This article has been
cited by other articles in ScienceCentral.
Abstract
Objective
Protocols for posterior circulation ischemic stroke have not been established by randomized clinical trials. Mechanical endovascular thrombectomy (MET) devices are evolving, and many of these devices already developed or in development are suitable for posterior circulation MET.
Materials and Methods
We investigated the medical records of patients who underwent MET for posterior circulation ischemic stroke from January 2012 to August 2016. Fifteen patients were included. MET was performed in patients with or without injected intravenous tissue plasminogen activator. MET was considered in patients with a National Institute of Health Stroke Scale score of 4 or more, older than 18 years, with definite occlusion of the basilar artery or posterior cerebral artery (PCA), and who arrived at the hospital within 24 hours from onset.
Results
The direct catheter aspiration technique was used in five cases, and the stent retrieval technique was used in seven cases. The stent retrieval technique with the direct aspiration technique was used in three cases. Recanalization failed in two cases. Basilar occlusion without PCA involvement is the only effective factor of successful recanalization (p = 0.03). Successful recanalization (p = 0.005) and the presence of a posterior communicating artery (p = 0.005) affected the good outcome at discharge.
Conclusion
An early diagnosis and active MET may improve the patient outcome. MET may help recanalization and good flow restoration and the potential for a good outcome.
Keywords: Ischemic stroke, Mechanical thrombectomy, Basilar artery occlusion, Endovascular treatment
INTRODUCTION
Protocols for anterior circulation ischemic stroke have been well established by several well-designed randomized clinical trials.
2)3)7)9)14) Mechanical endovascular thrombectomy (MET) is the standard treatment for acute ischemic stroke of the anterior circulation within 6 hours, and the challenge is to expand the treatment time window. Recently, a MET time window was expanded to within 24 hours.
1)13) However, protocols for posterior circulation ischemic stroke have not been established by randomized clinical trials.
6) MET devices are evolving, and many devices already developed or in development are suitable for posterior circulation MET. Thus, MET for posterior circulation acute stroke offers a challenge with the possibility of a good outcome for endovascular neurosurgeons. We report a study on the efficiency and implications of MET for posterior circulation ischemic stroke.
MATERIALS AND METHODS
We investigated the medical records of patients who underwent MET for posterior circulation ischemic stroke from January 2012 to August 2016. The inclusion criteria were as follows: 1) acute occlusion of the basilar artery or posterior cerebral artery (PCA) was confirmed by imaging modalities such as brain computed tomography (CT) angiography, magnetic resonance (MR) angiography or conventional cerebral angiography; 2) the patient arrived at the hospital within 24 hours of symptom onset; 3) the patient underwent MET; and 4) the patient underwent intravenous tissue plasminogen activator (IV-tPA) administration, or did not.
Sixteen patients who diagnosed with posterior circulation occlusion were investigated. One patient who underwent intra-arterial thrombolysis was excluded. All patients underwent plain brain CT at the emergency room to rule out, initially, intracranial hemorrhage. Patients who arrived at the hospital within 6 hours from symptom onset underwent brain CT angiography with CT perfusion. However, patients who arrived at the hospital 6 hours after onset underwent brain CT angiography with MR diffusion. Among the patients who were diagnosed with acute ischemic stroke of posterior circulation, IV-tPA was injected into these patients who had no contraindication and arrived at the hospital within 4.5 hours after symptom onset. IV-tPA was not injected into patients with a contraindication of IV-tPA or who arrived at the hospital 4.5 hours or longer after symptom onset. MET was performed in patients with or without the injection of IV-tPA. MET was considered in patients with a National Institute of Health Stroke Scale (NIHSS) score of 4 or greater, older than 18 years, with definite occlusion of the basilar artery or PCA who arrived at the hospital within 24 hours from onset.
