Abstract
To evaluate the carcinogenicity of p27 knockout (KO) mice with RNA-guided endonuclease (RGENs)-mediated p27 mutant exon I gene (IΔ), alterations in the carcinogenic phenotypes including tumor spectrum, tumor suppressor proteins, apoptotic proteins and cell cycle regulators were observed in p27 (IΔ) KO mice after treatment with 7,12-Dimethylbenz[a]anthracene (DMBA) and 12-O-tetradecanoylphorbol-13-acetate (TPA)(DT) for 5 months. The target region (544~571 nt) in exon I of the p27 gene was successfully disrupted in p27 (IΔ) KO mice using the RGEN-induced non-homologous end joining (NHEJ) technique. After DT exposure for 5 months, a few solid tumors (identified as squamous cell carcinoma) developed on the surface of back skin of DT-treated p27 (IΔ) KO mice. Also, squamous cell hyperplasia with chronic inflammation was detected in the skin dermis of DT-treated p27 (IΔ) KO mice, while the Vehicle+p27 (IΔ) KO mice and WT mice maintained their normal histological skin structure. A significant increase was observed in the expression levels of tumor suppressor protein (p53), apoptotic proteins (Bax, Bcl-2 and Caspase-3) and cell-cycle regulator proteins (Cyclin D1, CDK2 and CDK4) in the skin of DT-treated p27 (IΔ) KO mice, although their enhancement ratio was varied. Taken together, the results of the present study suggest that squamous cell carcinoma and hyperplasia of skin tissue can be successfully developed in new p27 (IΔ) KO mice produced by RGEN-induced NHEJ technique following DT exposure for 5 months.
p27Kip1 is a well-known member of the Cip/Kip family of cyclin-dependent kinase (Cdk) inhibitors [1]. It suppresses the cell cycle progression from G1 phase to the S phase by binding to the cyclin/Cdk complex, resulting in G1 arrest of the cell cycle [23]. In tumors, p27 deficiency enhances tumor aggressiveness, invasion of tumor cells and poor state of differentiation [45].
Several studies have been accomplished on the organ sensitivity in chemically induced carcinogenesis during p27 deficiency. Intestinal and lung adenocarcinoma and pituitary tumors were found in p27 KO mice with exon I and II deletion (I/IIΔ) challenged with γ-irradiation and N-ethyl-N-nitrosourea (ENU) injection [6], while skin tumor and intestinal adenoma were significantly increased in p27 (I/IIΔ) KO mice after DMBA and TPA treatment [7]. Also, dimethylhydrazine (DMH)-treated germline deletion p27 (exon I and II) mice presented only gastrointestinal adenocarcinoma malignancies, but N-methyl-N-nitrosourea (MNU) treatment induced hyperplasia and carcinoma in various organs [89]. Carcinoma of the urinary bladder and esophageal cancer were significantly higher in p27 (I/IIΔ) KO mice treated with N-butyl-N-(4-hydroxybutyl)nitrosamine (BBN) and methylbenzylnitrosamine (MBN) [1011]. Cotreatment with diethylnitrosamine (DEN) and phenobarbital (PB) resulted in promotion of genesis and progression of hepatocellular carcinoma in p27 (I/IIΔ) KO mice [12]. However, all above studies used only the p27 (I/IIΔ) KO mice with targeted disruption of exon I and II using the homologous recombination technique [1314]. The p27 (IΔ) KO mice generated by the RGEN technique have never been considered to investigate the organ sensitivity on chemically induced carcinogenesis.
This study therefore investigated the carcinogenicity in RGENS-mediated p27 (IΔ) KO mice after exposure to DT. These results provide novel evidence that squamous cell carcinoma of the skin tissue and alterations of cancer-related proteins in p27 (IΔ) KO mice can be successfully induced by DT treatment for 5 months, although other cancers were not observed in the various tissues.
To target the p27 gene in the mouse genome, the two most active RGEN-specific sequences to exon I of p27 were designed and synthesized (Figure 1A). After assessing their efficacy for small deletion at the target site in mouse NIH3T3 cells, each RGEN mRNA was injected into the cytoplasm of mouse pronuclear-stage embryos to produce mutant FVB/N founders (F0) with mutations in p27. The resultant p27 (IΔ) KO mice have a 28 bp deletion in exon 1 of the p27 gene (Figure 1A).
