1. Cheon CK, Jin HS, Kang EK, Kim HB, Kim BJ, Yu J, et al. Epidemic acute interstitial pneumonia in children occurred during the early 2006s. Korean J Pediatr. 2008; 51(4):383–390.

2. Kim BJ, Kim HA, Song YH, Yu J, Kim S, Park SJ, et al. Nationwide surveillance of acute interstitial pneumonia in Korea. Korean J Pediatr. 2009; 52(3):324–329.

3. Kim KW, Ahn K, Yang HJ, Lee S, Park JD, Kim WK, et al. Humidifier disinfectant-associated children's interstitial lung disease. Am J Respir Crit Care Med. 2014; 189(1):48–56.
4. Kim JY, Kim HH, Cho KH. Acute cardiovascular toxicity of sterilizers, PHMG, and PGH: severe inflammation in human cells and heart failure in zebrafish. Cardiovasc Toxicol. 2013; 13(2):148–160.

5. Paek D, Koh Y, Park DU, Cheong HK, Do KH, Lim CM, et al. Nationwide study of humidifier disinfectant lung injury in South Korea, 1994–2011. Incidence and dose-response relationships. Ann Am Thorac Soc. 2015; 12(12):1813–1821.

6. Park DU, Choi YY, Ahn JJ, Lim HK, Kim SK, Roh HS, et al. Relationship between exposure to household humidifier disinfectants and risk of lung injury: a family-based study. PLoS One. 2015; 10(5):e0124610.

7. Yang HJ, Kim HJ, Yu J, Lee E, Jung YH, Kim HY, et al. Inhalation toxicity of humidifier disinfectants as a risk factor of children's interstitial lung disease in Korea: a case-control study. PLoS One. 2013; 8(6):e64430.

8. Lee E, Seo JH, Kim HY, Yu J, Jhang WK, Park SJ, et al. Toxic inhalational injury-associated interstitial lung disease in children. J Korean Med Sci. 2013; 28(6):915–923.

9. Kim HJ, Lee MS, Hong SB, Huh JW, Do KH, Jang SJ, et al. A cluster of lung injury cases associated with home humidifier use: an epidemiological investigation. Thorax. 2014; 69(8):703–708.

10. Lee JH, Kim YH, Kwon JH. Fatal misuse of humidifier disinfectants in Korea: importance of screening risk assessment and implications for management of chemicals in consumer products. Environ Sci Technol. 2012; 46(5):2498–2500.

11. Park S, Lee K, Lee EJ, Lee SY, In KH, Kim HK, et al. Humidifier disinfectant-associated interstitial lung disease in an animal model induced by polyhexamethylene guanidine aerosol. Am J Respir Crit Care Med. 2014; 190(6):706–708.

12. Kim HR, Hwang GW, Naganuma A, Chung KH. Adverse health effects of humidifier disinfectants in Korea: lung toxicity of polyhexamethylene guanidine phosphate. J Toxicol Sci. 2016; 41(6):711–717.

13. Park DU, Friesen MC, Roh HS, Choi YY, Ahn JJ, Lim HK, et al. Estimating retrospective exposure of household humidifier disinfectants. Indoor Air. 2015; 25(6):631–640.

14. Park DU, Ryu SH, Lim HK, Kim SK, Choi YY, Ahn JJ, et al. Types of household humidifier disinfectant and associated risk of lung injury (HDLI) in South Korea. Sci Total Environ. 2017; 596-597:53–60.

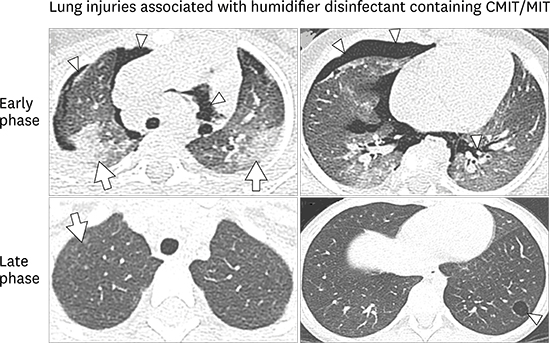
15. Yoon HM, Lee E, Lee JS, Do KH, Jung AY, Yoon CH, et al. Humidifier disinfectant-associated children's interstitial lung disease: computed tomographic features, histopathologic correlation and comparison between survivors and non-survivors. Eur Radiol. 2016; 26(1):235–243.

16. Hong SB, Kim HJ, Huh JW, Do KH, Jang SJ, Song JS, et al. A cluster of lung injury associated with home humidifier use: clinical, radiological and pathological description of a new syndrome. Thorax. 2014; 69(8):694–702.
17. Lee E, Seo JH, Kim HY, Yu J, Song JW, Park YS, et al. Two series of familial cases with unclassified interstitial pneumonia with fibrosis. Allergy Asthma Immunol Res. 2012; 4(4):240–244.

18. Nagorka R, Gleue C, Scheller C, Moriske HJ, Straff W. Isothiazolone emissions from building products. Indoor Air. 2015; 25(1):68–78.

19. Lundov MD, Mosbech H, Thyssen JP, Menné T, Zachariae C. Two cases of airborne allergic contact dermatitis caused by methylisothiazolinone in paint. Contact Dermat. 2011; 65(3):176–179.

20. Geier J, Lessmann H, Schnuch A, Uter W. Recent increase in allergic reactions to methylchloroisothiazolinone/methylisothiazolinone: is methylisothiazolinone the culprit? Contact Dermat. 2012; 67(6):334–341.

21. Bourke SJ, Convery RP, Stenton SC, Malcolm RM, Hendrick DJ. Occupational asthma in an isothiazolinone manufacturing plant. Thorax. 1997; 52(8):746–748.

23. Cho HJ, Park DU, Yoon J, Lee E, Yang SI, Kim YH, et al. Effects of a mixture of chloromethylisothiazolinone and methylisothiazolinone on peripheral airway dysfunction in children. PLoS One. 2017; 12(4):e0176083.

24. Poon R, Rigden M, Edmonds N, Charman N, Lamy S. Effects of 5-chloro-2-methyl-4-isothiazolin-3-one and other candidate biodiesel biocides on rat alveolar macrophages and NR8383 cells. Arch Toxicol. 2011; 85(11):1419–1427.

25. Jung HN, Zerin T, Podder B, Song HY, Kim YS. Cytotoxicity and gene expression profiling of polyhexamethylene guanidine hydrochloride in human alveolar A549 cells. Toxicol In Vitro. 2014; 28(4):684–692.

26. Kim MS, Jeong SW, Choi SJ, Han JY, Kim SH, Yoon S, et al. Analysis of genomic responses in a rat lung model treated with a humidifier sterilizer containing polyhexamethyleneguanidine phosphate. Toxicol Lett. 2017; 268:36–43.

27. Brent RL. Utilization of animal studies to determine the effects and human risks of environmental toxicants (drugs, chemicals, and physical agents). Pediatrics. 2004; 113(4):Suppl. 984–995.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download