1. Lee SY, Konstantinidis IT, Eaton AA, Gönen M, Kingham TP, D'Angelica MI, et al. Predicting recurrence patterns after resection of hepatocellular cancer. HPB (Oxford). 2014; 16:943–953. PMID:
25041404.

2. Goh BK, Teo JY, Chan CY, Lee SY, Jeyaraj P, Cheow PC, et al. Importance of tumor size as a prognostic factor after partial liver resection for solitary hepatocellular carcinoma: implications on the current AJCC staging system. J Surg Oncol. 2016; 113:89–93. PMID:
26611492.

3. Zhu RX, Seto WK, Lai CL, Yuen MF. Epidemiology of hepatocellular carcinoma in the Asia-Pacific region. Gut Liver. 2016; 10:332–339. PMID:
27114433.

4. Lu WP, Dong JH. Hepatectomy for hepatocellular carcinoma in the era of liver transplantation. World J Gastroenterol. 2014; 20:9237–9244. PMID:
25071316.
5. Golse N, Bucur PO, Adam R, Castaing D, Cunha AS, Vibert E. New paradigms in post-hepatectomy liver failure. J Gastrointest Surg. 2013; 17:593–605. PMID:
23161285.

6. Balzan S, Belghiti J, Farges O, Ogata S, Sauvanet A, Delefosse D, et al. The “50-50 criteria” on postoperative day 5: an accurate predictor of liver failure and death after hepatectomy. Ann Surg. 2005; 242:824–829. PMID:
16327492.
7. Rahbari NN, Garden OJ, Padbury R, Brooke-Smith M, Crawford M, Adam R, et al. Posthepatectomy liver failure: a definition and grading by the international study group of liver surgery (ISGLS). Surgery. 2011; 149:713–724. PMID:
21236455.

8. Simpson AL, Adams LB, Allen PJ, D'Angelica MI, DeMatteo RP, Fong Y, et al. Texture analysis of preoperative CT images for prediction of postoperative hepatic insufficiency: a preliminary study. J Am Coll Surg. 2015; 220:339–346. PMID:
25537305.

9. Limdi JK, Hyde GM. Evaluation of abnormal liver function tests. Postgrad Med J. 2003; 79:307–312. PMID:
12840117.

10. Zakim D, Boyer TD, editors. Hepatology: a textbook of liver disease. 4th ed. Philadelphia: Saunders;2003. p. 1765.
11. Gibbins JM, Mahaut-Smith MP, editors. Platelets and megakaryocytes. New Jersey: Humana Press;2004.
12. Behne T, Copur MS. Biomarkers for hepatocellular carcinoma. Int J Hepatol. 2012; 2012:859076. PMID:
22655201.

13. Kashyap R, Jain A, Nalesnik M, Carr B, Barnes J, Vargas HE, et al. Clinical significance of elevated alpha-fetoprotein in adults and children. Dig Dis Sci. 2001; 46:1709–1713. PMID:
11508671.
14. Johnson PJ. The role of serum alpha-fetoprotein estimation in the diagnosis and management of hepatocellular carcinoma. Clin Liver Dis. 2001; 5:145–159. PMID:
11218912.

15. Lee YJ, Lee HR, Shim JY, Moon BS, Lee JH, Kim JK. Relationship between white blood cell count and nonalcoholic fatty liver disease. Dig Liver Dis. 2010; 42:888–894. PMID:
20472517.

16. Zipprich A, Kuss O, Rogowski S, Kleber G, Lotterer E, Seufferlein T, et al. Incorporating indocyanin green clearance into the model for end stage liver disease (MELD-ICG) improves prognostic accuracy in intermediate to advanced cirrhosis. Gut. 2010; 59:963–968. PMID:
20581243.

17. Du ZG, Wei YG, Chen KF, Li B. An accurate predictor of liver failure and death after hepatectomy: a single institution's experience with 478 consecutive cases. World J Gastroenterol. 2014; 20:274–281. PMID:
24415882.

18. Kim HJ, Kim CY, Park EK, Hur YH, Koh YS, Kim HJ, et al. Volumetric analysis and indocyanine green retention rate at 15 min as predictors of post-hepatectomy liver failure. HPB (Oxford). 2015; 17:159–167. PMID:
24964188.
19. Garcea G, Ong SL, Maddern GJ. Predicting liver failure following major hepatectomy. Dig Liver Dis. 2009; 41:798–806. PMID:
19303376.

