopoietin pathway. Clin Transl Allergy. 2016; 6:2.

14. Lee JB, Chen CY, Liu B, Mugge L, Angkasekwinai P, Facchinetti V, Dong C, Liu YJ, Rothenberg ME, Hogan SP, et al. IL-25 and CD4 (+) TH2 cells enhance type 2 innate lymphoid cell-derived IL-13 production, which promotes IgE-mediated experimental food allergy. J Allergy Clin Immunol. 2016; 137:1216–1225. e5.
15. Noval Rivas M, Burton OT, Wise P, Charbonnier LM, Georgiev P, Oettgen HC, Rachid R, Chatila TA. Regulatory T cell reprogramming toward a Th2-cell-like lineage impairs oral tolerance and promotes food allergy. Immunity. 2015; 42:512–523.

16. Gri G, Piconese S, Frossi B, Manfroi V, Merluzzi S, Tripodo C, Viola A, Odom S, Rivera J, Colombo MP, et al. CD4+CD25+ regulatory T cells suppress mast cell degranulation and allergic responses through OX40-OX40L interaction. Immunity. 2008; 29:771–781.

17. Torgerson TR, Linane A, Moes N, Anover S, Mateo V, Rieux-Laucat F, Hermine O, Vijay S, Gambineri E, Cerf-Bensussan N, et al. Severe food allergy as a variant of IPEX syndrome caused by a deletion in a noncoding region of the FOXP3 gene. Gastroenterology. 2007; 132:1705–1717.

18. Qamar N, Fishbein AB, Erickson KA, Cai M, Szychlinski C, Bryce PJ, Schleimer RP, Fuleihan RL, Singh AM. Naturally occurring tolerance acquisition to foods in previously allergic children is characterized by antigen specificity and associated with increased subsets of regulatory T cells. Clin Exp Allergy. 2015; 45:1663–1672.

19. Noval Rivas M, Burton OT, Oettgen HC, Chatila T. IL-4 production by group 2 innate lymphoid cells promotes food allergy by blocking regulatory T-cell function. J Allergy Clin Immunol. 2016; 138:801–811. e9.

21. Gong JQ, Lin L, Lin T, Hao F, Zeng FQ, Bi ZG, Yi D, Zhao B. Skin colonization by Staphylococcus aureus in patients with eczema and atopic dermatitis and relevant combined topical therapy: a double-blind multicentre randomized controlled trial. Br J Dermatol. 2006; 155:680–687.

22. Byrd AL, Deming C, Cassidy SK, Harrison OJ, Ng WI, Conlan S, Belkaid Y, Segre JA, Kong HH. Staphylococcus aureus and Staphylococcus epidermidis strain diversity underlying pediatric atopic dermatitis. Sci Transl Med. 2017; 9:eaal4651.
23. Nakatsuji T, Chen TH, Narala S, Chun KA, Two AM, Yun T, Shafiq F, Kotol PF, Bouslimani A, Melnik AV, et al. Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis. Sci Transl Med. 2017; 9:eaah4680.
24. Kocianova S, Vuong C, Yao Y, Voyich JM, Fischer ER, DeLeo FR, Otto M. Key role of poly-gamma-DL-glutamic acid in immune evasion and virulence of Staphylococcus epidermidis. J Clin Invest. 2005; 115:688–694.

25. Inbaraj BS, Kao TH, Tsai TY, Chiu CP, Kumar R, Chen BH. The synthesis and characterization of poly(gamma-glutamic acid)-coated magnetite nanoparticles and their effects on antibacterial activity and cytotoxicity. Nanotechnology. 2011; 22:075101.
26. Linehan JL, Harrison OJ, Han SJ, Byrd AL, Vujkovic-Cvijin I, Villarino AV, Sen SK, Shaik J, Smelkinson M, Tamoutounour S, et al. Non-classical immunity controls microbiota impact on skin immunity and tissue repair. Cell. 2018; 172:784–796. e18.

27. Lai Y, Di Nardo A, Nakatsuji T, Leichtle A, Yang Y, Cogen AL, Wu ZR, Hooper LV, Schmidt RR, von Aulock S, et al. Commensal bacteria regulate Toll-like receptor 3-dependent inflammation after skin injury. Nat Med. 2009; 15:1377–1382.

28. Kurashima Y, Amiya T, Fujisawa K, Shibata N, Suzuki Y, Kogure Y, Hashimoto E, Otsuka A, Kabashima K, Sato S, et al. The enzyme Cyp26b1 mediates inhibition of mast cell activation by fibroblasts to maintain skin-barrier homeostasis. Immunity. 2014; 40:530–541.

