Abstract
Canine transmissible venereal tumor (CTVT) is a tumor that commonly occurs in genital and extragenital sites of both genders. Long interspersed nuclear elements (LINE-1) retrotransposon has a pivotal role in allogenic transfection among uncontrolled dog populations. This study aimed to perform pathomorphological, immunohistochemical, and in situ polymerase chain reaction (PCR) evaluation of CTVT (n = 18) in transfected dogs during chemotherapy. Immunohistochemically, tumor phases were investigated by using specific markers (CD3, CD4, CD8, CD79, and transforming growth factor beta [TGF-β]), and investigated an amplified specific sequence of TVT LINE-1 retrotransposon by in situ PCR. Polyhedral-shaped neoplastic cells that had large, round, hypo/hyperchromatic nuclei and eosinophilic cytoplasm were detected. All marker results were positive, especially in the early weeks of recovery. CD4 and TGF-β markers were conspicuously positive at the initial stage. In situ PCR LINE-1 sequence was initially positive in only four cases. It is believed that the CD and TGF-β markers provide phase identification at tumor initiation and during chemotherapy. It is thought that presence of T and B lymphocytes, which have roles in cellular and humoral immunity, is needed so that regression of the tumor is possible.
Canine transmissible venereal tumor (CTVT) is a highly contagious neoplasia seen in both sexes. It is transmitted by coitus, and forms as a round cell tumor of mucous membranes of extragenital tissues such as anal mucosa, conjunctiva, lips, lymph nodes, eye, ear, and skin. The tumor is not only seen in dogs, it is also observed in foxes, coyotes, and jackals. The tumor is more common in females and occurs especially in later ages; although dogs 2 to 5 years old are also affected [58]. No breed or sex predisposition has been documented [16]. The tumor grows progressively (P-phase) and then regresses spontaneously (R-phase). The transition from the P-phase to R-phase has not yet been elucidated.
Long interspersed nuclear elements (LINE-1), which are retrotransposons, are localized at the 5′ region to the exon of the c-myc locus of CTVT cells [34]. The retrotransposons destabilize in the whole cellular genome and cause cellular proliferation, differentiation, and interaction with the microenvironment, leading to malignant transformation of cells [212]. Recently, the presence of LINE-1 retrotransposons is utilized as a diagnostic marker for CTVT [4].
In this study, clinical, pathomorphological, immunohistochemical, and molecular investigations of CTVT cases of both genders were performed. We investigated the role of the CD markers CD3, CD4, CD8, and CD79 as well as transforming growth factor beta (TGF-β) expression in the progression, latent, and regression phases during chemotherapy. In particular, an association was found between these marker expressions and the initial phase of the tumor. It is thought that tumor regression requires T and B lymphocytes. LINE-1 positivities were observed in some cases. It is deduced that evaluation of LINE-1 retrotransposon might be useful when assessing tumor transmission.
The materials used in this study were biopsy specimens taken from the vagina and penis regions of 18 dogs of different ages, breeds, and genders that had been diagnosed with TVT. Evaluation period and recovery duration were different between cases because mass size and response to chemotherapy differed from animal to animal (Table 1). This study was conducted in accordance with the guidelines of the Animal Care and Use Committee of Ankara University and was obtained the Animal Ethics Committee Approval (No. 2010-59-295).
Observations of the majority of cases recorded in our clinic included: age and gender, social habits of dogs, tumor location, common symptoms, tumor growth and spread, and previous TVT treatment. From each of the masses observed in each dog, a separate smear was made. The resulting slides were fixed with ether-ethanol (1:1 ratio). TVT diagnosis was confirmed by observation of tumor cells in preparations stained with the Papanicolau (PAP) staining method. For treatment, vincristine sulphate was administered intravenously in 100 mL saline at a dose of 0.025 mg/kg or 0.5 mg/m2 (maximum dose, 1 mg). Chemotherapeutic applications were performed once a week until the masses were macroscopically absent. Cytologic examination and tumor size measurement were performed weekly, and the response to treatment was monitored. Before each chemotherapy application, masses were assessed by macroscopic, cytological, histopathological, immunohistochemical and in situ polymerase chain reaction (PCR) techniques. In accordance with anamnesis, tumor regression was considered partial if a reduction by 50% of the pretreatment tumor size was achieved. It was considered unresponsive to treatment if less than a 50% reduction was achieved. Animals received chemotherapy once per week until the tumor was macroscopically and cytologically absent. Mean treatment duration was 4.00 ± 1.45 weeks.