The target gene was identified by DNA-PCR analysis of the genomic DNA isolated from the tails of 3-week-old founder mice. The RGEN-mediated p27 mutant genes in FVB/N background strains were amplified using two primer sets. The p27 WT gene and mutant gene was identified by the forward primer (5′-CGC TTT TGT TCG GTT TTG TT-3′) and reverse primer (5′-CTC TCC ACC TCC TGC CAC-3′). Thereafter, 10 pmole of sense and antisense primers were added, and the reaction mixture was subjected to 30 cycles of amplification on a Thermal Cycler (T100™ Thermal Cycler, Bio-Rad, Hercules, CA, USA) as follows: 30 sec, 94℃; 30 sec, 62℃; 45 sec, 72℃. After amplification, the final PCR products of 180 bp and 155 bp were electrophoresed on 2% agarose gels.
The animal protocol used in this study was reviewed and approved in accordance with the protocols for ethical procedures and scientific care established by the Pusan National University-Institutional Animal Care and Use Committee (PNU-IACUC; Approval Number PNU-2017-1732). FVB/N mice were purchased from Samtako BioKorea Co. (Osan, Korea). All mice were handled at the Pusan National University (PNU)-Laboratory Animal Resources Center accredited by the Korea Food and Drug Administration (KFDA) (Accredited Unit Number-000231) and The Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International (Accredited Unit Number; 001525). Throughout the study, animals were provided with ad libitum access to water and a standard irradiated chow diet (Samtako BioKorea Co.) consisting of moisture (12.5%), crude protein (25.43%), crude fat (6.06%), crude fiber (3.9%), crude ash (5.31%), calcium (1.14%) and phosphorus (0.99%). During the experiment, mice were maintained in a specific pathogen-free (SPF) state under conditions of strict light cycle (lights on at 08:00 h and off at 20:00 h), 23±2℃ temperature, and 50±10% relative humidity.
p27 (IΔ) KO mice (n=14–18, 9-week-old age) were assigned to either a Vehicle-treated group (n=7-9) or DT-treated group (n=7-9). At day 1, all mice in the DT-treated p27 (IΔ) KO group were shaved, followed by a single application of DMBA (Sigma-Aldrich Co., St. Louis, MO, USA; 25 µg in 200 mL acetone), while those in the Vehicle-treated p27 (IΔ) KO group were treated with only acetone solution. Following DMBA treatment, the DT-treated p27 (IΔ) KO group received twice weekly applications of TPA (Sigma-Aldrich Co.; 200 mL of 10−4 M solution in acetone) for 5 months (Figure 1B). The formation of solid tumors on the skin of each mouse was observed at regular intervals. Finally, all animals were sacrificed using CO2 gas, after which blood, tumor and tissue samples were collected and stored in Eppendorf tubes at −70℃ until further assays.
After the DT treatment for 5 months, 9 organs (brain, ovary (or testis), kidney, adrenal gland, spleen, liver, thymus, heart and lung) were collected from the sacrificed mice and their weight were determined using an electrical balance (AB204, Mettler Toledo, Greifensee, Switzerland).
Tumor and skin tissues were excised from DT-treated p27 (IΔ) KO mice, fixed in 10% formalin, embedded in paraffin wax, processed routinely, and then sectioned into 4 µm thick slices. These sections were then stained with hematoxylin and eosin (H&E), after which their histopathological features were examined by light microscopy (Leica Microsystems, Wetzlar, Germany). The tumor type was identified by Prof. Beum Seok Han at the Department of Pharmaceutical Engineering, Hoseo University, Korea.
Proteins prepared from the skin tissue of mice were separated by 4–20% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) for 2 h, after which the resolved proteins were transferred to nitrocellulose membranes for 2 h at 40 V. Each membrane was then incubated separately, overnight at 4℃, with the following primary antibodies: anti-p27 (1:1,000, Sigma-Aldrich Co.), anti-p53 (1:1,000, Sigma-Aldrich Co.), anti-Bax (1:1,000, Abcam, Cambridge, UK), anti-Bcl-2 (1:1,000, Thermo Fisher Scientific, Waltham, MA, USA), anti-Caspase-3 (1:1,000, Cell Signaling Technology, Danvers, MA, USA), anti-Cyclin D1 (1:1,000, Cell Signaling Technology), anti-CDK2 (1:1,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA), anti-CDK4 (1:1,000, Santa Cruz Biotechnology) and anti-β-actin (1:1,000, Sigma-Aldrich Co.). The membranes were subsequently washed with washing buffer (137 mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, and 0.05% Tween 20) and incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG (1:2,000 dilution, Thermo Fisher Scientific) at room temperature for 1 h. Membrane blots were developed using Amersham ECL Select Western Blotting detection reagent (GE Healthcare, Little Chalfont, UK). The density of each band was quantified using the Image Analyzer System (Fluorchem FC2, Alpha Innotech, CA, USA) and was expressed as a fold-increase over control values.