29. Nakamura Y, Oscherwitz J, Cease KB, Chan SM, Munoz-Planillo R, Hasegawa M, Villaruz AE, Cheung GY, McGavin MJ, Travers JB, et al. Staphylococcus delta-toxin induces allergic skin disease by activating mast cells. Nature. 2013; 503:397–401.

30. Ando T, Matsumoto K, Namiranian S, Yamashita H, Glatthorn H, Kimura M, Dolan BR, Lee JJ, Galli SJ, Kawakami Y, et al. Mast cells are required for full expression of allergen/SEB-induced skin inflammation. J Invest Dermatol. 2013; 133:2695–2705.

31. Sanford JA, Zhang LJ, Williams MR, Gangoiti JA, Huang CM, Gallo RL. Inhibition of HDAC8 and HDAC9 by microbial short-chain fatty acids breaks immune tolerance of the epidermis to TLR ligands. Sci Immunol. 2016; 1:eaah4609.

32. Kao MS, Huang S, Chang WL, Hsieh MF, Huang CJ, Gallo RL, Huang CM. Microbiome precision editing: Using PEG as a selective fermentation initiator against methicillin-resistant Staphylococcus aureus. Biotechnol J. 2017; 12:1600399.
33. Ling Z, Li Z, Liu X, Cheng Y, Luo Y, Tong X, Yuan L, Wang Y, Sun J, Li L, et al. Altered fecal microbiota composition associated with food allergy in infants. Appl Environ Microbiol. 2014; 80:2546–2554.

34. Diesner SC, Bergmayr C, Pfitzner B, Assmann V, Krishnamurthy D, Starkl P, Endesfelder D, Rothballer M, Welzl G, Rattei T, et al. A distinct microbiota composition is associated with protection from food allergy in an oral mouse immunization model. Clin Immunol. 2016; 173:10–18.

35. Stefka AT, Feehley T, Tripathi P, Qiu J, McCoy K, Mazmanian SK, Tjota MY, Seo GY, Cao S, Theriault BR, et al. Commensal bacteria protect against food allergen sensitization. Proc Natl Acad Sci U S A. 2014; 111:13145–13150.

36. Tan J, McKenzie C, Vuillermin PJ, Goverse G, Vinuesa CG, Mebius RE, Macia L, Mackay CR. Dietary fiber and bacterial SCFA enhance oral tolerance and protect against food allergy through diverse cellular pathways. Cell Rep. 2016; 15:2809–2824.

37. Kelleher M, Dunn-Galvin A, Hourihane JO, Murray D, Campbell LE, McLean WH, Irvine AD. Skin barrier dysfunction measured by transepidermal water loss at 2 days and 2 months predates and predicts atopic dermatitis at 1 year. J Allergy Clin Immunol. 2015; 135:930–935. e1.
38. Eigenmann PA, Sicherer SH, Borkowski TA, Cohen BA, Sampson HA. Prevalence of IgE-mediated food allergy among children with atopic dermatitis. Pediatrics. 1998; 101:E8.

39. Strid J, Hourihane J, Kimber I, Callard R, Strobel S. Epicutaneous exposure to peanut protein prevents oral tolerance and enhances allergic sensitization. Clin Exp Allergy. 2005; 35:757–766.

40. Wilson SR, The L, Batia LM, Beattie K, Katibah GE, McClain SP, Pellegrino M, Estandian DM, Bautista DM. The epithelial cell-derived atopic dermatitis cytokine TSLP activates neurons to induce itch. Cell. 2013; 155:285–295.

41. Han H, Xu W, Headley MB, Jessup HK, Lee KS, Omori M, Comeau MR, Marshak-Rothstein A, Ziegler SF. Thymic stromal lymphopoietin (TSLP)-mediated dermal inflammation aggravates experimental asthma. Mucosal Immunol. 2012; 5:342–351.

42. Han H, Thelen TD, Comeau MR, Ziegler SF. Thymic stromal lymphopoietin-mediated epicutaneous inflammation promotes acute diarrhea and anaphylaxis. J Clin Invest. 2014; 124:5442–5452.

43. Noti M, Kim BS, Siracusa MC, Rak GD, Kubo M, Moghaddam AE, Sattentau QA, Comeau MR, Spergel JM, Artis D. Exposure to food allergens through inflamed skin promotes intestinal food allergy through the thymic stromal lymphopoietin-basophil axis. J Allergy Clin Immunol. 2014; 133:1390–1399. 1399.e1–6.

44. Ma L, Xue HB, Guan XH, Shu CM, Zhang JH, Yu J. Possible pathogenic role of T helper type 9 cells and interleukin (IL)-9 in atopic dermatitis. Clin Exp Immunol. 2014; 175:25–31.

45. Sismanopoulos N, Delivanis DA, Alysandratos KD, Angelidou A, Vasiadi M, Therianou A, Theoharides TC. IL-9 induces VEGF secretion from human mast cells and IL-9/IL-9 receptor genes are overexpressed in atopic dermatitis. PLoS One. 2012; 7:e33271.