Smears were obtained from each animal before and after chemotherapy application each week. Smear samples were fixed with an ether-ethanol absolute solution (1:1) for the PAP staining method. Impressed smears were air-dried for Giemsa staining. The density of CTVT cells was determined by examining 10 fields at 400× magnification.
Pre- and post-chemotherapy, weekly biopsies were evaluated for mass dimension, color, consistency, and surface features at each week.
Biopsy samples were fixed, processed routinely, and embedded in paraffin. All slides were stained according to the H&E or toluidine blue staining methods. All samples were scored as mild (+), moderate (++), and strong (+++) with respect to anaplasia, vascularization, inflammation, mitosis, and fibrosis.
Slides were stained with primary antibodies (CD3, CD4, CD8, CD79, TGF-β) at 56℃ for 1 h (Table 2) by applying the streptavidin-biotin complex peroxidase (StreptABC-P) method as described by the manufacturer (Novocastra Peroxidase Detection System; Leica Biosystems, UK). For negative control slides, PBS was used instead of the primary antibodies. As chromogen, diaminobenzidine was applied to sections. The remainder of the procedure was as previously described. All slides were observed under an optic light microscope (Leica DM4000B; Leica, Germany) and illustrated by using a camera attachment (Leica DFC-280; Leica). The percentage of positive cells was evaluated in 10 randomly selected fields at high magnification (400×).
Scoring of reactivity was classified according to staining intensity as (−), no positive staining; (+), 10% to 30% positivity; (++), 30% to 70% positivity; or (+++), 70% to 100% positivity. Also, this scoring was semiquantitatively done by calculating 10 fields at 400× magnification by two different observers during the histopathological and immunohistochemical examinations.
Tissue sections were deparaffinized and rehydrated and digested with proteinase K (Sigma-Aldrich, USA). They were acetylated with 0.3% acetic acid at room temperature. The 25 µL PCR mixture (2.5 µL 10× PCR buffer, 4.5 µL 25 mm MgCl2, 4 µL 10 mm dNTP, 5 mL 0.4 mm dig-dUTP, 5 mL 10 mm TVT-LINE forward primer: 5′-GCAGAAAGATCATTTTGAAAGAAGG-3′ for T8 and 5′-CTACCACCCCTAGTTCGTTT-3′ for T9), 1 µL (20 µM), 5 mL 10 mm reverse primer [3], 0.5 µL of 2.5 U/mL Jump Start Taq DNA Polymerase (Sigma-Aldrich, USA), and 11 µL diethyl pyrocarbonate (DEPC) were put on the slides. The predenaturation (95℃ for 7 min) and denaturation (94℃ for 3 min) for the first cycle were applied with 30 cycles for 1 min. This was followed by hybridization and extension (55℃ for 2 min) and a final extension (74℃ for 2 min). The reaction was then stopped (30℃ for 10 sec). Slides were treated with a 1× Standard Saline Citrate (SSC) solution [1].
After rehydration, the slides were incubated with anti-digoxigenin-alkaline phosphatase (AP) (diluted at 1:200, 37℃ for 1 h; Roche Diagnostics, Switzerland). A 0.2 N HCl was used for the endogenous phosphatase activity and NBT/BCIP (nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate) as chromogen. The slides were stained with 0.1% nuclear fast red and mounted. Control slides for the in situ PCR consisted in an omission of Taq polymerase or primers in the PCR mixture and in omission of AP or conjugated anti-digoxigenin antibody during the revelation step [1].
All of the masses in females were formed in the vagina; 3 cases were pendulous and 11 cases had a cauliflower-like appearance (panel A in Fig. 1). The masses were in dorsal (n = 3), ventral (n = 4) and lateral (n = 8) and diffuse (n = 7) locations. Masses in male dogs were diffusely localized at the penis and all were cauliflower-like in appearance.
