INTRODUCTION
Hypothermia therapy has already been documented to improve survival rates in unconscious patients resuscitated from cardiac arrest.
1)2) Previous animal studies reported that even a mild decrease in temperature, 32–35°C, could profoundly reduce myocardial ischemic injury during myocardial infarction.
1)2)3)4)5)6) Thus some have suggested that mild hypothermia therapy (body temperature < 35°C) in acute myocardial ischemia patients undergoing primary percutaneous coronary intervention (PCI) might reduce the myocardial infarct size and ultimately improve clinical outcomes.
7) However, previous large-scale randomized trials failed to prove that inducing mild hypothermia had an anti-infarct effect in acute ST-segment elevation myocardial infarction (STEMI) patients undergoing primary PCI.
8)9)10) Controversy still remains despite of previous study result regarding cooling method, target temperature, and cooling rate. In addition, the cardioprotective effect of natural mild hypothermia is completely unknown.
Cardiac magnetic resonance imaging (CMR) is useful for accurately assessing myocardial infarct size and salvaged myocardium, making it useful for evaluating the cardioprotective effects of natural mild hypothermia in STEMI patients. Our aim in this study was to investigate the relationship between natural mild hypothermia in the early phase after primary PCI and salvaged myocardium in patients with STEMI.
Go to :

METHODS
Study population
The study population consisted of patients on the Acute Myocardial Infarction–Cine Magnetic Resonance Imaging Registry at Samsung Medical Center in Seoul, Korea, from December 2007 to July 2014 (n=645). Eligible patients were those who had STEMI and underwent primary PCI. Exclusion criteria were the following: (1) STEMI patients undergoing primary fibrinolytic therapy; (2) previous coronary artery bypass grafting; (3) previous myocardial infarction history; (4) STEMI patients receiving reperfusion therapy more than 24 hours after symptom onset (5) insufficient information about vital signs before or after PCI; and (6) patients receiving induced hypothermia therapy. To evaluate the effect of natural hypothermia, patients who were treated with cooling therapy were excluded. In the end, a total of 291 patients were included in the present study and divided into 2 groups according to the presence of natural mild hypothermia (
Figure 1). This study was approved by the Samsung Medical Center review board, and written informed consent was obtained from all participants.
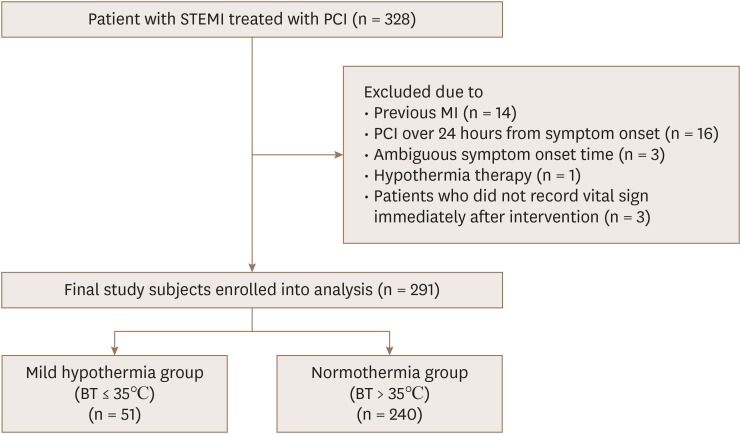 | Figure 1A schematic diagram of study patents selection. BT: body temperature, MI: myocardial infarction, PCI: percutaneous coronary intervention, STEMI: ST-segment elevation myocardial infarction.
|
Body temperature measurement
Each patient's initial vital signs were routinely measured, including body temperature at the emergency center, coronary angiography room just after the procedure, and cardiac intensive care unit. Vital signs were then regularly checked at the cardiac intensive care unit. Because we were interested in mild hypothermia during the early phase of the ischemic episode, we collected the body temperature readings taken over 24 hours. Body temperature was measured using a mercury thermometer from the axilla. Mild hypothermia was defined as a body temperature of 35°C or less.
6) Patients were divided into two groups according to the presence of natural mild hypothermia during the day after the PCI procedure. The natural mild hypothermia group had body temperature ≤ 35°C, and the normal temperature (normothermia) group had body temperature > 35°C.