MET was performed by two endovascular neurosurgeons using local anesthesia. Occlusion lesion was confirmed by vertebral artery angiography. A 5-French diagnostic catheter was used in cerebral angiography. Direct catheter aspiration or retrieval stent thrombectomy were used for MET. The Penumbra system (Penumbra Inc., Alameda, CA, USA) was used for direct thrombus aspiration. The guiding catheter was located at the distal vertebral artery as much as possible, and the 5-French Penumbra aspiration catheter with a Rebar™ 18 microcatheter (Medtronics, Irvine, CA, USA) and Synchro™ 14 microwire (Stryker Neurovascular, Fremont, CA, USA) was navigated to the occlusion site. The Penumbra aspiration catheter contacted the proximal portion of the thrombus after the microcatheter with a microwire passed through the occlusion site. The Pump Max™ (Penumbra Inc.) aspiration pump was turned on for thrombus aspiration using maximum negative pressure after removal of the microwire and microcatheter. When aspiration thrombectomy failed or aspiration thrombectomy was not suitable, we used stent retrieval thrombectomy with the Solitaire™ stent (Medtronics) or Trevo™ stent (Stryker Neurovascular). We chose retrieval stent that could completely cover the occlusion length sufficiently. We waited 5 minutes after stent installation and retrieved the stent by pulling it out gently with a microcatheter and sucking out the blood using a 50-mL syringe connected to the guiding catheter. The 50-mL syringe might prevent emboli migration and aspiration of the thrombus. When underlying arterial stenosis was found, the Solitaire stent was deployed without retrieval. Routine brain CT was performed within 1 hour after MET. Because of procedural complications, MR diffusion was performed within 24 hours after MET to check the infarction volume change.
The neurological condition was rated using NIHSS scores, and the neurologic outcome was rated using modified Rankin Scale (mRS) scores. We recorded the NIHSS score initially, at discharge, and 90 days after treatment. The mRS score was recorded at discharge and 90 days after treatment. The recanalization status based on the Thrombolysis In Cerebral Infarction (TICI) scale and TICI II (partial recanalization) or III (complete recanalization) was defined as successful recanalization. A good neurological outcome was defined as an mRS score of 0 to 2. We analyzed the geometric data, and improvements of the NIHSS scores and mRS score after MET, as well as the complications, successful recanalization, implications and efficacy of MET on posterior circulation ischemic stroke and presence of a posterior communicating artery (Pcom). SPSS version 20 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. Correlation analysis, regression analysis and logistic regression analysis, as appropriate for continuous or categorical variables, were used for statistical analysis. The null hypothesis of statistically no difference was rejected when the p value was less than 0.05.
RESULTS
Fifteen patients with acute posterior circulation occlusion were treated with MET (
Table 1). The mean age of the 15 patients was 71.93 ± 10.16 years. Ten patients were male, and five patients were female. They had vascular risk factors such as myositis, arrhythmia, atrial fibrillation, previous cerebral infarction, hypertension, angina, diabetes mellitus, cancer, hyperlipidemia, heart failure, and heart valve disease, with an average of 1.67 risk factors per patient. The patients had an average NIHSS score of 17.93 (range, 6–28). Two patients were occluded at the basilar top with PCA (
Fig. 1), four patients were occluded at the basilar top, and nine patients were occluded at the basilar trunk (
Fig. 2). Only six patients obtained IV-tPA. The average onset to puncture time was 245.26 minutes, and the average procedure time was 58 minutes (range, 30–127). The direct catheter aspiration technique was used in five cases, and the stent retrieval technique was used in seven cases. The stent retrieval technique with the direct catheter aspiration technique was used in three cases. Recanalization failed in two cases (Cases 1 and 3) because the thrombectomy devices could not reach the occlusion vessel (
Fig. 3). Successful recanalization (TICI ≥ 2a) was achieved in 10 patients. Although thrombectomy was attempted several times, TICI 1 was achieved in two patients (Cases 7 and 10), and procedural complication with recanalization failure occurred in one case (Case 12). All the cases that occluded at the basilar top with PCA achieved TICI 1. Two cases of recanalization failure underwent the direct aspiration technique. Two cases that achieved TICI 1 underwent the stent retrieval technique and stent retrieval with the direct aspiration technique. The procedural complication case underwent the stent retrieval technique. Hemorrhage was found in five patients after thrombectomy. However, all of them showed a very small amount of hematoma and no mass effect. Pcom was observed in only six patients. The average discharge NIHSS score was 11.58 and 11 patients showed an improved NHISS score at discharge. Four patients died after thrombectomy, and one patient did not show a change in the NIHSS score at discharge (Case 6). Two patients died from brain swelling, although the patients achieved a good angiographic outcome (TICI ≥ 2a). One patient died due to procedural complication. One patient died due to a medical complication after discharge. The average mRS score was 3.8 at discharge, and a good neurological outcome (mRS ≤ 2) was demonstrated in two cases at discharge. The average mRS score was 3.5 at 90 days after treatment. We lost one case at 90 days after treatment, and the mRS score was improved in the three cases.