46. Chen CY, Lee JB, Liu B, Ohta S, Wang PY, Kartashov AV, Mugge L, Abonia JP, Barski A, Izuhara K, et al. Induction of interleukin-9-producing mucosal mast cells promotes susceptibility to IgE-mediated experimental food allergy. Immunity. 2015; 43:788–802.

47. Yao W, Zhang Y, Jabeen R, Nguyen ET, Wilkes DS, Tepper RS, Kaplan MH, Zhou B. Interleukin-9 is required for allergic airway inflammation mediated by the cytokine TSLP. Immunity. 2013; 38:360–372.

48. Lee JB. Regulation of IgE-mediated food allergy by IL-9 producing mucosal mast cells and type 2 innate lymphoid cells. Immune Netw. 2016; 16:211–218.

49. Brough HA, Cousins DJ, Munteanu A, Wong YF, Sudra A, Makinson K, Stephens AC, Arno M, Ciortuz L, Lack G, et al. IL-9 is a key component of memory TH cell peanut-specific responses from children with peanut allergy. J Allergy Clin Immunol. 2014; 134:1329–1338. e10.

50. Sehra S, Yao W, Nguyen ET, Glosson-Byers NL, Akhtar N, Zhou B, Kaplan MH. TH9 cells are required for tissue mast cell accumulation during allergic inflammation. J Allergy Clin Immunol. 2015; 136:433–440. e1.

51. Matsune S, Ohori J, Yoshifuku K, Kurono Y. Effect of vascular endothelial growth factor on nasal vascular permeability. Laryngoscope. 2010; 120:844–848.

52. Lee J, Seto D, Bielory L. Meta-analysis of clinical trials of probiotics for prevention and treatment of pediatric atopic dermatitis. J Allergy Clin Immunol. 2008; 121:116–121. e11.

53. Takahashi N, Akahoshi M, Matsuda A, Ebe K, Inomata N, Obara K, Hirota T, Nakashima K, Shimizu M, Tamari M, et al. Association of the IL12RB1 promoter polymorphisms with increased risk of atopic dermatitis and other allergic phenotypes. Hum Mol Genet. 2005; 14:3149–3159.

54. Namkung JH, Lee JE, Kim E, Kim S, Shin ES, Cho EY, Yang JM. Association of single nucleotide polymorphisms in the IL-12 (IL-12A and B) and IL-12 receptor (IL-12Rbeta1 and beta2) genes and gene-gene interactions with atopic dermatitis in Koreans. J Dermatol Sci. 2010; 57:199–206.

55. Itazawa T, Adachi Y, Okabe Y, Hamamichi M, Adachi YS, Toyoda M, Morohashi M, Miyawaki T. Developmental changes in interleukin-12-producing ability by monocytes and their relevance to allergic diseases. Clin Exp Allergy. 2003; 33:525–530.

56. Park ST, Kim KE, Na K, Kim Y, Kim TY. Effect of dendritic cells treated with CpG ODN on atopic dermatitis of Nc/Nga mice. J Biochem Mol Biol. 2007; 40:486–493.

57. Kim CH, Park CD, Lee AY. Administration of poly(I:C) improved dermatophagoides farinae-induced atopic dermatitis-like skin lesions in NC/Nga mice by the regulation of Th1/Th2 balance. Vaccine. 2012; 30:2405–2410.

58. Hattori K, Nishikawa M, Watcharanurak K, Ikoma A, Kabashima K, Toyota H, Takahashi Y, Takahashi R, Watanabe Y, Takakura Y. Sustained exogenous expression of therapeutic levels of IFN-gamma ameliorates atopic dermatitis in NC/Nga mice via Th1 polarization. J Immunol. 2010; 184:2729–2735.

59. Panahi Y, Davoudi SM, Madanchi N, Abolhasani E. Recombinant human interferon gamma (Gamma Immunex) in treatment of atopic dermatitis. Clin Exp Med. 2012; 12:241–245.

60. Lee JH, Noh G, Noh J, Lee S, Choi WS, Kim HS, Lee K, Choi S, Jin H, Cho S. Clinical characteristics of oral tolerance induction of IgE-mediated and non-IgE-mediated food allergy using interferon gamma. Allergy Asthma Proc. 2010; 31:e39–e47.

61. Noh G, Jang EH. Dual specific oral tolerance induction using interferon gamma for IgE-mediated anaphylactic food allergy and the dissociation of local skin allergy and systemic oral allergy: tolerance or desensitization? J Investig Allergol Clin Immunol. 2014; 24:87–97.
62. Noh J, Noh G, Lee SJ, Lee JH, Kim A, Kim HS, Choi WS. Tolerogenic effects of interferon-gamma with induction of allergen-specific interleukin-10-producing regulatory B cell (Br1) changes in non-IgE-mediated food allergy. Cell Immunol. 2012; 273:140–149.