A statistical significance was evaluated using the One-way Analysis of Variance (ANOVA) (SPSS for Windows, Release 10.10, Standard Version, Chicago, IL, USA) followed by Turkey post hoc t-test for multiple comparison. All data are expressed as the means±SD. A P value less than 0.05 (P<0.05) is considered statistically significant.
To compare the changes in body and organ weight of p27 (IΔ) KO mice during chemical induced carcinogenesis, we measured the body weight and 9 organ weights in p27 (IΔ) KO mice treated with DT for 5 months. No significant differences were observed in the body weight (data not shown) and organ weights of Vehicle-treated p27 (IΔ) KO mice and DT-treated p27 (IΔ) KO mice (Figure 1C). All mice in WT, Vehicle- and DT-treated p27 (IΔ) KO groups consistently maintained the level of body weight. These findings suggest that weight loss of body and organs is not induced in p27 (IΔ) KO mice during DT-induced carcinogenesis.
Next, we examined the tumor development in p27 (IΔ) KO mice after DT exposure for 5 months. The formation of small solid tumors were observed on the backs of DT-treated p27 (IΔ) KO mice, but not in the other groups (Figure 2A). The tumors were identified as squamous cell carcinoma showing alterations of nuclear size, shape, margin, chromatin pattern, nucleoli, and perinucleolar space (Figure 2Ba and b). Furthermore, squamous cell hyperplasia with chronic inflammation was detected in the dermis region of DT-treated p27 (IΔ) KO mice, as compared to the WT and Vehicle-treated p27 (IΔ) KO mice (Figure 2Bc and d). These results indicate that squamous cell carcinoma and hyperplasia can be successfully induced by DT treatment in RGEN-mediated p27 (IΔ) KO mice.
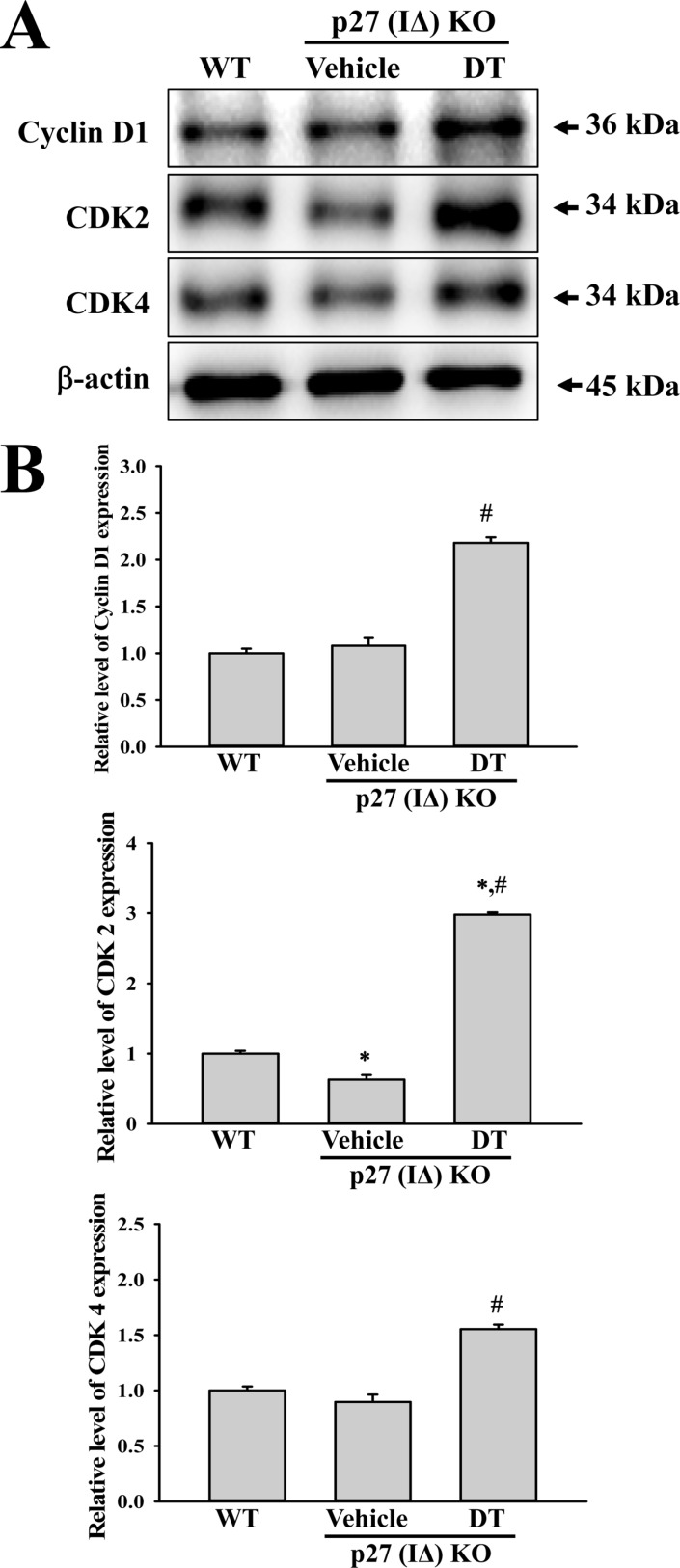
To investigate whether tumor development induced by DT treatment is accompanied with alterations in related protein levels, we investigated the expression of tumor suppressor proteins, apoptotic proteins and cell-cycle regulator proteins in p27 (IΔ) KO mice treated with DT. We observed that expression levels of p53 protein were significantly enhanced in DT-treated p27 (IΔ) KO mice compared with WT and Vehicle-treated p27 (IΔ) KO mice during the suppression of p27 proteins (Figure 3). Furthermore, we detected increased levels of the three apoptotic proteins (Bax, Bcl-2 and Caspase-3) in p27 (IΔ) KO mice after exposure to DT, although the increase rate was varied (Figure 4). A similar enhancement was observed in the expression of cell-cycle regulator proteins (Cyclin D1, CDK2 and CDK4); a remarkable increase was observed in DT-treated p27 (IΔ) KO mice compared with Vehicle-treated p27 (IΔ) KO mice (Figure 5). Together, these results indicate that the tumor development induced by DT treatment in RGEN-induced p27 (IΔ) KO mice is accompanied with alterations in the expression of tumor suppressor proteins, apoptotic proteins and cell-cycle regulator proteins.