Cardiac magnetic resonance imaging and analysis
CMR was performed during the index admission. The CMR scans were acquired with the use of a 1.5-tesla MRI scanner (Magnetom Avanto, Syngo MR B15 version; Siemens Medical Solutions, Erlangen, Germany). Cine images were obtained using a steady-state free-precession sequence with 8–10 contiguous short-axis slices to cover the entire left ventricle (LV) with a slice thickness of 6 mm and 4 mm inter-slice gap. T2-weighted imaging was performed in the cardiac short-axis direction using a dark-blood T2-weighted inversion recovery fast spin echo sequence and delayed gadolinium-enhanced imaging was acquired with the phase sensitive inversion recovery technique after injection of 0.15 mmol/kg Gadovist (gadobutrol; Bayer Schering Pharma, Berlin, Germany) in continuous short-axis image of 6mm in thickness with a 4-mm inter-slice gap. Late gadolinium enhancement and presence of microvascular obstruction (MVO) were evaluated at 10 minutes after gadolinium administration by using a multi-shot turbo field echo breath-hold sequence.
A validated software viewer (ARGUS; Siemens Medical System, Erlangen, Germany) was used to assess the data. The whole measurement procedure was conducted at the Samsung Medical Center CMR core laboratory. Two well-experienced radiologists performed measurement of CMR based on visual assessments. The radiologists were blinded to clinical characteristics and outcomes. To calculate the LV end-diastolic volume, LV end-systolic volume, and LV ejection fraction (EF), we used the Simpson method. Myocardial infarction size was defined as a hyper-intense myocardium area on the delayed gadolinium enhancement image, presented as percent of the hyper-enhanced area to the total LV myocardial volume. The infarction size was measured on each segment of the LV in several sliced planes of short-axis images. They were aggregated to calculate the infarct size and total LV myocardial volume. MVO was defined as a late hypo-intense area surrounded by a hyper-enhanced region. The extent of MVO was also assessed on delayed enhanced images. T2-weighted images were used to determine the area at risk (AAR) and presence of hemorrhagic infarction. The myocardial salvage index (MSI) was calculated as follows: MSI = (AAR − infarct size)/AAR × 100.
11)
Figure 2 shows an example CMR image.
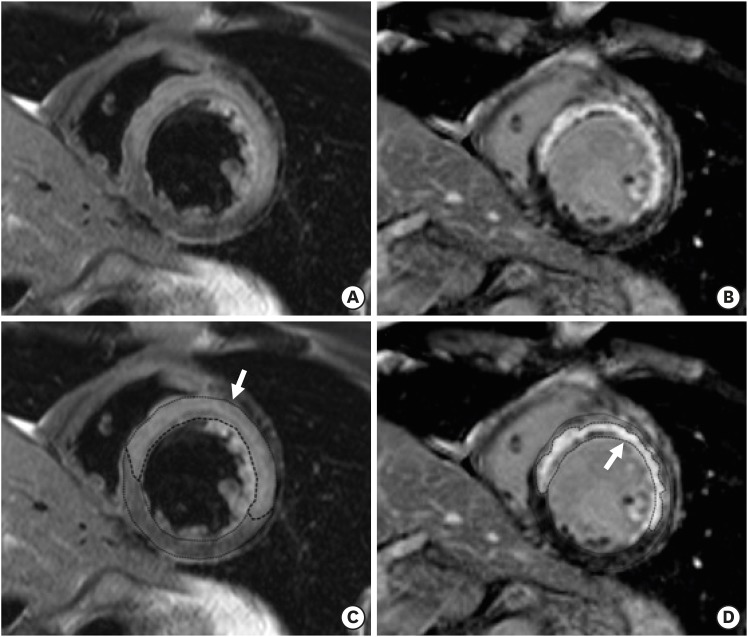 | Figure 2Example CMR images of anterior STEMI underwent PCI. Short-axis slices of T2-weighted image (A) and the corresponding delayed hyperenhancement image (B) in patients with anterior STEMI. The extent of area at risk (C) and infarct size (D) are the area surrounded by dot lines and indicated by white arrow. CMR: cardiac magnetic resonance imaging, PCI: percutaneous coronary intervention, STEMI: ST-segment elevation myocardial infarction.
|
Study outcomes
The primary study outcome was the MSI, measured using CMR. Secondary outcomes included the myocardial infarct size (% of LV volume), AAR (% of LV volume), presence of MVO, and presence of hemorrhagic infarction.
