Abstract
Backgrounds/Aims
Colorectal cancer is found with liver metastases about 20–25% due to characteristics of cancer itself. Approximately 20% of liver metastases are found to be resectable. The objective of this study was to evaluate short-term outcomes of patients who received liver resection with colorectal cancer operation in our center by laparoscopic surgery or open surgery.
Methods
Short-term outcomes of laparoscopic surgery of liver resection (LSLR) group who underwent liver resection for colorectal liver metastases (CRLM) at a single institute from 2013 to 2016 were compared to those of open surgery of liver resection (OSLR) group.
Results
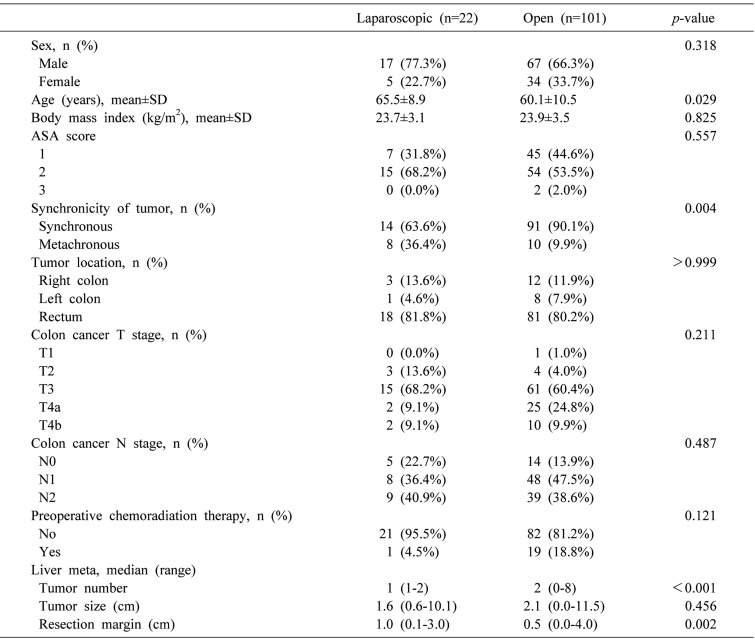
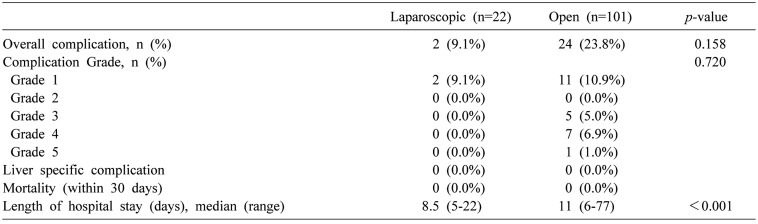
A total of 123 patients underwent liver resection for CRLM, including 101 (82.1%) patients in the OSLR group and 22 (17.9%) patients in the LSLR group. There were significant differences in tumor characteristics between the two groups, including synchronous and metachronous (p=0.004), tumor number (p<0.001), and tumor margin (p=0.002). For postoperative outcomes, only the length of hospital stay (LOS) was significantly different between the two groups (8.5 days in LSLR vs. 11 days in OSLR, p<0.001). There was no significant difference in overall rate of postoperative complications between the two groups (9.1% in LSLR vs. 23.8% in OSLR, p=0.158).
Colorectal cancer was the third most frequent cancer in Korea in 2013.1 In particular, colorectal cancer is found with liver metastases in 20–25% of patients due to characteristics of cancer itself. Approximately 20% of patients with liver metastases are resectable.23 The 5-year survival rate for patients with resectable liver metastases has increased to 30%–50%.45 Preoperative chemotherapy can reduce the extent of liver metastases if they are unresectable. It might make them resectable, thus increasing the survival rate of patients.6 Colorectal cancer research has also enhanced chemotherapeutic regimens and increased survival rates.
Surgical options for colorectal liver metastasis (CRLM) include simultaneous resection or a staged operation. A simultaneous resection of CRLM can reduce patient burden and the length of hospital stay. However, morbidity can increase slightly because the surgery is a major resection and the patient may have undergone preoperative chemotherapy.7 In addition, simultaneous operation can only be performed in patients with resectable CRLM which is a limitation. On the other hand, a staged operation for CRLM may lead to fewer postoperative complications and a better outcome. However, this has not been demonstrated yet. A staged operation involves two major operations. This will increase the length of hospital stay and cost.4
Laparoscopic liver surgery was first reported in 1996.8 Since then, laparoscopic devices and techniques have been substantially improved and surgeon's range of choice to perform a laparoscopic operation has also extended. Laparoscopic operation has been generalized to almost all surgeries. It has become one of standard surgical choices. Laparoscopic liver resection is also getting standardized because of advances in ablation techniques, anesthetic techniques, and postoperative care. However, laparoscopic liver surgery is difficult for surgeons who are newly performing it because it involves complex procedures and anatomy with difficulties for uncontrolled hemorrhage and a stressful learning curve to overcome.9 Some reports have shown encouraging results of laparoscopic liver resection.91011 Therefore, many hepatobiliary surgeons are attempting to use this approach. These challenges have been applied to living donor hepatectomy. A report has shown that laparoscopic living donor hepatectomy is feasible.12 Recently, researchers have been attempting to accomplish minimally invasive liver resection surgery. There have been efforts to decrease incision sizes during open surgery. Major liver resection is traditionally performed with an inverted L incision or an inverted T incision. One study has compared minimal open surgery with a previous large incision surgery13 and found that minimal open surgery is feasible for the surgeon and patient.