Until now, various animal models encompassing environmentally induced cancers have provided key information regarding the causes, mechanisms and conceptual frameworks of cancer [15]. Especially, genetically engineered mice (GEM) were applied to study the complex interaction between genotype and environmentally-induced cancer development, although they were unable to account for the complexity of all environmental exposures [15]. In p53 and p27 (I/IIΔ) KO mice, the spectrum of tumors was significantly increased after exposure to carcinogens or radiation [16171819]. Hence, novel animal models have received increased attention in chemical carcinogenesis studies. In this study, we investigated chemically-induced tumorigenesis in RGENs-mediated p27 (IΔ) KO mice after DT exposure for 5 months. Our results reveal that DT treatment induces squamous cell carcinoma as well as an alteration in the expression of tumor suppressor proteins, apoptotic proteins and cell-cycle regulator proteins in the skin of p27 (IΔ) KO mice.
In previous studies, two classes of p27 KO mice generated by the targeted disruption of exon I and II of p27 gene using the homologous recombination technique have been applied to study carcinogenesis. Mice of the first type showed various pathological phenotypes including the thymic hyperplasia, enhanced proliferation of hematopoietic progenitor cells in spleen, neoplastic growth of pituitary, ovulatory defect and female sterility at various ages [13]. The other type showed small alterations such as the pituitary tumor, female sterility and increased the weight of immune organs within 12-week-old age [14]. In the present study, we used p27 (IΔ) KO mice with RGEN specific deletion of exon I in p27 gene. These mice did not exhibit any significant alterations in the physiological and pathological phenotypes until 10 months of age. These results differed from previous studies using p27 (I/IIΔ) KO mice. Therefore, long-term studies are necessary to observe the development of spontaneous tumors at old age.
Various chemical and physical carcinogens induce several types of tumors in several organs of p27 (I/IIΔ) KO mice with targeted disruption of exon I and exon II. Tumor development of the intestine, lung, pituitary and reproductive tract was induced by 4Gy whole-body γ-irradiation in p27 (I/IIΔ) KO mice [6]. High tumor susceptibility was detected in various tissues of p27 (I/IIΔ) KO mice after ENU and MNU treatment [69]. However, most chemical carcinogens induced the development of only a single tumor type in p27 (I/IIΔ) KO mice; DMH, BBN, MBN and DEN treatment increased the progression of gastrointestinal tumor [8], urinary bladder tumor [10], esophageal cancer [11], and liver tumor [12], respectively. Furthermore, sequential treatment of DT resulted in the development of adenoma of the small intestine and colon as well as increase in the growth rate of benign skin tumors (papilloma and carcinoma) in p27 (I/IIΔ) KO mice [7]. In this study, we investigated the chemically-induced carcinogenicity in RGEN-mediated p27 (IΔ) KO mice after sequential treatment of DT treatment for 5 months. Squamous cell carcinoma and hyperplasia were observed on the surface of the back skin of p27 (IΔ) KO mice. These results are, in part, very similar to those of a previous study [7]. Skin carcinoma was commonly detected in both p27 (I/IIΔ) KO mice and RGEN-mediated p27 (IΔ) KO mice, although adenoma of the small intestine and colon, and papilloma of the skin tumor, were observed in only p27 (I/IIΔ) KO mice [7]. One reason for these differences might be that the two classes of KO mice have different deletion sites for the p27 gene.