Statistical analysis
Categorical variables are presented as absolute numbers (n) and percentages (%), and differences were analyzed using the χ2 test or Fisher's exact test. Continuous variables are described using the median and interquartile range (IQR), and comparisons between the natural mild hypothermia and normothermia groups were made using the Mann-Whitney U-test. To determine the independent predictors of the MSI, we performed multivariable linear regression analyses using 10 variables identified as important to prognosis in previous studies: age, heart rate, systolic blood pressure, lowest body temperature, body surface area, history of angina, Killip class ≥ 2, serum glucose, infarct-related artery (left main or left anterior descending artery), and an initial thrombolysis in myocardial infarction (TIMI) flow of 0. The multivariate linear regression was analyzed by a step-wise backward selection process based on p-values, which were set at 0.05 (inclusion) and 0.10 (exclusion). A p-value of < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS Statistics 23.0 (SPSS, Chicago, IL, USA).
Go to :

RESULTS
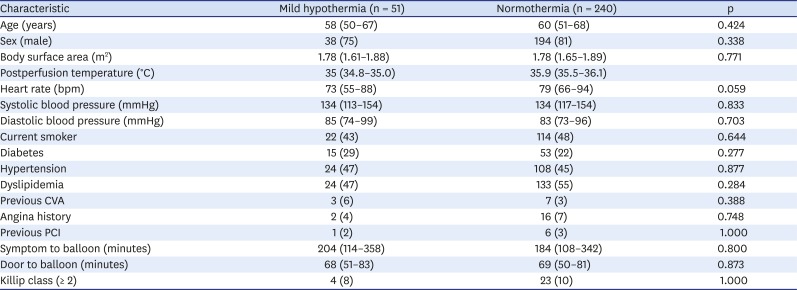
Among the 645 patients enrolled in the Acute Myocardial Infarction – Cine Magnetic Resonance Imaging Registry, 291 STEMI patients were included in this study and divided into a natural mild hypothermia group (n = 51) and a normothermia group (n = 240). The median post-PCI temperatures were 35.0°C and 35.9°C, respectively. The baseline characteristics of the patients are shown in
Table 1. The time from the onset of symptoms to the balloon was 204 (114–358) vs. 184 (108–342) minutes (median [IQR], natural mild hypothermia vs. normothermia, p = 0.800). The natural mild hypothermia group tended to have a lower heart rate than the normothermia group (73 [55–88] vs. 79 [66–94], p = 0.059). None of the other baseline characteristics showed statistical differences between the two groups.
Table 1
Baseline characteristics
|
Characteristic |
Mild hypothermia (n = 51) |
Normothermia (n = 240) |
p |
|
Age (years) |
58 (50–67) |
60 (51–68) |
0.424 |
|
Sex (male) |
38 (75) |
194 (81) |
0.338 |
|
Body surface area (m2) |
1.78 (1.61–1.88) |
1.78 (1.65–1.89) |
0.771 |
|
Postperfusion temperature (°C) |
35 (34.8–35.0) |
35.9 (35.5–36.1) |
|
|
Heart rate (bpm) |
73 (55–88) |
79 (66–94) |
0.059 |
|
Systolic blood pressure (mmHg) |
134 (113–154) |
134 (117–154) |
0.833 |
|
Diastolic blood pressure (mmHg) |
85 (74–99) |
83 (73–96) |
0.703 |
|
Current smoker |
22 (43) |
114 (48) |
0.644 |
|
Diabetes |
15 (29) |
53 (22) |
0.277 |
|
Hypertension |
24 (47) |
108 (45) |
0.877 |
|
Dyslipidemia |
24 (47) |
133 (55) |
0.284 |
|
Previous CVA |
3 (6) |
7 (3) |
0.388 |
|
Angina history |
2 (4) |
16 (7) |
0.748 |
|
Previous PCI |
1 (2) |
6 (3) |
1.000 |
|
Symptom to balloon (minutes) |
204 (114–358) |
184 (108–342) |
0.800 |
|
Door to balloon (minutes) |
68 (51–83) |
69 (50–81) |
0.873 |
|
Killip class (≥ 2) |
4 (8) |
23 (10) |
1.000 |

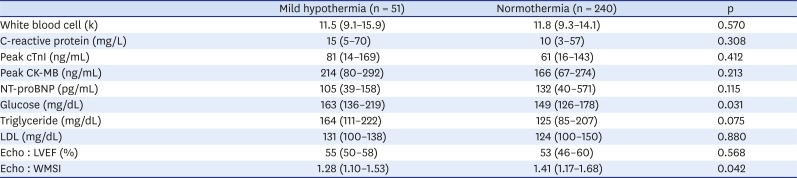
The biochemical and echocardiographic findings are summarized in
Table 2. There was no difference in inflammatory markers (white blood cell and C-reactive protein) or cardiac enzymes (troponin I, CK-MB) between the two groups. Patients with natural mild hypothermia had higher glucose levels than those with normothermia (median [IQR], 163 [136–219] vs. 149 [126–178]; p = 0.031). Echocardiography showed that the LV EF of the two groups was similar (median [IQR], 55 [50–58] vs. 53 [46–60]; p = 0.568). However, the regional wall motion score index was considerably lower in the mild hypothermia group than the normothermia group (median [IQR], 1.28 [1.10–1.53] vs. 1.41 [1.17–1.68]; p = 0.042).
