1. Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011; 365:2110–2121. PMID:
22129255.

2. Katz P, Julian L, Tonner MC, Yazdany J, Trupin L, Yelin E, et al. Physical activity, obesity, and cognitive impairment among women with systemic lupus erythematosus. Arthritis Care Res (Hoboken). 2012; 64:502–510. PMID:
22337564.

3. Rodríguez Huerta MD, Trujillo-Martín MM, Rúa-Figueroa Í, Cuellar-Pompa L, Quirós-López R, Serrano-Aguilar P. Spanish SLE CPG Development Group. Healthy lifestyle habits for patients with systemic lupus erythematosus: a systemic review. Semin Arthritis Rheum. 2016; 45:463–470. PMID:
26522137.

4. Pettersson S, Boström C, Eriksson K, Svenungsson E, Gunnarsson I, Henriksson EW. Lifestyle habits and fatigue among people with systemic lupus erythematosus and matched population controls. Lupus. 2015; 24:955–965. PMID:
25697772.

5. O'Dwyer T, Durcan L, Wilson F. Exercise and physical activity in systemic lupus erythematosus: a systematic review with meta-analyses. Semin Arthritis Rheum. 2017; 47:204–215. PMID:
28477898.
6. Urowitz MB, Gladman D, Ibañez D, Fortin P, Sanchez-Guerrero J, Bae S, et al. Accumulation of coronary artery disease risk factors over three years: data from an international inception cohort. Arthritis Rheum. 2008; 59:176–180. PMID:
18240193.

7. Eriksson K, Svenungsson E, Karreskog H, Gunnarsson I, Gustafsson J, Möller S, et al. Physical activity in patients with systemic lupus erythematosus and matched controls. Scand J Rheumatol. 2012; 41:290–297. PMID:
22651371.

8. Boström C, Dupré B, Tengvar P, Jansson E, Opava CH, Lundberg IE. Aerobic capacity correlates to self-assessed physical function but not to overall disease activity or organ damage in women with systemic lupus erythematosus with low-to-moderate disease activity and organ damage. Lupus. 2008; 17:100–104. PMID:
18250132.

9. Petri M, Spence D, Bone LR, Hochberg MC. Coronary artery disease risk factors in the Johns Hopkins Lupus Cohort: prevalence, recognition by patients, and preventive practices. Medicine (Baltimore). 1992; 71:291–302. PMID:
1522805.
10. Costenbader KH, Wright E, Liang MH, Karlson EW. Cardiac risk factor awareness and management in patients with systemic lupus erythematosus. Arthritis Rheum. 2004; 51:983–988. PMID:
15593366.

11. dos Santos Fde M, Borges MC, Correia MI, Telles RW, Lanna CC. Assessment of nutritional status and physical activity in systemic lupus erythematosus patients. Rev Bras Reumatol. 2010; 50:631–638. PMID:
21243304.
12. Ahn GE, Chmiel JS, Dunlop DD, Helenowski IB, Semanik PA, Song J, et al. Self-reported and objectively measured physical activity in adults with systemic lupus erythematosus. Arthritis Care Res (Hoboken). 2015; 67:701–707. PMID:
25251755.

13. Legge A, Blanchard C, Hanly JG. Physical activity and sedentary behavior in patients with systemic lupus erythematosus and rheumatoid arthritis. Open Access Rheumatol. 2017; 9:191–200. PMID:
29184453.

14. Tan EM, Cohen AS, Fries JF, Masi AT, McShane DJ, Rothfield NF, et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1982; 25:1271–1277. PMID:
7138600.

15. Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997; 40:1725.

16. Richter P, Werner J, Heerlein A, Kraus A, Sauer H. On the validity of the Beck Depression Inventory. A review. Psychopathology. 1998; 31:160–168. PMID:
9636945.
17. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992; 30:473–483. PMID:
1593914.
18. Alarcón GS, McGwin G Jr, Bartolucci AA, Roseman J, Lisse J, Fessler BJ, et al. Systemic lupus erythematosus in three ethnic groups. IX. Differences in damage accrual. Arthritis Rheum. 2001; 44:2797–2806. PMID:
11762940.
19. Gladman D, Ginzler E, Goldsmith C, Fortin P, Liang M, Urowitz M, et al. Systemic lupus international collaborative clinics: development of a damage index in systemic lupus erythematosus. J Rheumatol. 1992; 19:1820–1821. PMID:
1362779.
20. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987; 40:373–383. PMID:
3558716.