20. Dinant S, de Graaf W, Verwer BJ, Bennink RJ, van Lienden KP, Gouma DJ, et al. Risk assessment of posthepatectomy liver failure using hepatobiliary scintigraphy and CT volumetry. J Nucl Med. 2007; 48:685–692. PMID:
17475954.

21. Kauffmann R, Fong Y. Post-hepatectomy liver failure. Hepatobiliary Surg Nutr. 2014; 3:238–246. PMID:
25392835.
22. Chapelle T, De Beeck BO, Huyghe I, Francque S, Driessen A, Roeyen G, et al. Future remnant liver function estimated by combining liver volumetry on magnetic resonance imaging with total liver function on 99m tc-mebrofenin hepatobiliary scintigraphy: can this tool predict post-hepatectomy liver failure? HPB (Oxford). 2016; 18:494–503. PMID:
27317953.

23. Stoffels B, Enkirch SJ, Websky MW, Vilz TO, Pantelis D, Manekeller S, et al. Posthepatectomy liver failure in extended liver resections: an overview based on a retrospective single-centre analysis. Zentralbl Chir. 2016; 141:405–414. PMID:
27135865.
24. Lee SY. Central hepatectomy for centrally located malignant liver tumors: a systematic review. World J Hepatol. 2014; 6:347–357. PMID:
24868328.

25. Shirabe K, Shimada M, Gion T, Hasegawa H, Takenaka K, Utsunomiya T, et al. Postoperative liver failure after major hepatic resection for hepatocellular carcinoma in the modern era with special reference to remnant liver volume. J Am Coll Surg. 1999; 188:304–309. PMID:
10065820.

26. Malinchoc M, Kamath PS, Gordon FD, Peine CJ, Rank J, Ter Borg PC. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology. 2000; 31:864–871. PMID:
10733541.

27. Cucchetti A, Ercolani G, Vivarelli M, Cescon M, Ravaioli M, La Barba G, et al. Impact of model for endXMLLink_XYZstage liver disease (MELD) score on prognosis after hepatectomy for hepatocellular carcinoma on cirrhosis. Liver Transpl. 2006; 12:966–971. PMID:
16598792.
28. Bruix J, Llovet JM. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology. 2002; 35:519–524. PMID:
11870363.

29. Teh SH, Christein J, Donohue J, Que F, Kendrick M, Farnell M, et al. Hepatic resection of hepatocellular carcinoma in patients with cirrhosis: model of end-stage liver disease (MELD) score predicts perioperative mortality. J Gastrointest Surg. 2005; 9:1207–1215. PMID:
16332475.

30. Kaneko K, Shirai Y, Wakai T, Yokoyama N, Akazawa K, Hatakeyama K. Low preoperative platelet counts predict a high mortality after partial hepatectomy in patients with hepatocellular carcinoma. World J Gastroenterol. 2005; 11:5888–5892. PMID:
16270404.

31. Bennett JJ, Blumgart LH. Assessment of hepatic reserve prior to hepatic resection. J Hepatobiliary Pancreat Surg. 2005; 12:10–15. PMID:
15754093.

32. Tomimaru Y, Eguchi H, Gotoh K, Kawamoto K, Wada H, Asaoka T, et al. Platelet count is more useful for predicting post-hepatectomy liver failure at surgery for hepatocellular carcinoma than indocyanine green clearance test. J Surg Oncol. 2016; 113:565–569. PMID:
26751258.

33. Mullen JT, Ribero D, Reddy SK, Donadon M, Zorzi D, Gautam S, et al. Hepatic insufficiency and mortality in 1,059 noncirrhotic patients undergoing major hepatectomy. J Am Coll Surg. 2007; 204:854–862. PMID:
17481498.

34. Li B, Yu Y, He TF, Fan J, Wu ZQ, Zhou J, et al. Value of the conventional liver function tests in the assessment of hepatic reserve. Chin J Hepatobiliary Surg. 2011; 17:805–808.
35. Shen Y, Shi G, Huang C, Zhu X, Chen S, Sun H, et al. Prediction of post-operative liver dysfunction by serum markers of liver fibrosis in hepatocellular carcinoma. PLoS One. 2015; 10:e0140932. PMID:
26501145.

36. Motoyama H, Kobayashi A, Yokoyama T, Shimizu A, Furusawa N, Sakai H, et al. Liver failure after hepatocellular carcinoma surgery. Langenbeck Arch Surg. 2014; 399:1047–1055.