Taken together, the present study investigated the chemically-induced carcinogenicity in RGENs-mediated p27 (IΔ) KO mice after DT treatment for 5 months. Our results suggest that sequential treatment of DT accelerates squamous cell carcinoma and hyperplasia in the skin tissue of RGENs-mediated p27 (IΔ) KO mice, but there were no manifestations of any significant pathological features in other organs. Therefore, we believe that RGENs-mediated p27 (IΔ) KO mice can be considered a novel animal model for chemical carcinogenesis.
Acknowledgments
We thank the animal technician Jin Hyang Hwang, for directing the care and management of animals at the Laboratory Animal Resources Center in Pusan National University. This project was supported by a grant from BIOREIN (Laboratory Animal Bio Resources Initiative) from the Ministry of Food and Drug Safety in 2014~2018.
References
1. Sherr CJ, Roberts JM. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 1999; 13(12):1501–1512. PMID: 10385618.

2. Polyak K, Lee MH, Erdjument-Bromage H, Koff A, Roberts JM, Tempst P, Massagué J. Cloning of p27Kip1, a cyclin-dependent kinase inhibitor and a potential mediator of extracellular antimitogenic signals. Cell. 1994; 78(1):59–66. PMID: 8033212.
3. Toyoshima H, Hunter T. p27, a novel inhibitor of G1 cyclin-Cdk protein kinase activity, is related to p21. Cell. 1994; 78(1):67–74. PMID: 8033213.

4. Mori M, Mimori K, Shiraishi T, Tanaka S, Ueo H, Sugimachi K, Akiyoshi T. p27 expression and gastric carcinoma. Nat Med. 1997; 3(6):593. PMID: 9176477.

5. Kim DH, Lee HI, Nam ES, Shin HS, Sohn JH, Park CH, Yoon DS, Song SY, Park YE. Reduced expression of the cell-cycle inhibitor p27Kip1 is associated with progression and lymph node metastasis of gastric carcinoma. Histopathology. 2000; 36(3):245–251. PMID: 10692028.
6. Fero ML, Randel E, Gurley KE, Roberts JM, Kemp CJ. The murine gene p27Kip1 is haplo-insufficient for tumour suppression. Nature. 1998; 396(6707):177–180. PMID: 9823898.
7. Philipp J, Vo K, Gurley KE, Seidel K, Kemp CJ. Tumor suppression by p27Kip1 and p21Cip1 during chemically induced skin carcinogenesis. Oncogene. 1999; 18(33):4689–4698. PMID: 10467416.
8. Philipp-Staheli J, Kim KH, Payne SR, Gurley KE, Liggitt D, Longton G, Kemp CJ. Pathway-specific tumor suppression: Reduction of p27 accelerates gastrointestinal tumorigenesis in Apc mutant mice, but not in Smad3 mutant mice. Cancer cell. 2002; 1(4):355–368. PMID: 12086850.
9. Ogawa K, Murasaki T, Sugiura S, Nakanishi M, Shirai T. Organ differences in the impact of p27Kip1 deficiency on carcinogenesis induced by N-methyl-N-nitrosourea. J Appl Toxicol. 2013; 33(6):471–479. PMID: 22183835.
10. Hikosaka A, Ogawa K, Sugiura S, Asamoto M, Takeshita F, Sato SY, Nakanishi M, Kohri K, Shirai T. Susceptibility of p27Kip1 knockout mice to urinary bladder carcinogenesis induced by N-butyl-N-(4-hydroxybutyl)nitrosamine may not simply be due to enhanced proliferation. Int J Cancer. 2008; 122(6):1222–1228. PMID: 18027869.
11. Ellis FH Jr, Xu X, Kulke MH, LoCicero J 3rd, Loda M. Malignant transformation of the esophageal mucosa is enhanced in p27 knockout mice. J Thorac Cardiovasc Surg. 2001; 122(4):809–814. PMID: 11581618.

12. Guo J, Ma Q, Zhou X, Fan P, Shan T, Miao D. Inactivation of p27Kip1 promotes chemical hepatocarcinogenesis through enhancing inflammatory cytokine secretion and STAT3 signaling activation. J Cell Physiol. 2013; 228(10):1967–1976. PMID: 23460367.