21. Craig CL, Marshall AL, Sjöström M, Bauman AE, Booth ML, Ainsworth BE, et al. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003; 35:1381–1395. PMID:
12900694.

22. Chun MY. Validity and reliability of Korean version of international physical activity questionnaire short form in the elderly. Korean J Fam Med. 2012; 33:144–151. PMID:
22787536.

23. Hernández-Hernández V, Ferraz-Amaro I, Díaz-González F. Influence of disease activity on the physical activity of rheumatoid arthritis patients. Rheumatology (Oxford). 2014; 53:722–731. PMID:
24369410.
24. Mancuso CA, Perna M, Sargent AB, Salmon JE. Perceptions and measurements of physical activity in patients with systemic lupus erythematosus. Lupus. 2011; 20:231–242. PMID:
21183562.

25. Abrahão MI, Gomiero AB, Peccin MS, Grande AJ, Trevisani VF. Cardiovascular training vs. resistance training for improving quality of life and physical function in patients with systemic lupus erythematosus: a randomized controlled trial. Scand J Rheumatol. 2016; 45:197–201. PMID:
26525835.

26. Miossi R, Benatti FB, Lúciade de Sá Pinto A, Lima FR, Borba EF, Prado DM, et al. Using exercise training to counterbalance chronotropic incompetence and delayed heart rate recovery in systemic lupus erythematosus: a randomized trial. Arthritis Care Res (Hoboken). 2012; 64:1159–1166. PMID:
22438298.
27. Pate RR, Pratt M, Blair SN, Haskell WL, Macera CA, Bouchard C, et al. Physical activity and public health. A recommendation from the Centers for Disease Control and Prevention and the American College of Sports Medicine. JAMA. 1995; 273:402–407. PMID:
7823386.

28. Franco OH, de Laet C, Peeters A, Jonker J, Mackenbach J, Nusselder W. Effects of physical activity on life expectancy with cardiovascular disease. Arch Intern Med. 2005; 165:2355–2360. PMID:
16287764.

29. Svenungsson E, Jensen-Urstad K, Heimbürger M, Silveira A, Hamsten A, de Faire U, et al. Risk factors for cardiovascular disease in systemic lupus erythematosus. Circulation. 2001; 104:1887–1893. PMID:
11602489.

30. Volkmann ER, Grossman JM, Sahakian LJ, Skaggs BJ, FitzGerald J, Ragavendra N, et al. Low physical activity is associated with proinflammatory high-density lipoprotein and increased subclinical atherosclerosis in women with systemic lupus erythematosus. Arthritis Care Res (Hoboken). 2010; 62:258–265. PMID:
20191526.

31. Lewandowski LB, Kaplan MJ. Update on cardiovascular disease in lupus. Curr Opin Rheumatol. 2016; 28:468–476. PMID:
27227346.

32. Peutz-Kootstra CJ, de Heer E, Hoedemaeker PJ, Abrass CK, Bruijn JA. Lupus nephritis: lessons from experimental animal models. J Lab Clin Med. 2001; 137:244–260. PMID:
11283519.

33. Voorhees JL, Tarr AJ, Wohleb ES, Godbout JP, Mo X, Sheridan JF, et al. Prolonged restraint stress increases IL-6, reduces IL-10, and causes persistent depressive-like behavior that is reversed by recombinant IL-10. PLoS One. 2013; 8:e58488. PMID:
23520517.

34. Kasawara KT, Cotechini T, Macdonald-Goodfellow SK, Surita FG, Pinto E, Tayade C, et al. Moderate exercise attenuates lipopolysaccharide-induced inflammation and associated maternal and fetal morbidities in pregnant rats. PLoS One. 2016; 11:e0154405. PMID:
27124733.

35. Aqel SI, Hampton JM, Bruss M, Jones KT, Valiente GR, Wu LC, et al. Daily moderate exercise is beneficial and social stress is detrimental to disease pathology in murine lupus nephritis. Front Physiol. 2017; 8:236. PMID:
28491039.