Table 2
Biochemical data and echocardiographic finding
|
Mild hypothermia (n = 51) |
Normothermia (n = 240) |
p |
|
White blood cell (k) |
11.5 (9.1–15.9) |
11.8 (9.3–14.1) |
0.570 |
|
C-reactive protein (mg/L) |
15 (5–70) |
10 (3–57) |
0.308 |
|
Peak cTnI (ng/mL) |
81 (14–169) |
61 (16–143) |
0.412 |
|
Peak CK-MB (ng/mL) |
214 (80–292) |
166 (67–274) |
0.213 |
|
NT-proBNP (pg/mL) |
105 (39–158) |
132 (40–571) |
0.115 |
|
Glucose (mg/dL) |
163 (136–219) |
149 (126–178) |
0.031 |
|
Triglyceride (mg/dL) |
164 (111–222) |
125 (85–207) |
0.075 |
|
LDL (mg/dL) |
131 (100–138) |
124 (100–150) |
0.880 |
|
Echo : LVEF (%) |
55 (50–58) |
53 (46–60) |
0.568 |
|
Echo : WMSI |
1.28 (1.10–1.53) |
1.41 (1.17–1.68) |
0.042 |

Angiographic findings are described in
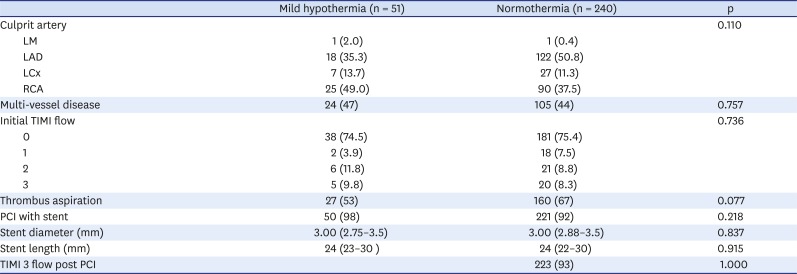
Table 3. The two groups had no significant differences. The proportion of culprit artery was slightly different between the two groups, but without statistical significance (p = 0.110). Although the normothermia group had a higher incidence of left anterior descending disease than the mild hypothermia group, the culprit lesion location in the proximal portion of the right coronary artery tended to be higher in the mild hypothermia group. (29.4% vs. 15.0%; natural mild hypothermia vs. normothermia, p = 0.103) (analysis not shown). A baseline TIMI flow of 0 was found in 74.5% and 75.4% of patients, respectively (p = 0.736). Thrombus aspiration was performed more frequently in the normothermia group (53% vs. 67%, p = 0.077). The prevalence of multivessel disease in the natural mild hypothermia group was similar to that in the normothermia group (47% vs. 44%, p = 0.757). In addition, the usage of a stent, the stent length, and stent diameter did not differ between the two groups.