13. Fero ML, Rivkin M, Tasch M, Porter P, Carow CE, Firpo E, Polyak K, Tsai LH, Broudy V, Perlmutter RM, Kaushansky K, Roberts JM. A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27Kip1-deficient mice. Cell. 1996; 85(5):733–744. PMID: 8646781.
14. Nakayama K, Ishida N, Shirane M, Inomata A, Inoue T, Shishido N, Horii I, Loh DY, Nakayama K. Mice lacking p27Kip1 display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell. 1996; 85(5):707–720. PMID: 8646779.
15. Kemp CJ. Animal models of chemical carcinogenesis: driving breakthroughs in cancer research for 100 years. Cold Spring Harb Protoc. 2015; 2015(10):865–874. PMID: 26430259.

16. Harvey M, McArthur MJ, Montgomery CA Jr, Butel JS, Bradley A, Donehower LA. Spontaneous and carcinogen-induced tumorigenesis in p53-deficient mice. Nat Genet. 1993; 5(3):225–229. PMID: 8275085.

17. Kemp CJ, Wheldon T, Balmain A. p53-deficient mice are extremely susceptible to radiation-induced tumorigenesis. Nat Genet. 1994; 8(1):66–69. PMID: 7987394.

18. Ziegler A, Jonason AS, Leffell DJ, Simon JA, Sharma HW, Kimmelman J, Remington L, Jacks T, Brash DE. Sunburn and p53 in the onset of skin cancer. Nature. 1994; 372(6508):773–776. PMID: 7997263.

19. Kiyokawa H, Kineman RD, Manova-Todorova KO, Soares VC, Hoffman ES, Ono M, Khanam D, Hayday AC, Frohman LA, Koff A. Enhanced growth of mice lacking the cyclin-dependent kinase inhibitor function of p27Kip1. Cell. 1996; 85(5):721–732. PMID: 8646780.
Figure 1
Scheme for deletion of the p27 gene, strategy for DT treatment, and evaluation of organ weights of DT-treated p27 (IΔ) KO mice. (A) Specific region (28 bp) of exon I in p27 (IΔ) KO mice were deleted with RGENs specific targeting. (B) After DMBA (25 µg) application on day 1, TP (10−4 M) was treated on the back skin of p27 (IΔ) KO mice for 5 months. (C) The weight of 9 organs collected from DT-treated p27 (IΔ) KO mice were measured using an electrical balance. The data shown represents the means±SD of three replicates. BR, Brain; LV, Liver; KD, Kidney; HT, Heart; LU, Lung; SP, Spleen; AD, Adrenal gland; TH, Thymus; GO, Gonad.
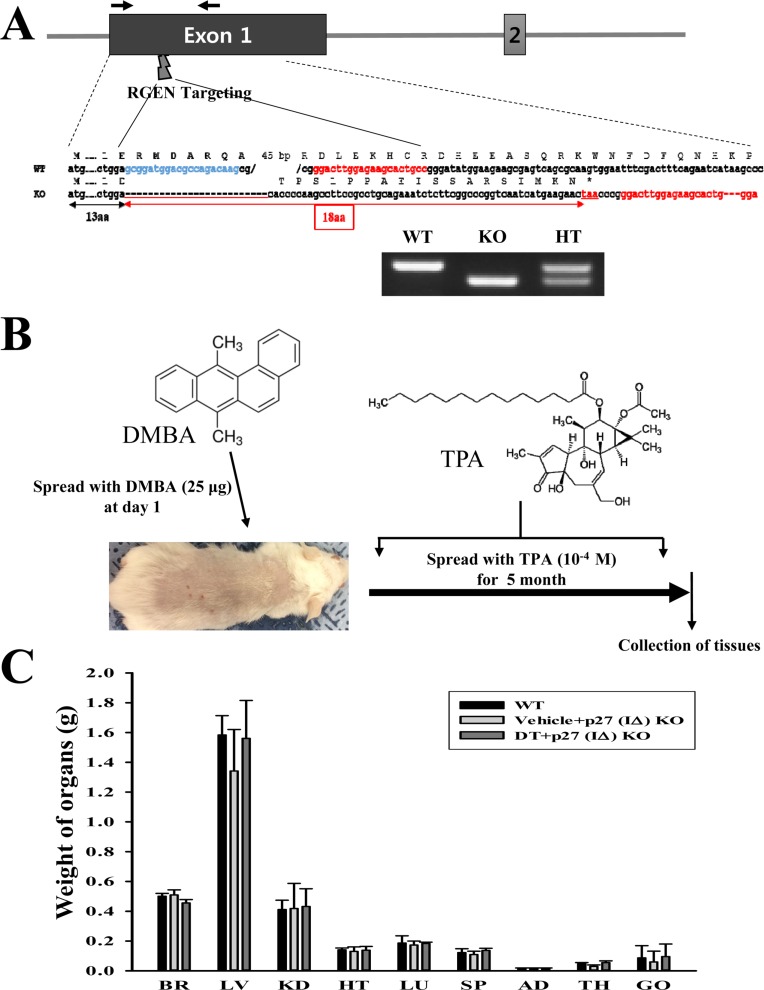
Figure 2
Tumor formation on the skin surface of DT-treated p27 (IΔ) KO mice. (A) Tumor morphology. Solid tumors were observed on the surface of the back skin. (B) Histopathology of solid tumors. H&E stained sections of skin and solid tumor from the DT-treated p27 (IΔ) KO mice were observed at 100× (left column) and 400× (right column) using a light microscope. Ep, epidermis; De, dermis; Hd, hypodermis; Ms, muscle layer.