63. Mann-Chandler MN, Kashyap M, Wright HV, Norozian F, Barnstein BO, Gingras S, Parganas E, Ryan JJ. IFN-gamma induces apoptosis in developing mast cells. J Immunol. 2005; 175:3000–3005.
64. Luttmann W, Dauer E, Schmidt S, Marx O, Hossfeld M, Matthys H, Virchow JC Jr. Effects of interferon-gamma and tumour necrosis factor-alpha on CD95/Fas ligand-mediated apoptosis in human blood eosinophils. Scand J Immunol. 2000; 51:54–59.

65. de Bruin AM, Buitenhuis M, van der Sluijs KF, van Gisbergen KP, Boon L, Nolte MA. Eosinophil differentiation in the bone marrow is inhibited by T cell-derived IFN-gamma. Blood. 2010; 116:2559–2569.

66. Moore ML, Newcomb DC, Parekh VV, Van Kaer L, Collins RD, Zhou W, Goleniewska K, Chi MH, Mitchell D, Boyce JA, et al. STAT1 negatively regulates lung basophil IL-4 expression induced by respiratory syncytial virus infection. J Immunol. 2009; 183:2016–2026.

67. Duerr CU, McCarthy CD, Mindt BC, Rubio M, Meli AP, Pothlichet J, Eva MM, Gauchat JF, Qureshi ST, Mazer BD, et al. Type I interferon restricts type 2 immunopathology through the regulation of group 2 innate lymphoid cells. Nat Immunol. 2016; 17:65–75.

68. Kudo F, Ikutani M, Seki Y, Otsubo T, Kawamura YI, Dohi T, Oshima K, Hattori M, Nakae S, Takatsu K, et al. Interferon-gamma constrains cytokine production of group 2 innate lymphoid cells. Immunology. 2016; 147:21–29.

69. Molofsky AB, Van Gool F, Liang HE, Van Dyken SJ, Nussbaum JC, Lee J, Bluestone JA, Locksley RM. Interleukin-33 and interferon-gamma counter-regulate group 2 innate lymphoid cell activation during immune perturbation. Immunity. 2015; 43:161–174.

70. Han M, Hong JY, Jaipalli S, Rajput C, Lei J, Hinde JL, Chen Q, Hershenson NM, Bentley JK, Hershenson MB. IFN-gamma blocks development of an asthma phenotype in rhinovirus-infected baby mice by inhibiting type 2 innate lymphoid cells. Am J Respir Cell Mol Biol. 2017; 56:242–251.
71. Murugaiyan G, Beynon V, Pires Da Cunha A, Joller N, Weiner HL. IFN-gamma limits Th9-mediated autoimmune inflammation through dendritic cell modulation of IL-27. J Immunol. 2012; 189:5277–5283.

72. Kaikiri H, Miyamoto J, Kawakami T, Park SB, Kitamura N, Kishino S, Yonejima Y, Hisa K, Watanabe J, Ogita T, et al. Supplemental feeding of a gut microbial metabolite of linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, alleviates spontaneous atopic dermatitis and modulates intestinal microbiota in NC/nga mice. Int J Food Sci Nutr. 2017; 68:941–951.

73. Schwarz A, Bruhs A, Schwarz T. The short-chain fatty acid sodium butyrate functions as a regulator of the skin immune system. J Invest Dermatol. 2017; 137:855–864.

74. Lee TY, Kim YH, Yoon SW, Choi JC, Yang JM, Kim CJ, Schiller JT, Sung MH, Poo H. Oral administration of poly-gamma-glutamate induces TLR4- and dendritic cell-dependent antitumor effect. Cancer Immunol Immunother. 2009; 58:1781–1794.

75. Kim S, Yang JY, Lee K, Oh KH, Gi M, Kim JM, Paik DJ, Hong S, Youn J. Bacillus subtilis-specific poly-gamma-glutamic acid regulates development pathways of naive CD4 (+) T cells through antigen-presenting cell-dependent and -independent mechanisms. Int Immunol. 2009; 21:977–990.

76. Lee SW, Park HJ, Park SH, Kim N, Hong S. Immunomodulatory effect of poly-gamma-glutamic acid derived from Bacillus subtilis on natural killer dendritic cells. Biochem Biophys Res Commun. 2014; 443:413–421.

77. Lee K, Kim SH, Yoon HJ, Paik DJ, Kim JM, Youn J. Bacillus-derived poly-gamma-glutamic acid attenuates allergic airway inflammation through a Toll-like receptor-4-dependent pathway in a murine model of asthma. Clin Exp Allergy. 2011; 41:1143–1156.