Table 3
Angiographic finding
|
Mild hypothermia (n = 51) |
Normothermia (n = 240) |
p |
|
Culprit artery |
|
|
0.110 |
|
LM |
1 (2.0) |
1 (0.4) |
|
LAD |
18 (35.3) |
122 (50.8) |
|
LCx |
7 (13.7) |
27 (11.3) |
|
RCA |
25 (49.0) |
90 (37.5) |
|
Multi-vessel disease |
24 (47) |
105 (44) |
0.757 |
|
Initial TIMI flow |
|
|
0.736 |
|
0 |
38 (74.5) |
181 (75.4) |
|
1 |
2 (3.9) |
18 (7.5) |
|
2 |
6 (11.8) |
21 (8.8) |
|
3 |
5 (9.8) |
20 (8.3) |
|
Thrombus aspiration |
27 (53) |
160 (67) |
0.077 |
|
PCI with stent |
50 (98) |
221 (92) |
0.218 |
|
Stent diameter (mm) |
3.00 (2.75–3.5) |
3.00 (2.88–3.5) |
0.837 |
|
Stent length (mm) |
24 (23–30 ) |
24 (22–30) |
0.915 |
|
TIMI 3 flow post PCI |
48 (94) |
223 (93) |
1.000 |

Assessment of CMR data
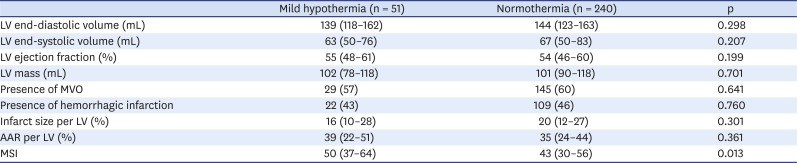
The CMR was performed during the index admission. There was no difference between the natural mild hypothermia and normothermia groups with regard to the timing of the CMR (3 days [3–4] vs. 4 days [3–5], p = 0.632). The primary endpoint (MSI) was significantly higher in the mild hypothermia group than in the normothermia group (median [IQR], 50 [37–64] vs. 43 [30–56], p = 0.013). The natural mild hypothermia group tended to have a lower infarct size per LV than the normothermia group (16 [10–28] vs. 20 [12–27]; p = 0.301). The extent of the AAR (39 [22–51] vs. 35 [24–44]; p = 0.361) showed higher tendency in the natural mild hypothermia group than in the normothermia group (
Figure 3). The LV EF and presence of MVO and hemorrhagic infarction were similar between the two groups. Further CMR findings are presented in
Table 4.
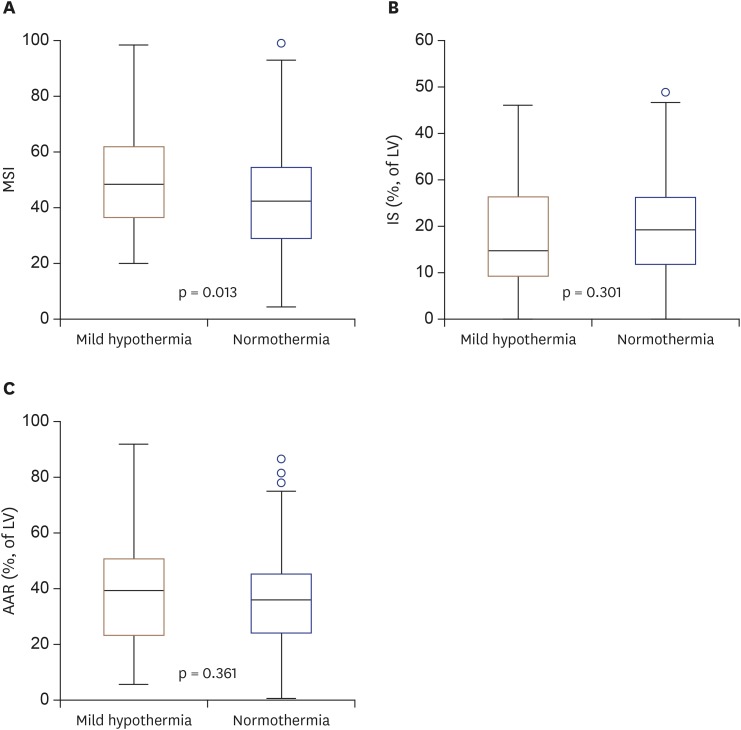 | Figure 3Boxplots of CMR findings according to the presence of mild hypothermia and MSI (A), IS (% of LV) (B), extent of AAR (% of LV) (C). AAR: area at risk, CMR: cardiac magnetic resonance imaging, IS: infarct size, LV: left ventricle, MSI: myocardial salvage index.