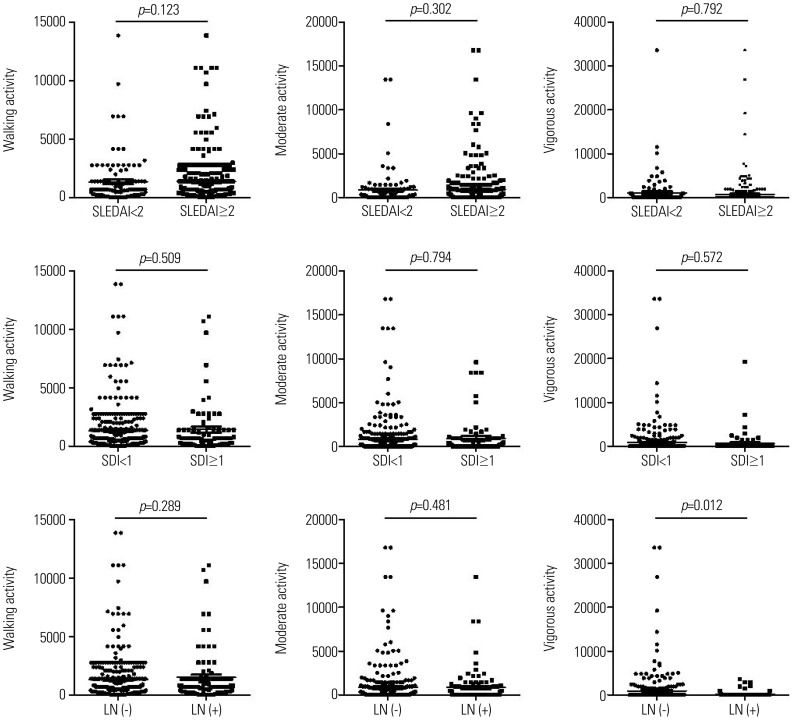
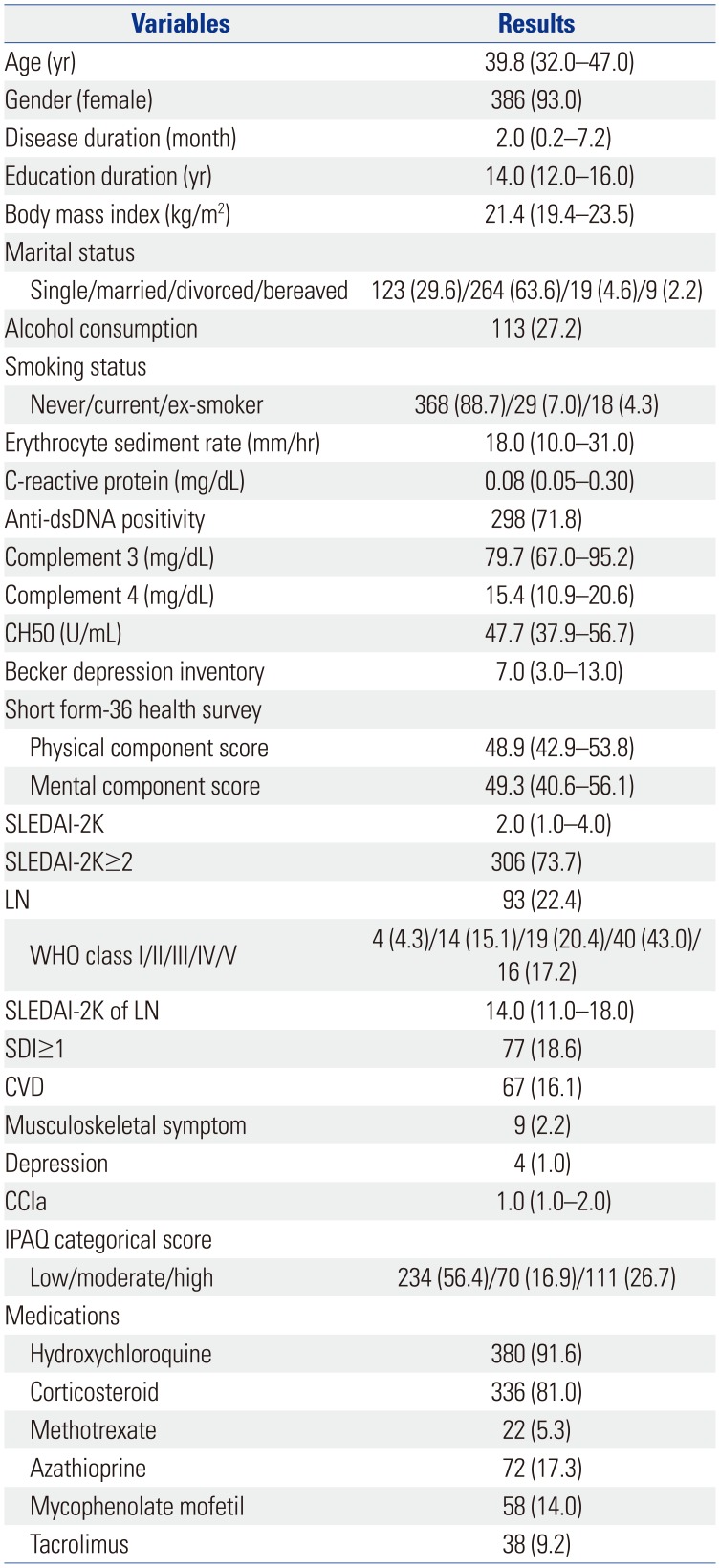
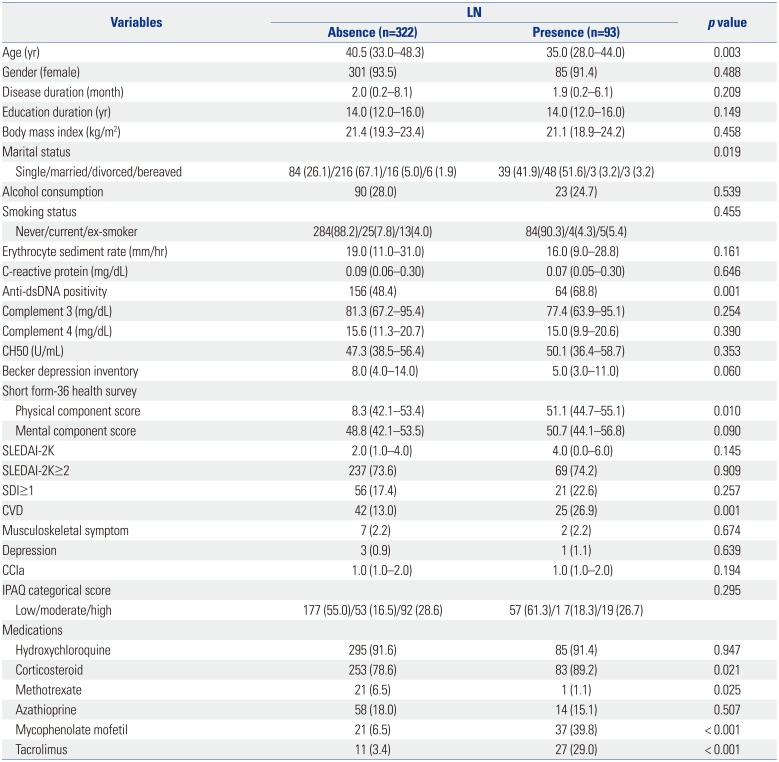