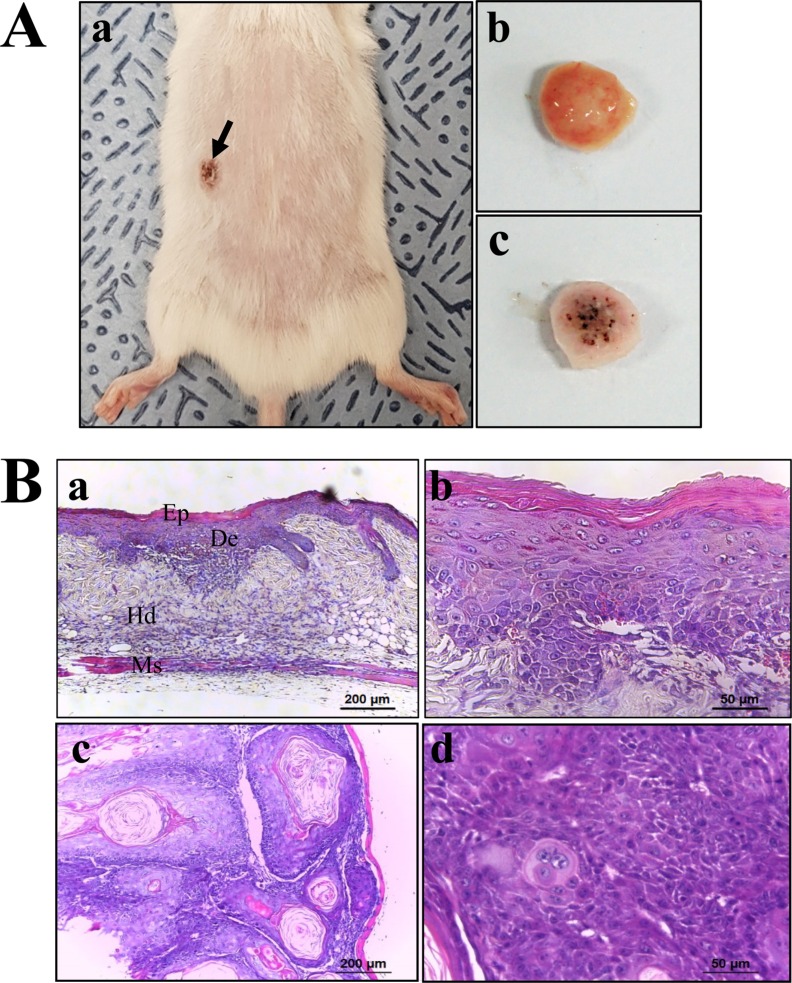
Figure 3
Expression analysis of tumor suppressor protein. An alteration in the expressions of p53 and p27 proteins were determined in DT-treated p27 (IΔ) KO mice by Western blot assays using HRP-labeled anti-rabbit IgG antibody. Band intensities were determined using an imaging densitometer, and the expression level of 6 proteins were evaluated relative to the intensity of actin bands. The data represents the means±SD (n=8). *, indicates P<0.05 compared to the WT mice. # indicates P<0.05 compared to the Vehicle-treated p27 (IΔ) KO mice.