|
Table 4
Cardiac magnetic resonance imaging finding
|
Mild hypothermia (n = 51) |
Normothermia (n = 240) |
p |
|
LV end-diastolic volume (mL) |
139 (118–162) |
144 (123–163) |
0.298 |
|
LV end-systolic volume (mL) |
63 (50–76) |
67 (50–83) |
0.207 |
|
LV ejection fraction (%) |
55 (48–61) |
54 (46–60) |
0.199 |
|
LV mass (mL) |
102 (78–118) |
101 (90–118) |
0.701 |
|
Presence of MVO |
29 (57) |
145 (60) |
0.641 |
|
Presence of hemorrhagic infarction |
22 (43) |
109 (46) |
0.760 |
|
Infarct size per LV (%) |
16 (10–28) |
20 (12–27) |
0.301 |
|
AAR per LV (%) |
39 (22–51) |
35 (24–44) |
0.361 |
|
MSI |
50 (37–64) |
43 (30–56) |
0.013 |

The relationship between the lowest body temperature during the 24 hours after PCI in STEMI patients and corresponding MSI values is shown in
Figure 4. The linear regression with MSI as the independent variable is shown in
Table 5. There was significant negative association between the lowest body temperature and corresponding MSI in STEMI patients (β = −0.205, p < 0.001). Among the results from our univariate linear regression analyses of other independent variables and MSI, BSA (p = 0.067) and an initial TIMI flow of 0 (p = 0.076) had p values of less than 0.10, meaning that they might correlate with MSI. The multivariable regression analyses found no such association with MSI. The multivariable linear regression did show a significant association between the lowest body temperature during the 24 hours after PCI and MSI in STEMI patients (β = −0.191, p = 0.001).
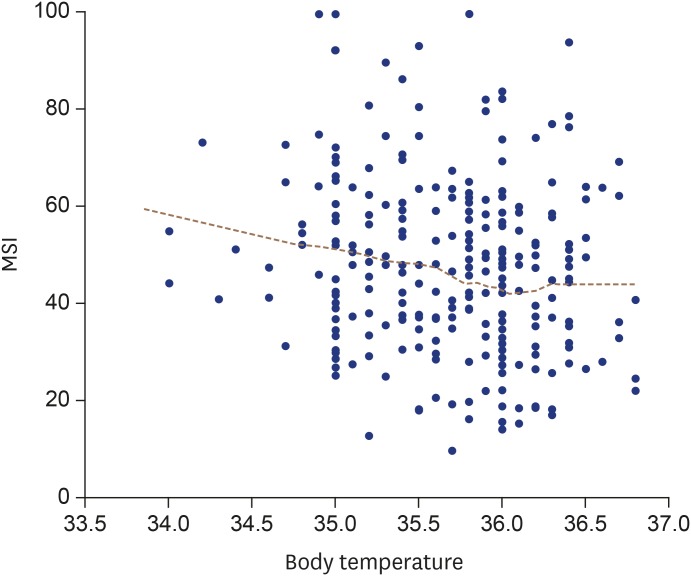 | Figure 4The relationship between MSI and body temperature in patients with STEMI. MSI: myocardial salvage index, STEMI: ST-segment elevation myocardial infarction.
