Abstract
Intraperitoneal metastatic hepatocellular carcinoma (HCC) is uncommon. Although rare, it can spontaneously rupture and cause hemoperitoneum similar to primary HCC in the liver. We present a case of intraperitoneal metastatic HCC that had spontaneously ruptured and appeared as an irregularly margined hemorrhagic mass with T1 high and T2 dark signal intensities on magnetic resonance imaging. Ruptured HCC is a life-threatening emergency with high mortality rate. Spontaneously ruptured intraperitoneal metastatic HCC should be considered if a patient with a history of HCC presents with acute abdomen, although rare.
Hepatocellular carcinoma (HCC) is the most common primary malignancy of the liver, composed of cells with characteristics similar to those of normal hepatocytes (1). Extrahepatic metastases are reported in 64% of patients with HCC, and the most common sites include lungs, abdominal lymph nodes, and bones. Peritoneal metastases are uncommon (2). Rarely, seeding tumor in the peritoneum may spontaneously rupture, similar to primary HCC in the liver (34). However, no studies have described magnetic resonance imaging (MRI) findings of this condition. Ruptured HCC is a life-threatening emergency with high mortality rate (25–75%) (5). We report a rare case of ruptured intraperitoneal metastatic HCC as a cause of hemoperitoneum with MRI findings.
A 40-year-old man presented with acute abdominal pain that started three days prior to presentation. On physical examination, diffuse tenderness was observed in the whole abdomen. The patient was a hepatitis B virus carrier with a history of recurrent HCC. Since the first diagnosis of HCC and right hepatectomy five years ago, he had an intrahepatic recurrence three times. Recurrent tumors were treated with wedge resection twice and radiofrequency ablation.
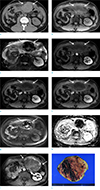
Computed tomography (CT) was performed without contrast enhancement because the patient had a history of anaphylactic shock related to iodine-based contrast media. On CT, a newly developed 8-cm irregularly margined high-density mass was detected in the omentum with small amount of hemoperitoneum (Fig. 1a). Abdominal MRI (Ingenia 3T, Philips Healthcare, Best, the Netherlands) was performed with gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid (Gd-EOB-DTPA), wherein the irregularly margined mass revealed slightly high signal intensity on T1-weighted image (Fig. 1b) and dark signal intensity on T2-weighted image (Fig. 1c), suggesting hemorrhage in the mass. No significant enhanced portion in the mass was seen on dynamic contrast enhancement study (Fig. 1d–f). Diffusion restriction was not observed on diffusion weighted images (DWI) with a high b-value (800 s/mm2) (Fig. 1g) and apparent diffusion coefficient (ADC) map (Fig. 1h). Because the mass was irregularly margined with surrounding hematoma, ruptured peritoneal seeding mass from the HCC was suspected, considering the patient's history. His vital signs were stable, but hemoglobin (Hb) level continued to decrease; he underwent arterial embolization, and Hb level was stabilized. Follow-up abdominal MRI was performed two months later, that revealed the size of the mass had enlarged to 15 cm. The mass had a well-defined margin compared to the previous study. During dynamic enhancement study, a highly enhanced solid portion was noted at the periphery of the mass on arterial phase (Fig. 1i). Surgical excision was performed, and the mass was pathologically confirmed as metastatic HCC with gross hemorrhagic component (Fig. 1j).
Intraperitoneal metastatic HCC is rare with an incidence of 5–14% (3). The most common cause of intraperitoneal metastasis is rupture of primary mass in the liver, leading to direct spillage and spread of cancer cells into the peritoneal cavity. However, intraperitoneal seeding of HCC is also possible without previous history of tumor rupture. Percutaneous needle biopsy, radiofrequency ablation, percutaneous ethanol injection, tumor resection, and transarterial chemoembolization are other suggested factors related to intraperitoneal metastatic HCC (46). In our case, the patient did not have a previous history of ruptured HCC. However, a history of tumor recurrence at the resection margin of the right hepatectomy site and previous treatment with tumor resection and radiofrequency ablation could have been risk factors of tumor seeding.