Biopsy samples ranged from 1 to 10 mg in weight and from 2 to 20 mm in diameter. It was observed that the masses decreased and disappeared (panel B in Fig. 1) in size after chemotherapy. Some cases also presented with hemorrhages. The biopsy samples were elastic in consistency and cut sections were grayish-white in color.
Cytological examination revealed typical round to slightly polyhedral cells. Oval to round nuclei contained coarsely aggregated chromatin usually arranged in a cord-like pattern and centrally placed; most had a single, large, blue nucleolus. The cytoplasm was a light blue color and contained distinct clear vacuoles. The nuclear to cytoplasmic ratio of tumor cells was large. Positive mitotic figures (+/++) were prominent, especially in samples taken at 1 and 2 weeks.
A decrease in the number of tumor cells was gradually observed following chemotherapy in the Giemsa and PAP (panels C and D in Fig. 1) stained samples. Lymphocytes, plasma cells, macrophages, and neutrophil leukocytes were also present. In some cases, spindle-shaped cells were observed during the last few weeks of chemotherapy.
Tumor cells had polyhedral shapes with large and hypo/hyperchromatic nuclei and prominent amphophilic cytoplasm in P-phase (panel E in Fig. 1). Mitotic figures were plentiful. Vascularization and hyperemia were observed in some cases. The degree of intratumoral perivascular infiltration by mature lymphocytes, plasma cells, neutrophil leukocytes, and macrophages was variable. Neutrophil leukocytes and/or mononuclear cells were observed from the second week of chemotherapy onward. There were no inflammatory reactions observed at the beginning of chemotherapy (3 cases).
Tumor cells were remarkably decreased in the R-phase (12 cases). Vascularization and fibrosis increased until the final one or two weeks of chemotherapy in many cases (panel F in Fig. 1). Sometimes, inflammatory cells were absent or the samples only included lymphocytes and plasma cells.
T lymphocytes were positively stained with CD3 (panel G in Fig. 1). In P-phase, there was a reaction in three of the cases. During R-phase, reactions were obtained in 14 cases. In the final weeks of chemotherapy, 12 cases were CD3 positive.
Helper T lymphocytes were similarly positive with CD4. In P-phase, there was a reaction in 16 cases (panel H in Fig. 1), while in the R-phase, reactions were observed in 12 cases.
Cytotoxic and suppressor T lymphocytes showed positivity for CD8. Positivities were localized in cell cytoplasm as was observed in other cells. In the P-phase, positivities were present in 16 cases, while in R-phase, 13 cases were positive (panel I in Fig. 1).
CD79 positivities were determined in plasma and in B cells. The positivities were localized to the cytoplasm, and, in P-phase, there was positivity in 17 cases. In R-phase, 15 cases were positive (panel J in Fig. 1).
TGF-β showed positive reactions in the cytoplasm of T and B lymphocytes. In P-phase, positive reactions were observed in 17 cases (panel K in Fig. 1), while in R-phase, 14 cases were positive.
Based on the primary antibodies used and the chemotherapy therapy results, the phases and severity of detected positive markers are shown in detail in Fig. 2.
LINE-1 retrotransposons were detected in only four cases. The positive reactions were observed in the first week and they were completely localized in nuclei (panel L in Fig. 1). Positive reaction results were observed in the periphery of masses in one of the cases, in the central and peripheral areas in one of the cases, and in the central area in 2 cases. Homogeneous or granular positivities appeared reddish-purple. There were no reactions observed in tumor cells, stromal cells, or inflammatory cells, before or after chemotherapy in the R-phase.
Transmissible canine venereal tumors are evaluated according to cell tumor classifications and transmitted by coitus between dogs. The CTVT is generally localized in the genital organs and sometimes in extragenital tissues. Moreover, it is frequently seen in uncontrolled dog populations [5818]. Samples in this study were collected from dog shelters at which stray dogs were held. The tumors were more common in bitches in their sexually active period. In this study, the dogs were male (n = 4) and female (n = 14), and the mean age of the study group was 4.08 ± 1.89 years.