|
Table 5
Linear regression analysis with MSI on CMR as dependent variable
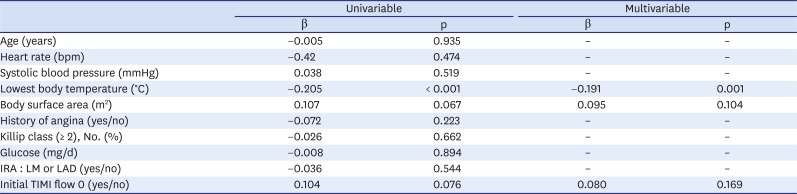
|
Univariable |
Multivariable |
|
β |
p |
β |
p |
|
Age (years) |
−0.005 |
0.935 |
– |
– |
|
Heart rate (bpm) |
−0.42 |
0.474 |
– |
– |
|
Systolic blood pressure (mmHg) |
0.038 |
0.519 |
– |
– |
|
Lowest body temperature (°C) |
−0.205 |
< 0.001 |
−0.191 |
0.001 |
|
Body surface area (m2) |
0.107 |
0.067 |
0.095 |
0.104 |
|
History of angina (yes/no) |
−0.072 |
0.223 |
– |
– |
|
Killip class (≥ 2), No. (%) |
−0.026 |
0.662 |
– |
– |
|
Glucose (mg/d) |
−0.008 |
0.894 |
– |
– |
|
IRA : LM or LAD (yes/no) |
−0.036 |
0.544 |
– |
– |
|
Initial TIMI flow 0 (yes/no) |
0.104 |
0.076 |
0.080 |
0.169 |

Subgroup analysis of natural mild hypothermia
Fifty-one of the STEMI patients treated with primary PCI in this study had natural mild hypothermia (less than 35°C) during the 24 hours after PCI. The median body temperatures measured in study patients during 24 hours are shown in a flow chart (
Figure 5). In the natural mild hypothermia group, body temperature was the lowest immediately after PCI. Thirty-two STEMI patients (62% of the mild hypothermia group) reached mild hypothermia within 6 hours of the PCI. Seven patients (14%) reached mild hypothermia between 6 hours and 12 hours after the PCI, and 12 patients (24%) reached mild hypothermia between 12 hours and 24 hours after the PCI. Therefore, we divided the natural mild hypothermia group into an early mild hypothermia group (patients who experienced mild hypothermia within 12 hours of the PCI) and a late mild hypothermia group (the rest of the mild hypothermia group). We then analyzed the CMR findings according to the onset time of mild hypothermia. The early mild hypothermia group tended to have higher MSI than the late mild hypothermia group (52.2 [41.3–64.2] vs. 37.1 [28.3–55.5], p = 0.055), but the other CMR findings were similar between the two groups (
Table 6).
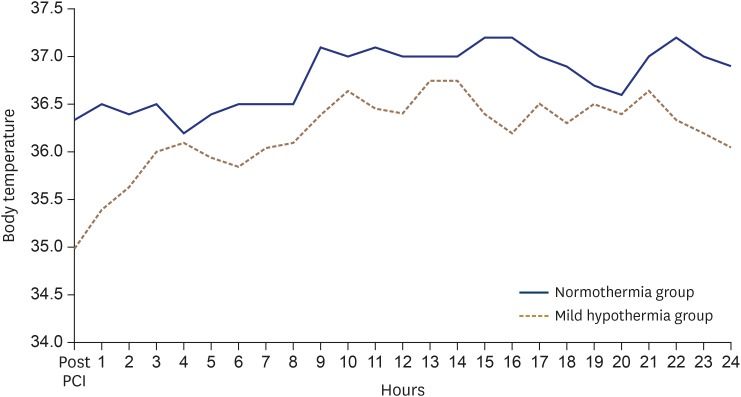 | Figure 5The median value of body temperature measured during 24 hours. The blue solid line indicates the normothermia group. The brown dotted line indicate mild hypothermia group. PCI: percutaneous coronary intervention.
|
Table 6
CMR finding in patient with mild hypothermia (n = 51)

|
Early mild hypothermia (n = 39) |
Late mild hypothermia (n = 12) |
p |
|
Infarct size per LV (%) |
15.7 (9.7–28.6) |
19.7 (11.1–28.2) |
0.665 |
|
AAR per LV (%) |
39.0 (26.4–51.3) |
32.1 (16.9–48.7) |
0.520 |
|
MSI |
52.2 (41.3–64.2) |
37.1 (28.3–55.5) |
0.055 |

Go to :

DISCUSSION
In this study, we evaluated the association between salvaged myocardium, which can be precisely measured by CMR images, and natural mild hypothermia in patients with STEMI who underwent primary PCI. The major finding of this study is that the presence of natural mild hypothermia (≤ 35°C) during the 24 hours after primary PCI is associated with high MSI in STEMI patients. Also, our multivariable linear analysis found a significant association between the lowest body temperature and corresponding MSI in STEMI patients. However, there was no significant difference in infarct size, AAR, MVO, or hemorrhagic infarction. This study is the first to use CMR to evaluate the effects of natural mild hypothermia on salvaged myocardium, and we found that mild hypothermia might have a myocardial protective effect in STEMI patients undergoing primary PCI.
For several decades, mild hypothermia therapy, 32–35°C, has been suggested for cardioprotective method in acute myocardial infarction patients undergoing primary PCI. However, previous large-scale trials failed to demonstrate an anti-infarct effect of mild hypothermia therapy in that population.