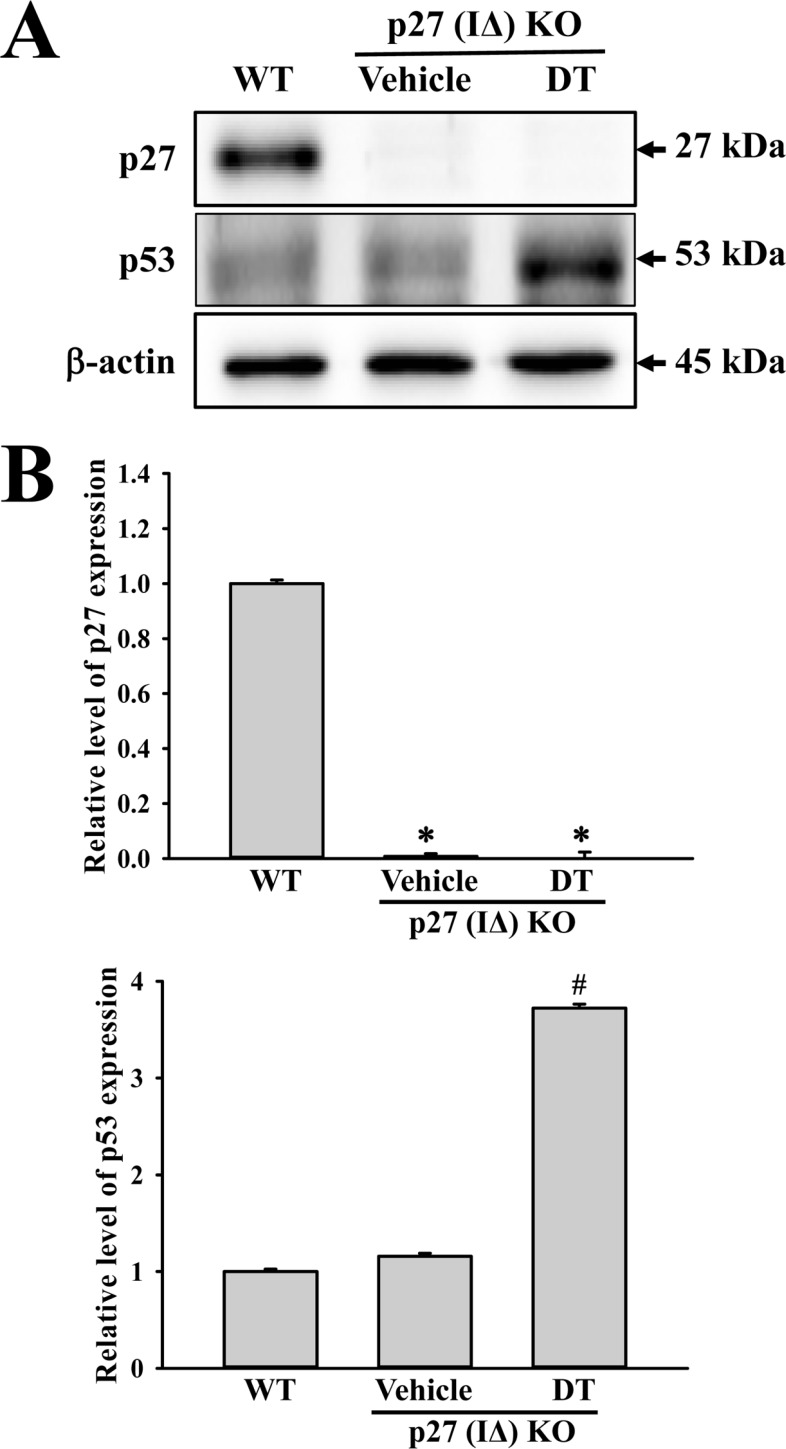
Figure 4
Expression analysis of apoptotic proteins. Alterations in the expressions of Bax, Bcl-2 and Caspase-3 proteins were determined in DT-treated p27 (IΔ) KO mice by Western blot assays using HRP-labeled anti-rabbit IgG antibody. Band intensities were determined using an imaging densitometer, and the expression level of 6 proteins were evaluated relative to the intensity of actin bands. The data represents the means±SD (n=8). *, indicates P<0.05 compared to the WT mice. # indicates P<0.05 compared to the Vehicle-treated p27 (IΔ) KO mice.
Figure 5
Expression analysis of cell-cycle regulator proteins. An alteration in the expressions of Cyclin D1, CDK2 and CDK4 proteins were determined in DT-treated p27 (IΔ) KO mice by Western blot assays using HRP-labeled anti-rabbit IgG antibody. Band intensities were determined using an imaging densitometer, and the expression level of 6 proteins were evaluated relative to the intensity of actin bands. The data represents the means±SD (n=8). *, indicates P<0.05 compared to the WT mice. # indicates P<0.05 compared to the Vehicle-treated p27 (IΔ) KO mice.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download