Clinically, CTVT cases are treated with radiotherapy and chemotherapy, although spontaneous regression was documented in an experimental study [15]. The most effective chemotherapeutic agent is vincristine sulphate and with occasional continuation with doxorubicin. Recoveries at high rates have been documented by using vincristine sulphate at a 0.025 mg/kg body weight dosage over 2 to 6 weeks [1317]. In this study, this was the preferred dosage of vincristine sulphate and with treatment, a decrease in mass diameter occurred; masses were completely regressed in 3 weeks (n = 5), 4 weeks (n = 6), 5 weeks (n = 4), 6 weeks (n = 2), or 7 weeks (n = 1).
Macroscopically, CTVT can be 0.5 to 10 cm in diameter, but masses were 2 to 20 mm diameter in the study. CTVT is sometimes observed together with ulceration, and the tumors are generally known to be firm in consistency [518]. In addition, their weights and diameters decreased until the end of chemotherapy. Cytologically, the tumor cells were round or oval, with eosinophilic vacuolar thin cytoplasm, and round large hyperchromatic nucleus and nucleolus as reported by Mukaratirwa and Gruys [11]. However, the presence of fibrocyte-like spindle-shaped cells was notable in the transition from P-phase to R-phase. Histopathologically; tumor cells at the developmental stage are mostly polygonal, hyperchromatic, and with pale eosinophilic cytoplasm, indistinct cell borders, and large nuclei and prominent nucleoli. Generally, when tumor cells proliferate so as to form cords, they are separated from each other by fine fibrous tissue as well as by marked mitotic activity.
Vascular stroma has been reported to be inadequate in early tumor development. In the R-phase of the tumor, T lymphocytes and other inflammatory cells are encountered [5818]. In this study, the presence of anaplastic cells and mitosis intensified at the beginning (progressive stage) while toward the last weeks (12 cases) the intensity decreased. Inversely proportional, it was noted that vascularization and fibrosis increased in the following weeks. Among the inflammatory cells encountered in the tumors, the presence or absence of lymphocyte-predominant mononuclear cells was observed in the R-phase. In the weeks following treatment, neutrophil leukocytes were noticed in the inflammatory response. Pérez et al. [14] separated TVT dogs in the same way as in this study into 2 groups: Group 1 (regressive phase) and Group 2 (progressive phase). Similar histopathological studies have shown that cell infiltrations are more intense in their Group 1 than in their Group 2, whereas, to the contrary, mitotic activity was observed to be lower in Group 1 than in Group 2. The results were obtained in the study of Pérez et al. [14] in which the malignant cells continued to decrease while connective tissue increased in the last week. It was noted that in the first week, the cases were in the P-phase and with chemotherapy gradual transition to regression phase was reported. Immunohistochemical evaluation is important in determining the phase in which T and B lymphocytes are encountered and determining their relative amounts in each phase [61014]. CD molecules are known as surface differentiation antigens, and TGF-β is a multifunctional protein that controls cellular differentiation and proliferation. In this study, the markers used for immunohistochemical diagnosis were CD3, CD4, CD8, CD79, and TGF-β. Those antibodies were preferred because they are often used in the diagnosis of such tumors. At the same time, markers are also important contributors to the differentiation of T and B cells.
The proteins have an antiproliferative effect on T and B lymphocytes and an activation effect of fibrocytes and fibroblasts. CD3+ is found in tumor cells during the transition from P-phase to R-phase, while CD79 is found in tumor cells, lymphocytes, and plasma cells at the periphery of a tumor. CD4+ and CD8+ are positive in T lymphocytes of the P-phase [1420]. TGF-β is active in CTVT cells in all of the P-phase, the transition, and the R-phase [20]. However, CD3+ mostly in transition and R-phase and CD79+ mostly in R-phase were present in peripheral lymphocytes in this study in contrast to previous reports. However, in this study, TGF-β+ was present in all phases as was reported by Trompieri-Silveira et al. [20]. Also, there were CD4+ cells mainly in the P-phase and infrequently in the transition from P-phase to R-phase. CD8+ cells were primarily found in transition and the R-phase. These results indicated that CD3 continues its role against CTVT cells not only in the transition phase but also in the R-phase. CD79, which is known as a specific marker for B lymphocytes, could also be present in T lymphocytes in case of a reaction to a tumor. It was thought that both markers can be jointly effective in chemotherapy for diminishment of a tumor.