We analyzed the effective factors of successful recanalization (TICI ≥ 2a) (
Table 2). Basilar occlusion without PCA involvement was the only statistically significant effective factor of successful recanalization (
p=0.03). We also analyzed which factors affect a good outcome at discharge (
Table 3). An initial low NIHSS score led to a good outcome at discharge (
p=0.03), and an initial high NIHSS score led to a poor outcome at discharge (
p=0.03). Hemorrhage after MET did not affect a poor outcome (
p>0.05). Successful recanalization (
p=0.005) and the presence of Pcom (
p=0.005) led to a good outcome at discharge. We analyzed which factors affected a good outcome (mRS ≤ 2) 90 days after treatment. Only a good mRS score at discharge (mRS ≤ 2) led to a good outcome 90 days after treatment (
p=0.03).
DISCUSSION
Mechanical thrombectomy was confirmed to be effective in acute ischemic stroke of anterior circulation and achieved the recognition of standard treatment modalities.
2)3)6)7)9)14) In addition, its therapeutic window is widening based on well-designed studies.
1)13) However, its effectiveness has not been proven in posterior circulation ischemic stroke. Randomized clinical trials for posterior circulation ischemic stroke have not been performed, and standard therapeutic protocols or devices have not been established.
6) This information has not been achieved for the following reasons: 1) posterior circulation occlusion is an uncommon stroke event, accounting for approximately 20% of all ischemic stroke events, a comparatively low rate.
10) 2) The pathophysiology of posterior circulation ischemic stroke is different from that of anterior circulation. The territory of the basilar artery and PCA is different from that of anterior circulation. Thus, the brain function is different, and stroke symptoms are different.
12) 3) The vascular movement of posterior circulation is different from that of anterior circulation, and the vascular diameter is also different. Thus, the selection of devices and treatment strategy may be different from that of anterior circulation thrombectomy.
17) 4) The territory of posterior circulation is very small compared to that of anterior circulation. However, a small infarction can lead to a life-threatening result. The prognosis of posterior circulation occlusion is very poor, and the mortality rate is very high (85–95%). Middle cerebral artery occlusion without recanalization showed a mortality rate of 41% to 79%, which is lower than that of posterior circulation infarction.
8)10)18) And 5) No optimized diagnostic tools exist to detect the penumbra zone of posterior circulation territories. The brainstem, cerebellum and PCA territory receive a relatively small blood supply volume. Perfusion modalities do not reflect posterior circulation perfusion and diffusion states. Many perfusion-based studies select patients with anterior circulation ischemic stroke.
3)4)16) Although these reasons make it difficult to treat posterior circulation infarction, its treatment now poses a challenge with the possibility of a favorable outcome for neuro-endovascular surgeons.
A good outcome at 90 days after treatment was associated only with a good mRS score at discharge in our study result. Thus, functional abilities might be not be recovered easily after discharge; therefore, the best attempts are made for a good mRS score at discharge. The study result demonstrated a close association between a good outcome at discharge and several parameters. The patients obtained a good outcome with a low initial NIHSS score, successful recanalization (TICI ≥ 2a) and the presence of Pcom in our study. A low initial NIHSS score indicates that patients had a salvageable penumbra zone or did not progress to irreversible infarction before mechanical thrombectomy. Mechanical thrombectomy helped to restore the blood flow before irreversible infarction. Successful recanalization helps to achieve a good outcome in patients because high-grade TICI indicates blood flow close to normalization on the penumbra zone. The development of Pcom means that the patients had a collateral blood pathway of the posterior circulation system.