In this report, we evaluated short-term outcomes and complications of patients who received liver resection for colorectal cancer in our center using laparoscopic or open surgical technique. This study was funded by National Cancer Center, Korea (Grant No.1810203-1).
A total of 123 patients underwent liver resection for CRLM at a single institute from March 1, 2013 to June 31, 2016. We reviewed and analyzed electronic medical records of all patients retrospectively. These patients were divided into two groups according to whether they underwent laparoscopic surgery for liver resection (LSLR, n=22) or open surgery for liver resection (OSLR, n=101).
All patients were diagnosed with CRLM. Ninety-seven patients were diagnosed with liver metastasis at the same time colorectal cancer was diagnosed by preoperative computed tomography or magnetic resonance imaging. The other 26 patients were diagnosed with liver metastasis during outpatient follow-up after they underwent colorectal cancer surgery. A simultaneous operation was our first choice if the liver metastasis was resectable. The first choice for an unresectable metastasis was colorectal cancer resection followed by adjuvant chemotherapy. Regarding the second choice, patients received neo-adjuvant chemotherapy first and CRLM was then resected next. In this report, we evaluated only differences in outcomes and complications between LSLR and OSLR groups. Therefore, differences between synchronous and metachronous groups were not evaluated. Differences between simultaneous and staged operation were not evaluated neither.
Colorectal cancer operation was basically done laparoscopically or with open conversion or open surgery depending on cancer status. Liver resection was performed only after colorectal surgery. OSLR incision only used an upper midline incision. LSLR used an additional port incision because the port incision for colorectal surgery was not suitable for laparoscopic liver resection. Additionally, three cases were converted from LSLR to OSLR. These cases were included in the OSLR group.
Extracorporeal Pringle maneuver was performed to reduce bleeding during liver parenchymal transection in the LSLR procedure. An additional port was inserted in the left lower quadrant of the abdominal wall and the hepatoduodenal ligament was encircled with umbilical tape. The tape was externalized through a long plastic pipe used as an injection extension and the external tip was clamped with mosquito forceps. This Pringle maneuver was performed every 20 min and relieved every 5 min during liver parenchymal transection. Liver transection was performed with a cavitron ultrasonic surgical aspirator (CUSA) and energy devices such as Thunderbeat. Small vessels were ligated with CUSA electrocautery or laparoscopic clip devices. The main glissonian pedicle and hepatic vein were ligated with endo-GIA stapler.
Colorectal cancer tumor staging was analyzed according to the American Joint Committee on Cancer, 7th edition. Postoperative complications were defined as any deviation from the normal postoperative course that needed additional physiotherapy, interventions, or endoscopic or additional surgical management. If a patient had multiple complications, only complication with the highest grade was included. Complications were graded using the Clavien-Dindo scale ranging from grade 1 (mild) to grade 5 (death).11 Postoperative complications were checked for 30 days. Postoperative mortality was defined as death within 30 days after surgery. If a patient was discharged within 30 days, postoperative complications or outcomes were examined in the outpatient clinic until 30 days. Operation record was reviewed for perioperative analysis. We reviewed liver resection operation record for estimated blood loss, operation time, major or minor resection, and need for perioperative transfusions. Operation time of liver resection in simultaneous operation was calculated by excluding time of primary colon operation from total operation time. Size, number, and resection margin of the metastatic tumor were reviewed when pathological diagnosis of the liver resection specimen was confirmed.