In recent years, are often encountered with molecular field studies. The most attractive of these molecular level indicators is the LINE-1 sequence, which is a nonviral retrotransposon. LINE-1 has two reading frames open reading frames localized on the 5′ end of the c-myc oncogene of TVT cells [347]. The formation of these retrotransposons leads to instability in the genomic structure of the whole cell. LINE-1 insertion is found specifically and constantly in the 59 end of the CTVT cell c-myc gene, outside the first exon. A rearranged LINE–c-myc gene sequence has been used with PCR to diagnose CTVT. The total length of the inserted LINE gene is not constant in CTVT cells. Transformation of the cell into proliferation, differentiation, and interaction status within the microenvironment becomes inevitable [234712]. Therefore, the LINE-1 sequence is a useful diagnostic marker for malignancy in such tumors [4]. However, in one study, point mutations in the p53 gene with the LINE-1 sequence were evaluated together and both were shown to be malignant. In this study, the primer designs used by Choi and Kim [3] to detect the LINE-1 sequence were selected. LINE-1 sequences are commonly evaluated [34]; however, evaluation of TVT by in situ PCR has been restricted to only one study [7]. In that study, the nuclei of the cultured TVT cells were LINE-1 positive, but no positivity was observed in other inflammatory cells or connective tissue cells. However, in this study, in spite of testing for LINE-1 every week in every sample, positivity was detected in only 4 cases, and all of those were in the first week.
The localization of the LINE-1 positives was in the nuclei of TVT cells as was also reported by Liao et al. [7]. This indicates support for the notion of partial deletion of the LINE sequence, which is located at the 3′-end, as was suggested by Smit [19] because positives are not present in every event or in every selected week of the same event. However, the reason why LINE is left in this way remains unknown [7].
In this study, LINE-1 positivity was detected in the nucleoli of nuclei of CTVT cells, but no positivity was observed in other inflammatory cells or connective tissue cells. However, positivity was noted in 4 cases, and all of these positives were in the P-phase. It is believed that these results might provide useful information because this is the first evaluation of CTVT during chemotherapy by using in situ PCR to detect the active phase. However, it is suggested that the roles of other retrotransposons, such as SINE, or proteins of retinoblastoma (Rb, p107, p130), which are activators of LINE, should also be investigated in these type of tumors [921].
In conclusion, it is believed that the roles of critical CDs and TGF-β in different phases were clarified through this study. It is clear that T and B lymphocyte infiltrations and the releasing of the cytokine TGF-β are needed for regression of CTVT. Based on the presence of a few positive cases, it is thought that LINE-1 retrotransposon and its microenvironmental interactions are under pressure during chemotherapy. However, our advice to researchers is to focus research on the transmission of the tumor and on correlations between retrotransposons, the retinoblastoma family, and the p53 gene. Regardless, the outcome of most importance to clinicians is that the chemotherapy has an important role in tumor regression.
Figures and Tables
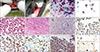 | Fig. 1Macroscopic and microscopic appearances of transmissible venereal tumor (TVT). (A and B) Macroscopical appearance of a representative tumor before (A) and after (B) chemotherapy. (C and D) Cytologic appearance of tumoral cells in P-phase (C) and in R-phase (D). (E) Tumoral cells, P-phase. (F) Decreased tumoral cells and fibrosis (arrow), R-phase. (G) CD3+ lymphocytes at the periphery of a tumor (arrow). (H) CD4+ T lymphocytes (arrows) in P-phase. (I) CD8+ B lymphocytes in R-phase (arrows). (J) CD79+ T and B lymphocytes and canine TVT cells in R-phase (arrows). (K) Transforming growth factor beta positive cells in P-phase. (L) Long interspersed nuclear element-1 positivities from in situ polymerase chain reaction (PCR) results in P-phase (arrows). Papanicolaou stain (C and D), H&E stain (E and F), ABC-P stain (G–K), In situ PCR stain (L). 400× (C–E and H–K), 100× (F and G), 125× (L), 500× (inset). |
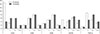 | Fig. 2Scoring of primary antibodies at different phases. Scoring: −, no positive staining; +, 10% to 30% positivity (10 fields at high magnification); ++, 30% to 70% positivity (10 fields at high magnification); +++, 70% to 100% positivity (10 fields at high magnification). TGF-β, transforming growth factor beta. |
Acknowledgments
This study was supported by the Ankara University Scientific Research Projects (project No. 10B3338008/2014), Turkey.