Although rare, intraperitoneal metastatic mass can spontaneously rupture and cause hemoperitoneum, similar to primary HCC in the liver. Ruptured HCC is a potentially life-threatening emergency because its mortality rate in the acute phase is as high as 25–75% (5). Early diagnosis and proper management including hemodynamic resuscitation are crucial for this emergency. The exact mechanism of spontaneous rupture of HCC is unclear; however, several hypotheses exist. Rupture of a parasitic vessel feeding or draining the tumor, laceration of a tumor surface by minor trauma either from outside the body or respiratory movement, and direct tumor invasion to the hepatic vein leading to venous congestion and increased intratumoral pressure are some of the most known speculations (7). According to one immunohistochemical study, vascular dysfunction has been postulated to be caused by elastin proliferation and degeneration, degradation of type IV collagen, and increased collagenase expression leading to easy split of blood vessels in ruptured HCC (8).
Similar to those of ruptured primary HCC in the liver, imaging findings of ruptured intraperitoneal metastatic HCC include hemoperitoneum, peritumoral hematoma, extravasation of contrast material, and discontinuity of the mass surface. Hemorrhagic mass may be observed as a hyperattenuating mass on non-enhanced CT and increased signal intensity on T1-weighted image. Signal intensity of blood may vary on T1 and T2 signal intensities depending on the age of hematoma. In our case, peritoneal mass was mostly observed as a hematoma without enhanced solid portion at the time of presentation. However, the mass had rapidly enlarged in size two months later with highly enhanced peripheral portion on the early arterial phase image, that was matched with typical imaging findings of HCC.
Regarding treatment of intraperitoneal metastatic HCC, benefits of systemic chemotherapy or sorafenib are poor, and surgical resection is challenging (6). Ruptured intraperitoneal metastatic HCC has no standardized treatment, because such cases are rare. In our case, transarterial chemoembolization was performed first because of Hb level changes; however, the mass continued to grow, and surgical excision was performed eventually.
In conclusion, spontaneous rupture of intraperitoneal metastatic HCC may result in an acute abdomen in a patient with a history of HCC despite its rarity. It was presented as an irregularly margined poorly enhanced mass revealing high signal intensity on T1-weighted image and dark signal intensity on T2-weighted image in MRI due to internal hemorrhage, with surrounding hematoma. Early recognition of this life-threatening condition may lead to proper management.
Figures and Tables
Fig. 1
A 40-year-old man with spontaneous rupture of intraperitoneal metastatic mass secondary to HCC. Axial non-enhanced CT scan (a) reveals an irregularly margined 8-cm high-density mass (arrows) in the omentum with hemoperitoneum. On MRI, the mass (arrows) reveals high signal intensity on pre-contrast T1-weighted image (b) and dark signal intensity on T2-weighted image (c). No significantly enhanced portion in the mass (arrows) was noted during dynamic enhancement study (d, arterial phase; e, portal phase; f, 3-min delayed phase). On DWI (b = 800 s/mm2) (g) and ADC map (h), the mass (arrows) does not reveal remarkable diffusion restriction. Follow-up MRI of the patient performed two months later revealed enlarged size, and more well-defined margin of the mass. Highly enhanced portion is well depicted at the periphery of the mass (arrows) on arterial phase image of dynamic enhancement study (i). Gross specimen (j) reveals a 15-cm metastatic HCC that appears to be a well-defined, firm, pink-tan, hemorrhagic mass.
References
1. van Malenstein H, van Pelt J, Verslype C. Molecular classification of hepatocellular carcinoma anno 2011. Eur J Cancer. 2011; 47:1789–1797.

2. Katyal S, Oliver JH 3rd, Peterson MS, Ferris JV, Carr BS, Baron RL. Extrahepatic metastases of hepatocellular carcinoma. Radiology. 2000; 216:698–703.

3. Okano J, Shiota G, Horie Y, et al. Rupture of metastatic nodule on the peritoneal surface secondary to hepatocellular carcinoma. Intern Med. 1996; 35:783–784.

4. Chen HW, Yang CF, Chao CC. Spontaneous rupture of peritoneal seeding hepatocellular carcinoma: report of two cases. J Acute Medicine. 2016; 6:64–66.

5. Tanaka A, Takeda R, Mukaihara S, et al. Treatment of ruptured hepatocellular carcinoma. Int J Clin Oncol. 2001; 6:291–295.

6. Spiliotis J, Nikolaou G, Kopanakis N, Vassiliadou D, Terra A, Efstathiou E. Hepatocellular carcinoma peritoneal metastasis: role of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (HIPEC). Gulf J Oncolog. 2017; 1:20–23.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download