37. An S, Rong W, Wang L, Wu F, Yu W, Feng L, et al. Analysis of clinicopathological features and prognosis between alpha-fetoprotein negative and positive hepatocellular carcinoma patients after ro radical hepatectomy. Zhonghua Zhong Liu Za Zhi. 2015; 37:308–311. PMID:
26462898.
38. Abbasi A, Bhutto AR, Butt N, Munir SM. Corelation of serum alpha fetoprotein and tumor size in hepatocellular carcinoma. J Pak Med Assoc. 2012; 62:33–36. PMID:
22352098.
39. Lai Q, Melandro F, Pinheiro RS, Donfrancesco A, Fadel BA, Levi Sandri GB, et al. Alpha-fetoprotein and novel tumor biomarkers as predictors of hepatocellular carcinoma recurrence after surgery: a brilliant star raises again. Int J Hepatol. 2012; 2012:893103. PMID:
22792474.

40. Toro A, Ardiri A, Mannino M, Arcerito MC, Mannino G, Palermo F, et al. Effect of pre-and post-treatment α-fetoprotein levels and tumor size on survival of patients with hepatocellular carcinoma treated by resection, transarterial chemoembolization or radiofrequency ablation: a retrospective study. BMC Surg. 2014; 14:40. PMID:
24993566.

41. Kohla MA, Abu Zeid MI, Al-Warraky M, Taha H, Gish RG. Predictors of hepatic decompensation after TACE for hepatocellular carcinoma. BMJ Open Gastroenterol. 2015; 2:e000032.

42. Kadalayil L, Benini R, Pallan L, O'beirne J, Marelli L, Yu D, et al. A simple prognostic scoring system for patients receiving transarterial embolisation for hepatocellular cancer. Ann Oncol. 2013; 24:2565–2570. PMID:
23857958.

43. Nanashima A, Sumida Y, Abo T, Tanaka K, Takeshita H, Hidaka S, et al. Clinicopathological and intraoperative parameters associated with postoperative hepatic complications. Hepatogastroenterology. 2007; 54:839–843. PMID:
17591075.
44. Nanashima A, Tobinaga S, Abo T, Nonaka T, Takeshita H, Hidaka S, et al. Reducing the incidence of post-hepatectomy hepatic complications by preoperatively applying parameters predictive of liver function. J Hepatobiliary Pancreat Sci. 2010; 17:871–878. PMID:
20734210.

45. Imamura H, Sano K, Sugawara Y, Kokudo N, Makuuchi M. Assessment of hepatic reserve for indication of hepatic resection: decision tree incorporating indocyanine green test. J Hepatobiliary Pancreat Surg. 2005; 12:16–22. PMID:
15754094.

46. Seyama Y, Kokudo N. Assessment of liver function for safe hepatic resection. Hepatol Res. 2009; 39:107–116. PMID:
19208031.

47. Yokoyama Y, Ebata T, Igami T, Sugawara G, Mizuno T, Yamaguchi J, et al. The predictive value of indocyanine green clearance in future liver remnant for posthepatectomy liver failure following hepatectomy with extrahepatic bile duct resection. World J Surg. 2016; 40:1440–1447. PMID:
26902630.

48. Uchida Y, Furuyama H, Yasukawa D, Nishino H, Ando Y, Hata T, et al. Hepatectomy based on future liver remnant plasma clearance rate of indocyanine green. HPB Surg. 2016; 2016:7637838. PMID:
27418717.

49. Leung U, Simpson AL, Araujo RL, Gönen M, McAuliffe C, Miga MI, et al. Remnant growth rate after portal vein embolization is a good early predictor of post-hepatectomy liver failure. J Am Coll Surg. 2014; 219:620–630. PMID:
25158914.

50. Narita M, Oussoultzoglou E, Fuchshuber P, Pessaux P, Chenard MP, Rosso E, et al. What is a safe future liver remnant size in patients undergoing major hepatectomy for colorectal liver metastases and treated by intensive preoperative chemotherapy? Ann Surg Oncol. 2012; 19:2526–2538. PMID:
22395987.