78. Lee SW, Park HJ, Park SH, Hong S. Oral administration of poly-gamma-glutamic acid prevents the development of atopic dermatitis in NC/Nga mice. Exp Dermatol. 2013; 22:561–563.

79. Park HJ, Lee SW, Park SH, Hong S. iNKT cells are responsible for the apoptotic reduction of basophils that mediate Th2 immune responses elicited by papain in mice following gammaPGA stimulation. PLoS One. 2016; 11:e0152189.
80. Katsuta M, Takigawa Y, Kimishima M, Inaoka M, Takahashi R, Shiohara T. NK cells and gamma delta+ T cells are phenotypically and functionally defective due to preferential apoptosis in patients with atopic dermatitis. J Immunol. 2006; 176:7736–7744.

81. Brown AF, Murphy AG, Lalor SJ, Leech JM, O'Keeffe KM, Mac Aogain M, O'Halloran DP, Lacey KA, Tavakol M, Hearnden CH, et al. Memory Th1 cells are protective in invasive Staphylococcus aureus infection. PLoS Pathog. 2015; 11:e1005226.
82. Naik S, Bouladoux N, Wilhelm C, Molloy MJ, Salcedo R, Kastenmuller W, Deming C, Quinones M, Koo L, Conlan S, et al. Compartmentalized control of skin immunity by resident commensals. Science. 2012; 337:1115–1119.

83. Tanaka A, Jung K, Benyacoub J, Prioult G, Okamoto N, Ohmori K, Blum S, Mercenier A, Matsuda H. Oral supplementation with Lactobacillus rhamnosus CGMCC 1.3724 prevents development of atopic dermatitis in NC/NgaTnd mice possibly by modulating local production of IFN-gamma. Exp Dermatol. 2009; 18:1022–1027.

84. Prescott SL, Dunstan JA, Hale J, Breckler L, Lehmann H, Weston S, Richmond P. Clinical effects of probiotics are associated with increased interferon-gamma responses in very young children with atopic dermatitis. Clin Exp Allergy. 2005; 35:1557–1564.

85. Shalapour S, Deiser K, Sercan O, Tuckermann J, Minnich K, Willimsky G, Blankenstein T, Hammerling GJ, Arnold B, Schuler T. Commensal microflora and interferon-gamma promote steady-state interleukin-7 production in vivo. Eur J Immunol. 2010; 40:2391–2400.

86. Nakajima S, Igyártó BZ, Honda T, Egawa G, Otsuka A, Hara-Chikuma M, Watanabe N, Ziegler SF, Tomura M, Inaba K, et al. Langerhans cells are critical in epicutaneous sensitization with protein antigen via thymic stromal lymphopoietin receptor signaling. J Allergy Clin Immunol. 2012; 129:1048–1055.e6.

87. Martin R, Nauta AJ, Ben Amor K, Knippels LM, Knol J, Garssen J. Early life: gut microbiota and immune development in infancy. Benef Microbes. 2010; 1:367–382.

88. Scharschmidt TC, Vasquez KS, Truong HA, Gearty SV, Pauli ML, Nosbaum A, Gratz IK, Otto M, Moon JJ, Liese J, et al. A wave of regulatory t cells into neonatal skin mediates tolerance to commensal microbes. Immunity. 2015; 43:1011–1021.

89. Penders J, Thijs C, van den Brandt PA, Kummeling I, Snijders B, Stelma F, Adams H, van Ree R, Stobberingh EE. Gut microbiota composition and development of atopic manifestations in infancy: the KOALA Birth Cohort Study. Gut. 2007; 56:661–667.

90. Wickens K, Black PN, Stanley TV, Mitchell E, Fitzharris P, Tannock GW, Purdie G, Crane J. A differential effect of 2 probiotics in the prevention of eczema and atopy: a double-blind, randomized, placebo-controlled trial. J Allergy Clin Immunol. 2008; 122:788–794.

91. Prakoeswa CR, Herwanto N, Prameswari R, Astari L, Sawitri S, Hidayati AN, Indramaya DM, Kusumowidagdo ER, Surono IS. Lactobacillus plantarum IS-10506 supplementation reduced SCORAD in children with atopic dermatitis. Benef Microbes. 2017; 8:833–840.

92. Kwon HK, Lee CG, So JS, Chae CS, Hwang JS, Sahoo A, Nam JH, Rhee JH, Hwang KC, Im SH. Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc Natl Acad Sci U S A. 2010; 107:2159–2164.

93. Shin JH, Chung MJ, Seo JG. A multistrain probiotic formulation attenuates skin symptoms of atopic dermatitis in a mouse model through the generation of CD4 (+)Foxp3 (+) T cells. Food Nutr Res. 2016; 60:32550.