Notes
References
1. Alcigir ME, Atalay Vural S. Evaluation of liver and heart lesions induced by experimental fowl adenovirus-4 infection in broilers and virus detection by immunohistochemistry, immunofluorescence and in situ PCR. Revue Méd Vét. 2013; 164:348–357.
3. Choi YK, Kim CJ. Sequence analysis of canine LINE-1 elements and p53 gene in canine transmissible venereal tumor. J Vet Sci. 2002; 3:285–292.

4. Fonseca LS, Mota LSLS, Colodel MM, Ferreira I, Brandão CVS, Rocha NS. Spontaneous canine transmissible venereal tumor: association between different phenotypes and the insertion LINE-1/c-myc. Rev Colom Cienc Pecua. 2012; 25:402–408.
5. Goldschmidt MH, Hendrick MJ. Tumors of the skin and soft tissues. In : Meuten DJ, editor. Tumors of Domestic Animals. 4th ed. Ames: Iowa State Press;2002. p. 115–117.
6. Gonzalez CM, Griffey SM, Naydan DK, Flores E, Cepeda R, Cattaneo G, Madewell BR. Canine transmissible venereal tumour: a morphological and immunohistochemical study of 11 tumours in growth phase and during regression after chemotherapy. J Comp Pathol. 2000; 122:241–248.

7. Liao KW, Lin ZY, Pao HN, Kam SY, Wang FI, Chu RM. Identification of canine transmissible venereal tumor cells using in situ polymerase chain reaction and the stable sequence of the long interspersed nuclear element. J Vet Diagn Invest. 2003; 15:399–406.

8. McEntee K, Nielsen SW. Tumors of the female genital tract. Bull World Health Organ. 1976; 53:217–226.
9. Montoya-Durango DE, Liu Y, Teneng I, Kalbfleisch T, Lacy ME, Steffen MC, Ramos KS. Epigenetic control of mammalian LINE-1 retrotransposon by retinoblastoma proteins. Mutat Res. 2009; 665:20–28.

10. Mozos E, Méndez A, Gómez-Villamandos JC, Martín De Las Mulas J, Pérez J. Immunohistochemical characterization of canine transmissible venereal tumor. Vet Pathol. 1996; 33:257–263.

11. Mukaratirwa S, Gruys E. Canine transmissible venereal tumour: cytogenetic origin, immunophenotype, and immunobiology. A review. Vet Q. 2003; 25:101–111.

12. Murphy SK, Jirtle RL. Imprinting evolution and the price of silence. Bioessays. 2003; 25:577–588.

13. Nak D, Nak Y, Cangul IT, Tuna B. A Clinico-pathological study on the effect of vincristine on transmissible venereal tumour in dogs. J Vet Med A Physiol Pathol Clin Med. 2005; 52:366–370.

14. Pérez J, Day MJ, Mozos E. Immunohistochemical study of the local inflammatory infiltrate in spontaneous canine transmissible venereal tumour at different stages of growth. Vet Immunol Immunopathol. 1998; 64:133–147.

15. Richardson RC. Canine transmissible veneral tumor. Compend Contin Educ Prac Vet. 1981; 3:951–956.
16. Rogers KS. Transmissible venereal tumor. Compend Contin Educ Pract Vet. 1997; 19:1036–1045.
17. Scarpelli KC, Valladão ML, Metze K. Predictive factors for the regression of canine transmissible venereal tumor during vincristine therapy. Vet J. 2010; 183:362–363.

18. Schlafer DH, Miller RB. Female genital system. In : Maxie MG, editor. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals. 5th ed. New York: Elsevier Saunders;2007. p. 429–564.
19. Smit AF. Interspersed repeats and other mementos of transposable elements in mammalian genomes. Curr Opin Genet Dev. 1999; 9:657–663.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download