15) Although proximal posterior circulation was occluded, collateral flow may maintain minimal perfusion of posterior circulation territories.
Successful recanalization affected the occlusion site in our study result. Basilar top or basilar trunk occlusion were recanalized successfully (
p=0.03). Basilar artery occlusion with PCA combined showed poor recanalization. Because, Basilar artery occlusion with PCA combined have high possibility of thrombus burden and show similar disease pattern to distal internal carotid artery occlusion with middle cerebral artery and anterior cerebral artery combined. Cross et al.
5) insisted that proximal basilar occlusion occurred via atherosclerosis, while distal or middle basilar artery occlusion occurred via an embolic source. Lindsberg et al.
11) also insisted that distal occlusion of basilar artery occurred via an embolus and was easier to recanalize than proximal occlusion. Basilar trunk or top occlusion were recanalized more easily than PCA combined occlusion in our study. PCA combined occlusion can demonstrate different characteristics. PCA combined occlusion is characterized by a long-segment thrombus compared with basilar occlusion. In addition, long-segment thrombus is likely to occur because of atherosclerosis. These data might explain why PCA combined lesion recanalization is more challenging than basilar occlusion.
One patient showed severe vessel tortuosity with atherosclerosis, and there was a failure to guide the catheter to the vertebral artery and perform thrombectomy. Four patients died after mechanical thrombectomy: one patient died due to a procedural complication; two patients died of brain swelling after mechanical thrombectomy. Angiographic results showed successful recanalization in these two patients; however, brain swelling developed despite blood flow restoration. The fourth patient died of pneumonia after discharge. Post-procedural brain CT found hemorrhagic change in eight patients; however, the amount of hemorrhage was minimal and did not have a mass effect. When we consider the technical aspects, mechanical thrombectomy of posterior circulation is sufficiently safe.
The result of this study showed high TICI grades because we not only selected the appropriate device according to the lesion but also repeated the procedure with another instrument to determine whether the lesion had been recanalized.
This study has some limitations. 1) This was a retrospectively designed study. 2) The study cases were limited by being chosen over a short period, with a single-center experience. 3) Various techniques and various devices were used (two retrieval stents and one suction catheter). And 4) There is no proper image-detecting tool for the posterior circulation perfusion status. The perfusion study may not reflect the state of posterior circulation. The penumbra area was not accurately calculated on imaging modalities. Therefore, patients should be well identified through clinical symptoms and CTA, and an appropriate indication for treatment should be selected.
CONCLUSION
Successful recanalization, the presence of Pcom and the initial neurological state are important factors affecting patient outcomes of posterior circulation ischemic stroke. Therefore, an early diagnosis and active MET may improve the patient outcome. MET may help vascular recanalization and cause good blood flow restoration, leading to a good outcome.
Fig. 1
Patient who occluded at the basilar top with PCA. (A) Top of basilar artery was occluded on initial angiography. (B) Top of basilar artery was recanalization after direction aspiration. However, right PCA was occluded. (C) Right PCA was partial recanalization after retrieval stent apply. (D) Right PCA was not recanalization on final angiography. PCA = posterior cerebral artery.
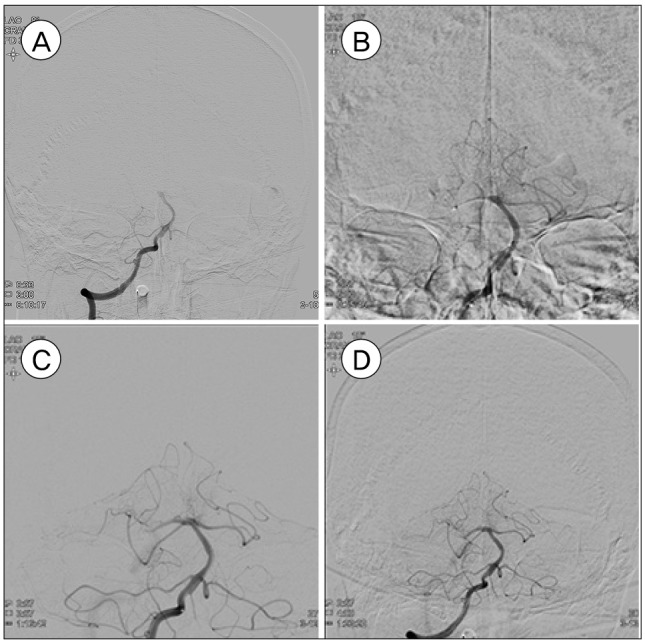
Fig. 2
Patient who occluded at basilar trunk. (A) Trunk of basilar artery was not shown on initial angiography. (B) Thrombus was captured in retrieval stent. (C) Basilar artery was recanalization completely after thrombectomy.