94. Ren C, Zhang Q, de Haan BJ, Zhang H, Faas MM, de Vos P. Identification of TLR2/TLR6 signalling lactic acid bacteria for supporting immune regulation. Sci Rep. 2016; 6:34561.

95. Fu L, Peng J, Zhao S, Zhang Y, Su X, Wang Y. Lactic acid bacteria-specific induction of CD4 (+)Foxp3 (+) T cells ameliorates shrimp tropomyosin-induced allergic response in mice via suppression of mTOR signaling. Sci Rep. 2017; 7:1987.
96. Kluger MA, Nosko A, Ramcke T, Goerke B, Meyer MC, Wegscheid C, Luig M, Tiegs G, Stahl RA, Steinmetz OM. RORgammat expression in Tregs promotes systemic lupus erythematosus via IL-17 secretion, alteration of Treg phenotype and suppression of Th2 responses. Clin Exp Immunol. 2017; 188:63–78.

97. Jung MK, Kwak JE, Shin EC. IL-17A-producing Foxp3 (+) regulatory T cells and human diseases. Immune Netw. 2017; 17:276–286.

98. Cortes-Perez NG, Lozano-Ojalvo D, Maiga MA, Hazebrouck S, Adel-Patient K. Intragastric administration of Lactobacillus casei BL23 induces regulatory FoxP3+RORgammat+ T cells subset in mice. Benef Microbes. 2017; 8:433–438.

99. Hacini-Rachinel F, Gheit H, Le Luduec JB, Dif F, Nancey S, Kaiserlian D. Oral probiotic control skin inflammation by acting on both effector and regulatory T cells. PLoS One. 2009; 4:e4903.

100. Jin HE, Choi JC, Lim YT, Sung MH. Prebiotic effects of poly-gamma-glutamate on bacterial flora in murine gut. J Microbiol Biotechnol. 2017; 27:412–415.