Categorical variables are presented as sex, American Society of Anesthesiologists (ASA) score, synchronicity, tumor location, and colon cancer T, N stage. These variables were compared using Fisher's exact and χ2 tests. Continuous variables are presented as age, body mass index (BMI), metastatic tumor number, size, and resection margin. These variables were compared using independent sample t-test and Mann-Whitney U test. Risk factors found to be statistically significant in univariate analysis were entered into a multivariate binary logistic regression model to predict major complications after surgery. Variables found to be significant on univariate analysis (p<0.100) were considered in a multivariable model. Differences were considered significant at p-value<0.05. All statistical analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) and R version 3.3.3 software.
Propensity score matching using patients' clinicopathologic variables including sex, age, BMI, synchronicity of tumor, colon cancer T, N stage, the number of tumors, and liver resection range was performed to reduce differences between the two groups for more accurate comparison. However, there were no statistically significant differences in perioperative data, disease-free survival (DFS), or overall survival (OS) between the two groups. A comparison of DFS after propensity score matching is shown in Supplementary Table 1.
A total of 123 patients underwent liver resection for CRLM, including 101 (82.1%) patients in the OSLR group and 22 (17.9%) patients in the LSLR group. No significant differences in sex, ASA score, BMI, or maximum size of liver metastases were detected. Statistically significant differences were observed in age, synchronicity of tumor, metastatic tumor number, and tumor margin (p=0.029, p=0.004, p<0.001, and p=0.002, respectively). No significant differences were observed in primary colon cancer T stage or N stage between the two groups (Table 1).
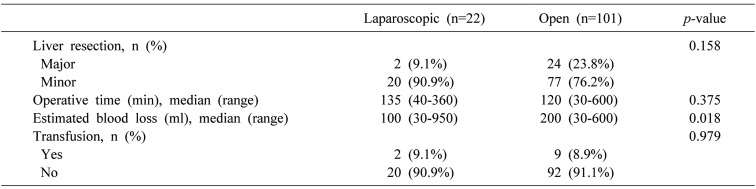
Operation time and transfusion rate were not statistically different between the two groups (p=0.375, p=0.979, respectively). The ratio of major to minor liver surgery was not significantly different between the two groups either (p=0.158). However, estimated blood loss in the LSLR group was less than that in the OSLR group (p=0.018) (Table 2).
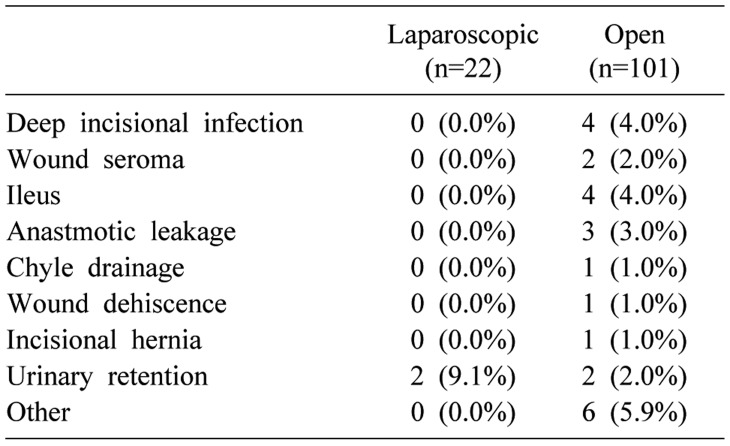
Postoperative outcomes and types of complication are listed in Tables 3 and 4. There was no mortality observed in 30 days after surgery. Only one patient developed a severe complication with colonic anastomotic leakage. However, the patient recovered well after reoperation. There were no statistically significant differences in postoperative outcomes such as complication grade between the two groups. Only the length of hospital stay was significantly shorter in the LSLR group compared to that in the OSLR group (8.5 days in LSLR group vs. 11 days in OSLR group, p<0.001).
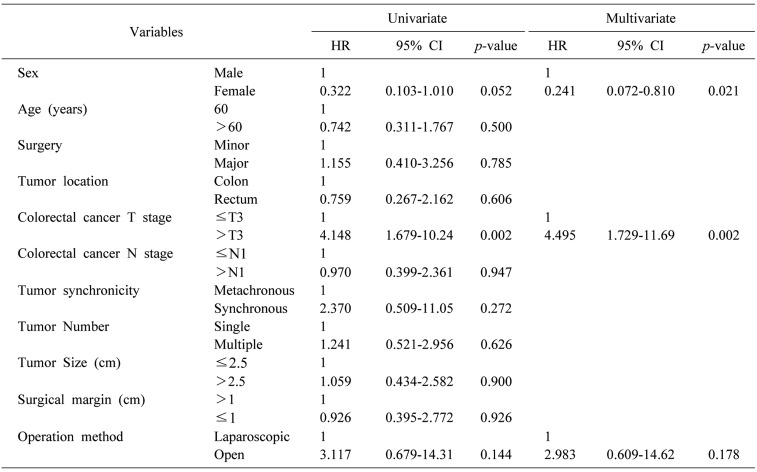
Univariate analysis was performed to evaluate risk factors for postoperative complications (Table 5). Some variables such as sex and primary colorectal cancer T stage showed statistical significance in both univariate and multivariate analyses. However, operation method was not statistically significant in these analyses.
We compared outcomes between LSLR and OSLR in patients with CRLM. The number of liver metastases, resection margin, and length of hospital stay were significantly different between the two groups. Our results suggest that LSLR may be favorable for patients with CRLM and a small number of metastases.