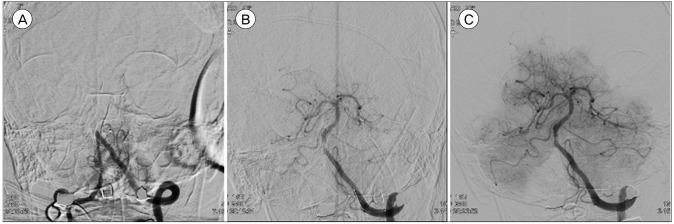
Fig. 3
Recanalization failed because the thrombectomy devices could not reach the occlusion vessel. (A) Although guiding wire reached right vertebral artery, diagnostic catheter wasn't able to reach the right vertebral artery because of vascular tortuosity. (B) Left vertebral artery wasn't shown on road map image. (C) angiographic image was taken by right subclavian artery angiography with inflated blood pressure cuff on right arm.
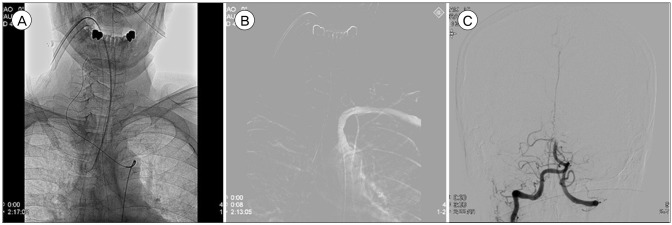
Table 1
Demographical characteristics
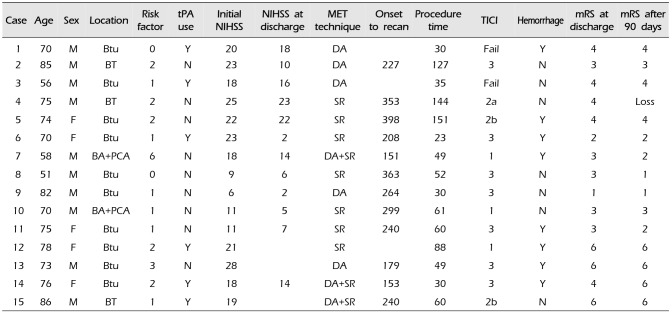
|
Case |
Age |
Sex |
Location |
Risk factor |
tPA use |
Initial NIHSS |
NIHSS at discharge |
MET technique |
Onset to recan |
Procedure time |
TICI |
Hemorrhage |
mRS at discharge |
mRS after 90 days |
|
1 |
70 |
M |
Btu |
0 |
Y |
20 |
18 |
DA |
|
30 |
Fail |
Y |
4 |
4 |
|
2 |
85 |
M |
BT |
2 |
N |
23 |
10 |
DA |
227 |
127 |
3 |
N |
3 |
3 |
|
3 |
56 |
M |
Btu |
1 |
Y |
18 |
16 |
DA |
|
35 |
Fail |
N |
4 |
4 |
|
4 |
75 |
M |
BT |
2 |
N |
25 |
23 |
SR |
353 |
144 |
2a |
N |
4 |
Loss |
|
5 |
74 |
F |
Btu |
2 |
N |
22 |
22 |
SR |
398 |
151 |
2b |
Y |
4 |
4 |
|
6 |
70 |
F |
Btu |
1 |
Y |
23 |
2 |
SR |
208 |
23 |
3 |
Y |
2 |
2 |
|
7 |
58 |
M |
BA+PCA |
6 |
N |
18 |
14 |
DA+SR |
151 |
49 |
1 |
Y |
3 |
2 |
|
8 |
51 |
M |
Btu |
0 |
N |
9 |
6 |
SR |
363 |
52 |
3 |
N |
3 |
1 |
|
9 |
82 |