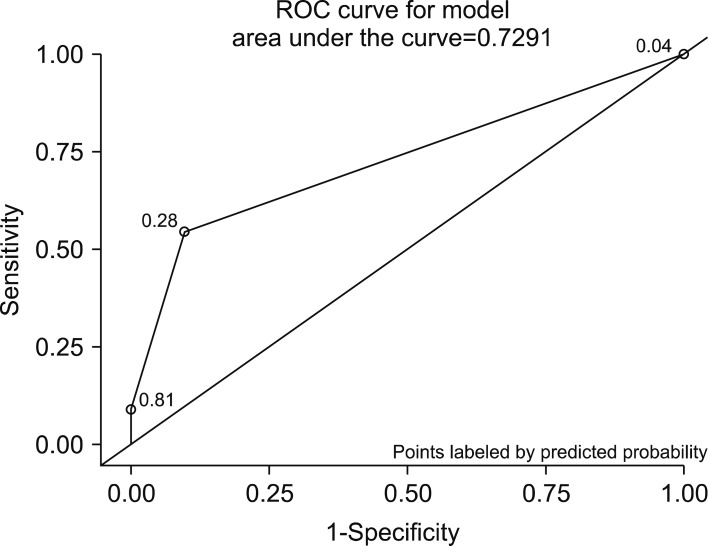
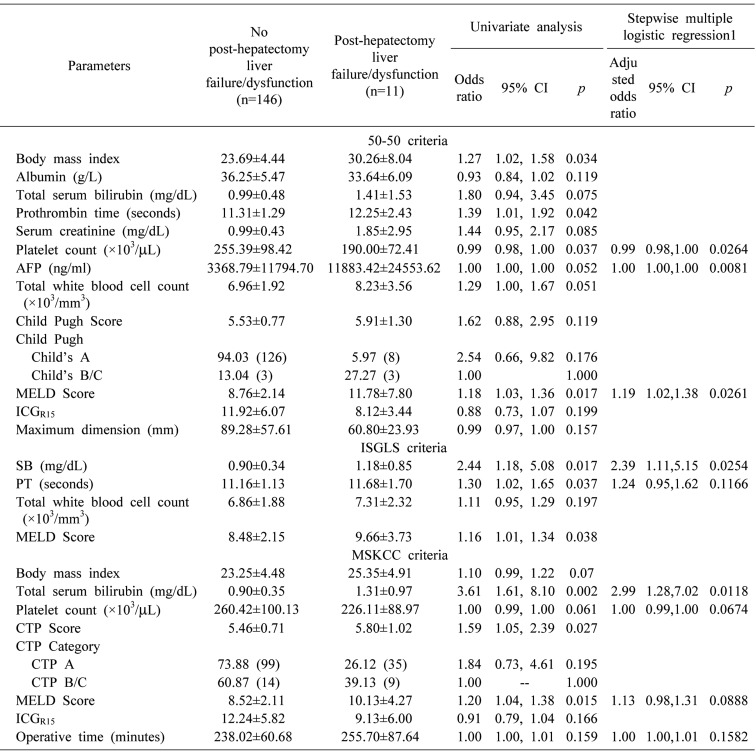
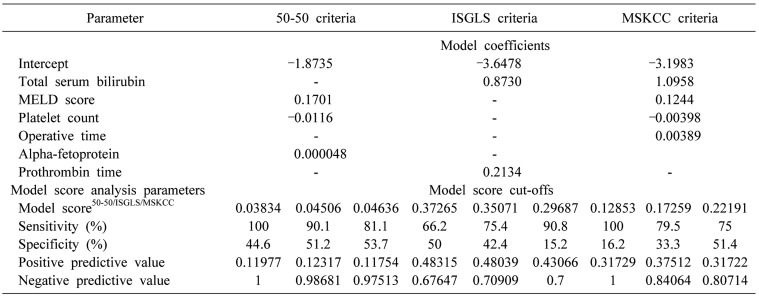
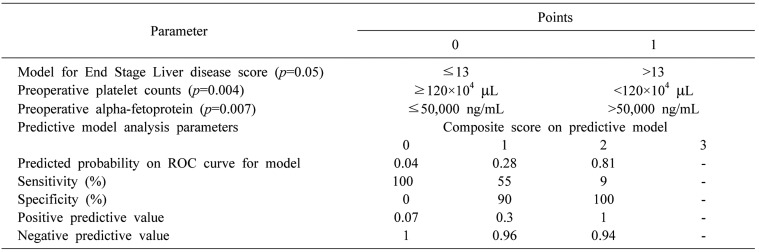
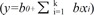 ) from which the probability of PHLF/I was calculated (p=ey/(1+ey)). Receiver operating characteristic (ROC) curves were plotted for each of the above models, where area under curve (AUC) was calculated to determine their validity as a predictive model.
) from which the probability of PHLF/I was calculated (p=ey/(1+ey)). Receiver operating characteristic (ROC) curves were plotted for each of the above models, where area under curve (AUC) was calculated to determine their validity as a predictive model.



 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download