A favorable long-term oncological outcome or cure for cancer is expected only if patients with CRLM will undergo surgical resection of the cancerous region. However, whether a simultaneous resection or a staged operation should be performed remains controversial. Several reports have shown that a staged operation is safer with fewer perioperative complications.141516 However, with development in surgical devices and techniques, pre- and post-operative management, and anesthetic techniques, safer and more aggressive liver resection operations are now possible17 and minimally invasive surgery for liver resection is now available for patients with CRLM. Laparoscopic surgery is a minimally invasive surgery. Most new devices and techniques are now focused on laparoscopic surgery. In fact, abdominal surgery is now mostly performed laparoscopically. It is also desired by most patients. Hepatobiliary surgery has progressed slower compared to other minimally invasive operations primarily due to complexity of procedures and anatomy, difficulties with uncontrolled hemorrhage, and stressful learning curve.9 Some reports have shown favorable outcomes. However, those results usually represent activity at highly experienced centers. With recent progress in techniques and a better understanding of hepatobiliary surgery, minimally invasive surgery has shown encouraging results.1011
Several reports have compared LSLR to OSLR in patients with CRLM.181920 Most of these studies have reported that patients who undergo LSLR have less perioperative blood loss, fewer complications, less consumption of analgesics, and shorter length of hospital stay with comparable long-term outcomes. Laparoscopic living donor hepatectomy has now been performed during liver transplantation. Laparoscopic liver resection will take the place of living donor hepatectomy during liver transplantation in the future.12
We performed OSLR with only an upper midline incision, even for a major resection. Major liver resection such as right hemihepatectomy is typically performed with large incision such as an inverted L incision. This is not special as an upper midline incision is generalized for liver resection. One study has compared a minimal incision and inverted L incision.13 All single OSLR procedures were performed with only an upper midline incision which was a minimal incision for a right hemihepatectomy during major liver resection.
Several limitations of our study should be mentioned. The major limitation was that LSLR group sample size was too small compared to the OSLR group. Although propensity score matching analysis was applied to overcome this major limitation and analysis showed no statistical differences between the two groups, this small sample size made this study hard to be generally acceptable. Additionally, this study revealed only small advantage of laparoscopic liver resection available for selected group. Another important controversy about simultaneous versus staged operation was not evaluated in this study. Further well-designed large study is needed to reveal the actual advantage of laparoscopic liver resection. In addition, this study was designed retrospectively. Therefore, there might be selection bias.
In conclusion, no significant differences in postoperative outcomes were observed between LSLR and OSLR except length of hospital stay, the number of liver metastasis, and the resection margin. Our results suggest that LSLR may be favorable in highly selected patients with CRLM having small metastatic region.
ACKNOWLEDGEMENTS
This study was approved by the institutional review board of National Cancer Center (No. NCC2017-0092).
References
1. Oh CM, Won YJ, Jung KW, Kong HJ, Cho H, Lee JK, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2013. Cancer Res Treat. 2016; 48:436–450. PMID: 26987395.