8)9)10) Two large-scale randomized controlled trials (Cooling as an Adjunctive Therapy to Percutaneous Intervention in Patients With Acute Myocardial Infarction
8) and Intravascular cooling adjunctive to percutaneous coronary intervention for acute myocardial infarction
9)) used primary PCI and primary PCI with endovascular cooling groups and found no significant difference in infarct size. However, a subgroup analysis of patients with anterior myocardial infarction who received induced hypothermia suggested infarct size reductions in both trials. Because the previous studies showed no effect, a shorter cooling induction time was tested. The CHILL-MI
10) trial induced hypothermia using a combination of a cold saline infusion and endovascular cooling before reperfusion in patients with STEMI, but it also showed no significant reduction in infarct size. Controversy thus remains.
On the basis of those previous studies, we wonder whether induced mild hypothermia might have a cardioprotective effect in patients with STEMI undergoing primary PCI. Moreover, we assumed that naturally developed mild hypothermia might also have an anti-infarct effect. Therefore, we investigated whether natural mild hypothermia protected against myocardial damage in STEMI patients and evaluated the MSI using CMR images. Our findings suggest that natural mild hypothermia is an independent predictor of greater salvaged myocardium in patients with STEMI undergoing primary PCI. Early-phase natural mild hypothermia in patients with MI is not a simple phenomenon, but CMR demonstrates that it is a defense mechanism as the body that attempts to salvage myocardium by reducing myocardial damage during myocardial ischemia.
Several pathophysiological mechanisms could explain this result. First, the crucial mechanism by which hypothermia induces cardioprotection is associated with a positive inotropic effect.
7) Animal studies showed that cardiac output was maintained under mild hypothermia because of an increase in stroke volume, despite a decrease in the heart rate in acute MI settings.
12) The positive inotropic effect of mild hypothermia is related to increased Ca
2+ sensitivity of the myofilament proteins
13) and improved Ca
2+-activated force generation.
14) In sheep, mild hypothermia during early ischemia and reperfusion was shown to enhance the LV contractile function by inhibiting post-infarction LV remodeling.
15) Hamamoto et al.
15) performed a study investigating the effects of very mild hypothermia (37.5°C) on healing in sheep. Eight weeks after MI, echocardiography showed lower LV end-systolic volume and LV end-diastolic volume and higher LV EF in hearts that experienced hypothermia than in normothermic hearts. Also, energy preservation and protective signal transduction were suggested as the metabolic mechanism of hypothermia-induced cardioprotection in that study.
6) In this study, the mild hypothermia group had similar blood pressure and a lower heart rate than the normothermia group. These results might be evidence of the positive inotropic effect of mild hypothermia suggested in animal experiments.
We further analyzed the CMR findings in the mild hypothermia group according to the time taken to reach mild hypothermia. That subgroup analysis showed that the early mild hypothermia group had a higher MSI than the late mild hypothermia group. Thus, early mild hypothermia (within 12 hours of PCI) has an anti-infarct effect in STEMI patients undergoing PCI. The results of this paper thus provide evidence for future hypothermia trials. Large studies will be needed to verify these results.
Our study has a few limitations. First, this study design was a retrospective, observational study, in which several biases influenced the results. Second, these results should be interpreted with caution because of the small sample size of the mild hypothermia group. Third, CMR might have been performed only in relatively stable STEMI patients. Unstable MI patients with large myocardial infarctions might not have been included. Fourth, we did not evaluate the association between CMR findings and clinical outcomes. Fifth, although we divided the study group into two groups according to the presence of mild hypothermia (≤ 35°C) during the 24 hours after PCI, the body temperature of patients with natural mild hypothermia was maintained above 35°C for most of the time, as shown in the flow chart of median body temperature (
Figure 5). Thus, the onset time, duration, and degree of hypothermia required to produce a cardioprotective effect remain controversial. Finally, the question of whether therapeutically induced mild hypothermia has the equal cardioprotective effect as naturally developed mild hypothermia in STEMI patients remains unanswered.
In conclusion, our study suggests that mild natural hypothermia within 24 hours of primary PCI in STEMI patients is associated with greater salvaged myocardium. This could be caused by an anti-infarct effect of low body temperature, although other contributing mechanisms cannot be excluded. Further large studies will be needed to verify these results.
Go to :














 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download