Go to : 
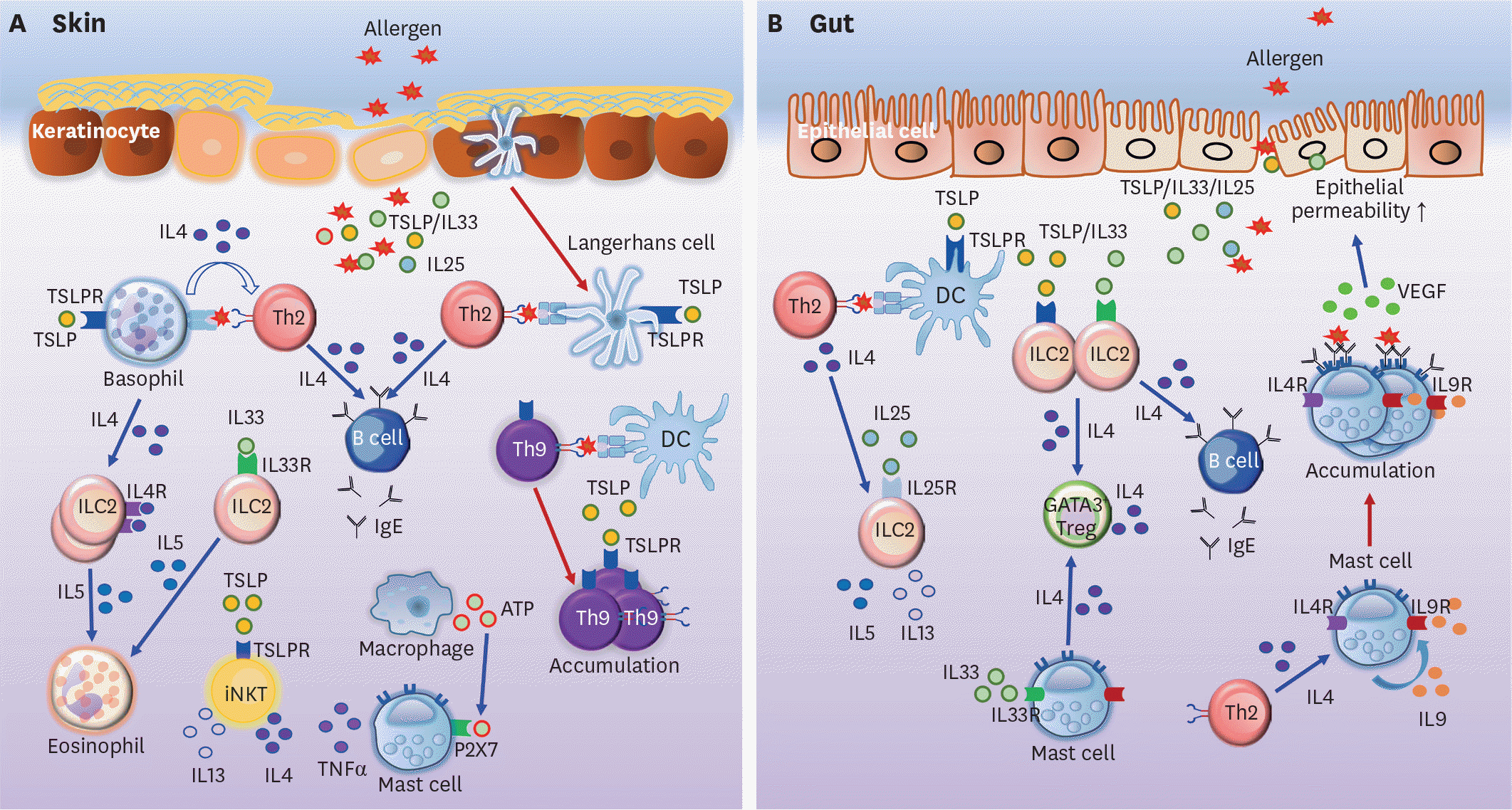 | Figure 1.Allergic immune responses in the skin and gut. (A) Basophils and LCs activated by lesional skin-derived TSLP induce the Th2 differentiation of allergen-specific CD4+ T cells. Th2 cells stimulate B cells to switch, and to thus produce IgE. Basophil-derived IL4 can induce IL5 production, promoting the accumulation of eosinophils in the skin. Besides, IL5 produced by IL33-activated ILC2s recruits eosinophils into the skin. Th9 cells induced by DCs accumulate under TSLP stimulation in the skin. TSLP also promotes iNKT cells to secrete IL4 and IL13. Furthermore, macrophage-derived ATP causes the release of TNFα by mast cells via P2X7 signaling, consequently resulting in skin inflammation. (B) TSLP triggers DCs to induce naive CD4+ T cell differentiation into Th2 cells. Th2 cells and IL25 stimulate ILC2s to secrete IL5 and IL13. IL4 produced by IL33-activated ILC2s and mast cells also induces GATA3+ Treg differentiation in the gut. IL4 secreted by IL33-triggered ILC2s increases IgE production by B cells in the intestine. Mast cells stimulated by Th2-derived IL4 produce IL9, which further induces the accumulation of mast cells in the intestine, in an autocrine fashion. IL9-producing mast cells increase gut permeability via the expression of VEGF. Note that blue arrows indicate induction or stimulation while red arrows represent migration or proliferation. ATP, adenosine triphosphate; TSLPR, TSLP receptor. |
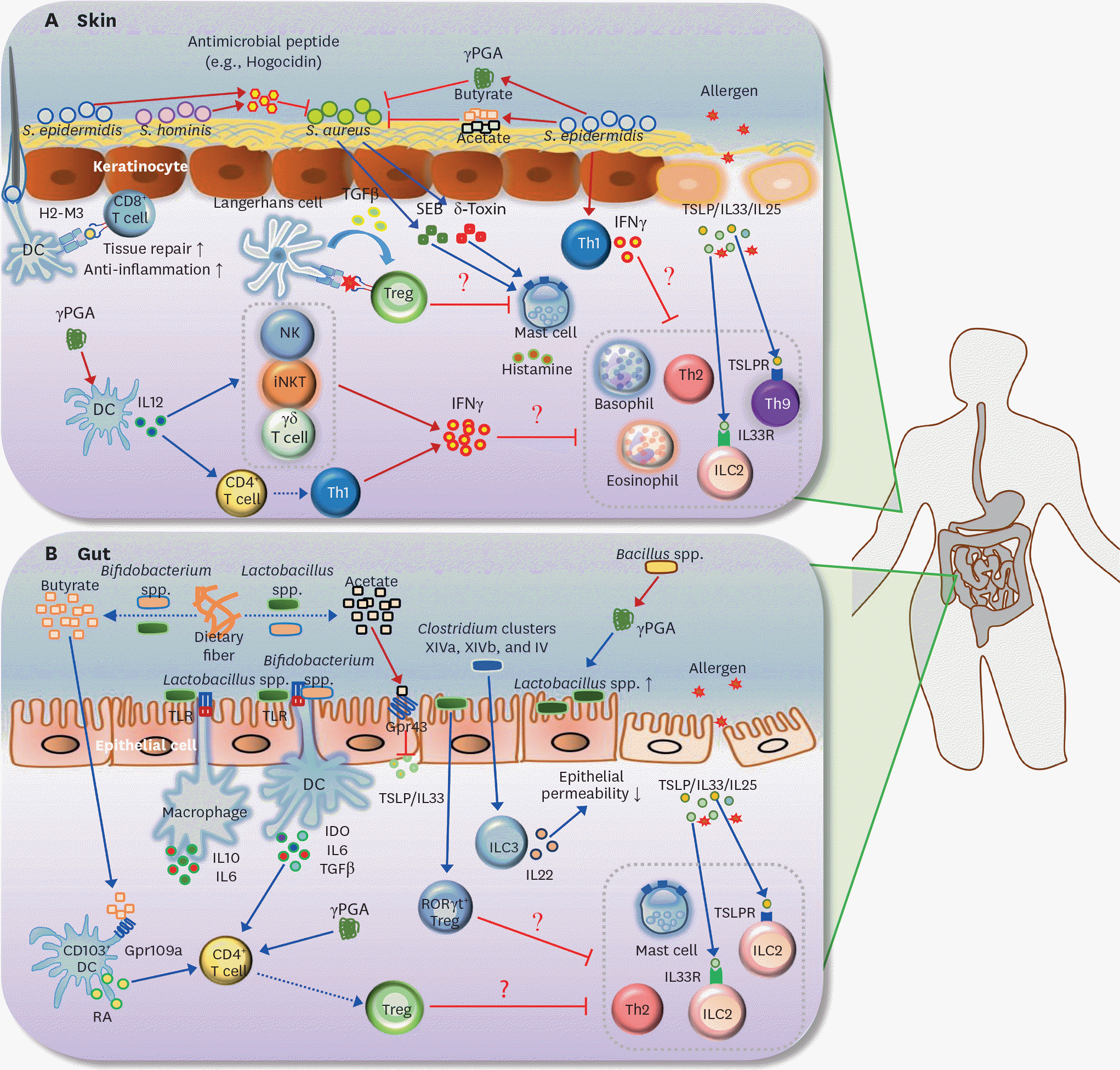 | Figure 2.Roles of the microbiota and its metabolites in AD and FA. (A) S. aureus is known to be the main pathogen that induces AD. SEB and δ-toxin, secreted by S. aureus, induce degranulation of mast cells in the skin. Survival of S. aureus in the skin is selectively inhibited by antimicrobial peptide (e.g., hogocidin) derived from commensal bacteria including S. epidermidis and S. hominis. S. epidermidis-derived γ PGA suppresses survival of S. aureus. S. epidermidis also produces SCFAs (butyrate and acetate), which suppress the colonization of S. aureus in the skin. In addition, H2-M3-restricted commensal-specific CD8+ T cells, induced by S. epidermidis-stimulated DCs, contribute to both anti-inflammatory