M |
Btu |
1 |
N |
6 |
2 |
DA |
264 |
30 |
3 |
N |
1 |
1 |
|
10 |
70 |
M |
BA+PCA |
1 |
N |
11 |
5 |
SR |
299 |
61 |
1 |
N |
3 |
3 |
|
11 |
75 |
F |
Btu |
1 |
N |
11 |
7 |
SR |
240 |
60 |
3 |
Y |
3 |
2 |
|
12 |
78 |
F |
Btu |
2 |
Y |
21 |
|
SR |
|
88 |
1 |
Y |
6 |
6 |
|
13 |
73 |
M |
Btu |
3 |
N |
28 |
|
DA |
179 |
49 |
3 |
Y |
6 |
6 |
|
14 |
76 |
F |
Btu |
2 |
Y |
18 |
14 |
DA+SR |
153 |
30 |
3 |
Y |
4 |
6 |
|
15 |
86 |
M |
BT |
1 |
Y |
19 |
|
DA+SR |
240 |
60 |
2b |
N |
6 |
6 |
Table 2
Statistical analysis of factors affecting successful recanalization
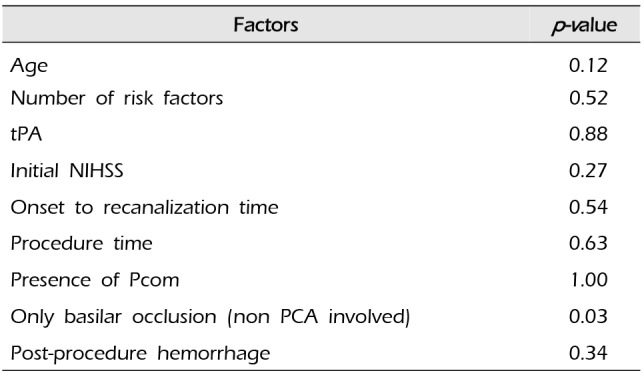
|
Factors |
p-value |
|
Age |
0.12 |
|
Number of risk factors |
0.52 |
|
tPA |
0.88 |
|
Initial NIHSS |
0.27 |
|
Onset to recanalization time |
0.54 |
|
Procedure time |
0.63 |
|
Presence of Pcom |
1.00 |
|
Only basilar occlusion (non PCA involved) |
0.03 |
|
Post-procedure hemorrhage |
0.34 |
Table 3
Statistical analysis of factors affecting a good outcome (mRS ≤ 2) at discharge
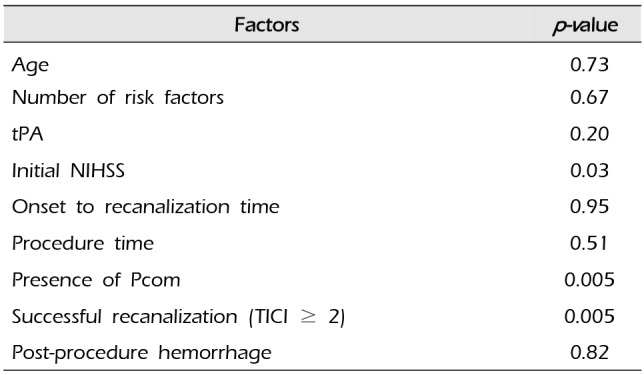
|
Factors |
p-value |
|
Age |
0.73 |
|
Number of risk factors |
0.67 |
|
tPA |
0.20 |
|
Initial NIHSS |
0.03 |
|
Onset to recanalization time |
0.95 |
|
Procedure time |
0.51 |
|
Presence of Pcom |
0.005 |
|
Successful recanalization (TICI ≥ 2) |
0.005 |
|
Post-procedure hemorrhage |
0.82 |










 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download