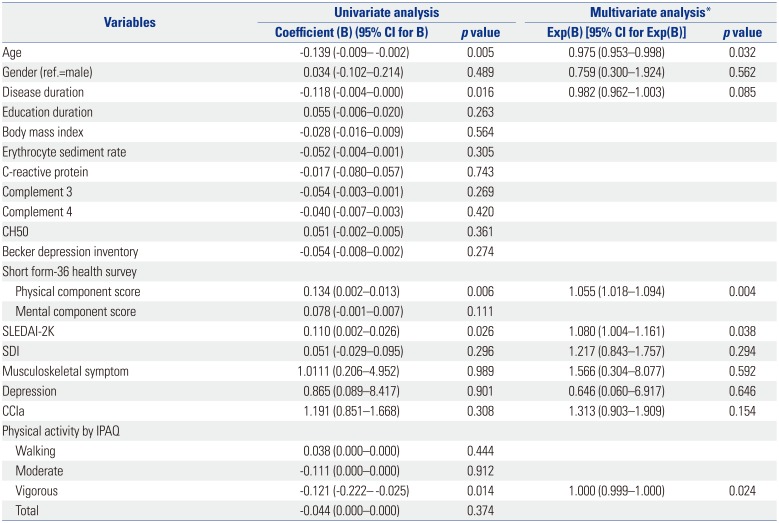




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download