and tissue repair functions. The skin Treg population can be increased by the resting LCs via TGFβ; these Tregs may be responsible for the suppression of mast cell degranulation. Skin-resident commensal S. epidermidis increases IFNγ production by dermal T cells. In addition, γ PGA from the skin commensal bacteria can activate DCs to induce differentiation of Th1 and activation of IFNγ-producing cells such as NK, iNKT, and γδ T cells. Consequently, IFNγ derived from S. epidermidis-activated Th1 cells, γ PGA-induced Th1, and IFNγ-producing cells may lead to the suppression of skin allergic effector cells (e.g., ILC2, basophils, eosinophil, Th2 cells, and Th9 cells). (B) Butyrate and acetate are produced from dietary fiber by commensal bacteria including Lactobacillus spp. and Bifidobacterium spp. Acetate suppresses TSLP and IL33 via epithelial GPR43 signaling, and butyrate triggers CD103+ DCs to produce retinoic acid via GPR109a signaling. Lactobacillus spp. stimulates macrophages to produce IL10 and IL6 in a TLR2/TLR6-dependent manner. Moreover, commensal bacteria including Lactobacillus spp. and Bifidobacterium spp. induce differentiation of naive CD4+ T cells into Foxp3+ Tregs via IDO, IL10, and TGFβ. γ PGA from gut commensal bacteria can directly induce the generation of adaptive Foxp3+ Tregs from naive CD4+ T cells. Gut extracellular γ PGA, derived from Bacillus spp., induces compositional change of microbiota, such as increase of Lactobacillus spp. Non-toxin-producing Clostridium spp., including Clostridium clusters XIVa, XIVb, and IV decrease intestinal permeability via increased IL22 production by ILC3s. Lactobacillus spp. (e.g., Lactobacillus casei) promotes RORγ t+ Treg differentiation in the gut. Thus, skin Tregs, induced by commensal microbiota and its metabolites (γ PGA and SCFAs), can participate in the inhibition of gut allergic effector cells (e.g., ILC2, mast cells, and Th2 cells). Note that blue arrows indicate induction or stimulation, red arrows represent secretion, and red flat lines indicate inhibition. Moreover, dotted arrows indicate decomposition or differentiation. SEB, S. aureus exotoxin B; TSLPR, TSLP receptor. |




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download