2. Leporrier J, Maurel J, Chiche L, Bara S, Segol P, Launoy G. A population-based study of the incidence, management and prognosis of hepatic metastases from colorectal cancer. Br J Surg. 2006; 93:465–474. PMID: 16523446.

3. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013; 63:11–30. PMID: 23335087.

4. Tanaka K, Shimada H, Matsuo K, Nagano Y, Endo I, Sekido H, et al. Outcome after simultaneous colorectal and hepatic resection for colorectal cancer with synchronous metastases. Surgery. 2004; 136:650–659. PMID: 15349115.

5. de Santibañes E, Fernandez D, Vaccaro C, Quintana GO, Bonadeo F, Pekolj J, et al. Short-term and long-term outcomes after simultaneous resection of colorectal malignancies and synchronous liver metastases. World J Surg. 2010; 34:2133–2140. PMID: 20532766.

6. Litvak DA, Wood TF, Tsioulias GJ, Chung M, Chawla SP, Foshag LJ, et al. Systemic irinotecan and regional floxuridine after hepatic cytoreduction in 185 patients with unresectable colorectal cancer metastases. Ann Surg Oncol. 2002; 9:148–155. PMID: 11888871.

7. Karoui M, Penna C, Amin-Hashem M, Mitry E, Benoist S, Franc B, et al. Influence of preoperative chemotherapy on the risk of major hepatectomy for colorectal liver metastases. Ann Surg. 2006; 243:1–7. PMID: 16371728.

8. Azagra JS, Goergen M, Gilbart E, Jacobs D. Laparoscopic anatomical (hepatic) left lateral segmentectomy-technical aspects. Surg Endosc. 1996; 10:758–761. PMID: 8662435.

9. Tzanis D, Shivathirthan N, Laurent A, Abu Hilal M, Soubrane O, Kazaryan AM, et al. European experience of laparoscopic major hepatectomy. J Hepatobiliary Pancreat Sci. 2013; 20:120–124. PMID: 23053354.

10. Dagher I, Di Giuro G, Dubrez J, Lainas P, Smadja C, Franco D. Laparoscopic versus open right hepatectomy: a comparative study. Am J Surg. 2009; 198:173–177. PMID: 19268902.

11. Pearce NW, Di Fabio F, Teng MJ, Syed S, Primrose JN, Abu Hilal M. Laparoscopic right hepatectomy: a challenging, but feasible, safe and efficient procedure. Am J Surg. 2011; 202:e52–e58. PMID: 21861979.

12. Han HS, Cho JY, Yoon YS, Hwang DW, Kim YK, Shin HK, et al. Total laparoscopic living donor right hepatectomy. Surg Endosc. 2015; 29:184. PMID: 24993170.

13. Suh SW, Lee KW, Lee JM, Choi Y, Yi NJ, Suh KS. Clinical outcomes of and patient satisfaction with different incision methods for donor hepatectomy in living donor liver transplantation. Liver Transpl. 2015; 21:72–78. PMID: 25348280.

14. Bolton JS, Fuhrman GM. Survival after resection of multiple bilobar hepatic metastases from colorectal carcinoma. Ann Surg. 2000; 231:743–751. PMID: 10767796.

15. Reddy SK, Pawlik TM, Zorzi D, Gleisner AL, Ribero D, Assumpcao L, et al. Simultaneous resections of colorectal cancer and synchronous liver metastases: a multi-institutional analysis. Ann Surg Oncol. 2007; 14:3481–3491. PMID: 17805933.

16. Muangkaew P, Cho JY, Han HS, Yoon YS, Choi Y, Jang JY, et al. Outcomes of simultaneous major liver resection and colorectal surgery for colorectal liver metastases. J Gastrointest Surg. 2016; 20:554–563. PMID: 26471363.

17. Silberhumer GR, Paty PB, Temple LK, Araujo RL, Denton B, Gonen M, et al. Simultaneous resection for rectal cancer with synchronous liver metastasis is a safe procedure. Am J Surg. 2015; 209:935–942. PMID: 25601556.

18. Castaing D, Vibert E, Ricca L, Azoulay D, Adam R, Gayet B. Oncologic results of laparoscopic versus open hepatectomy for colorectal liver metastases in two specialized centers. Ann Surg. 2009; 250:849–855. PMID: 19801934.

19. Kazaryan AM, Marangos IP, Røsok BI, Rosseland AR, Villanger O, Fosse E, et al. Laparoscopic resection of colorectal liver metastases: surgical and long-term oncologic outcome. Ann Surg. 2010; 252:1005–1012. PMID: 21107111.
20. Inoue Y, Hayashi M, Tanaka R, Komeda K, Hirokawa F, Uchiyama K. Short-term results of laparoscopic versus open liver resection for liver metastasis from colorectal cancer: a comparative study. Am Surg. 2013; 79:495–501. PMID: 23635585.

SUPPLEMENTARY MATERIAL
Supplementary Table 1
Univariable analysis of prognostic factors for disease-free survival in propensity score matched patients with colorectal cancer liver metastases




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download