Abstract
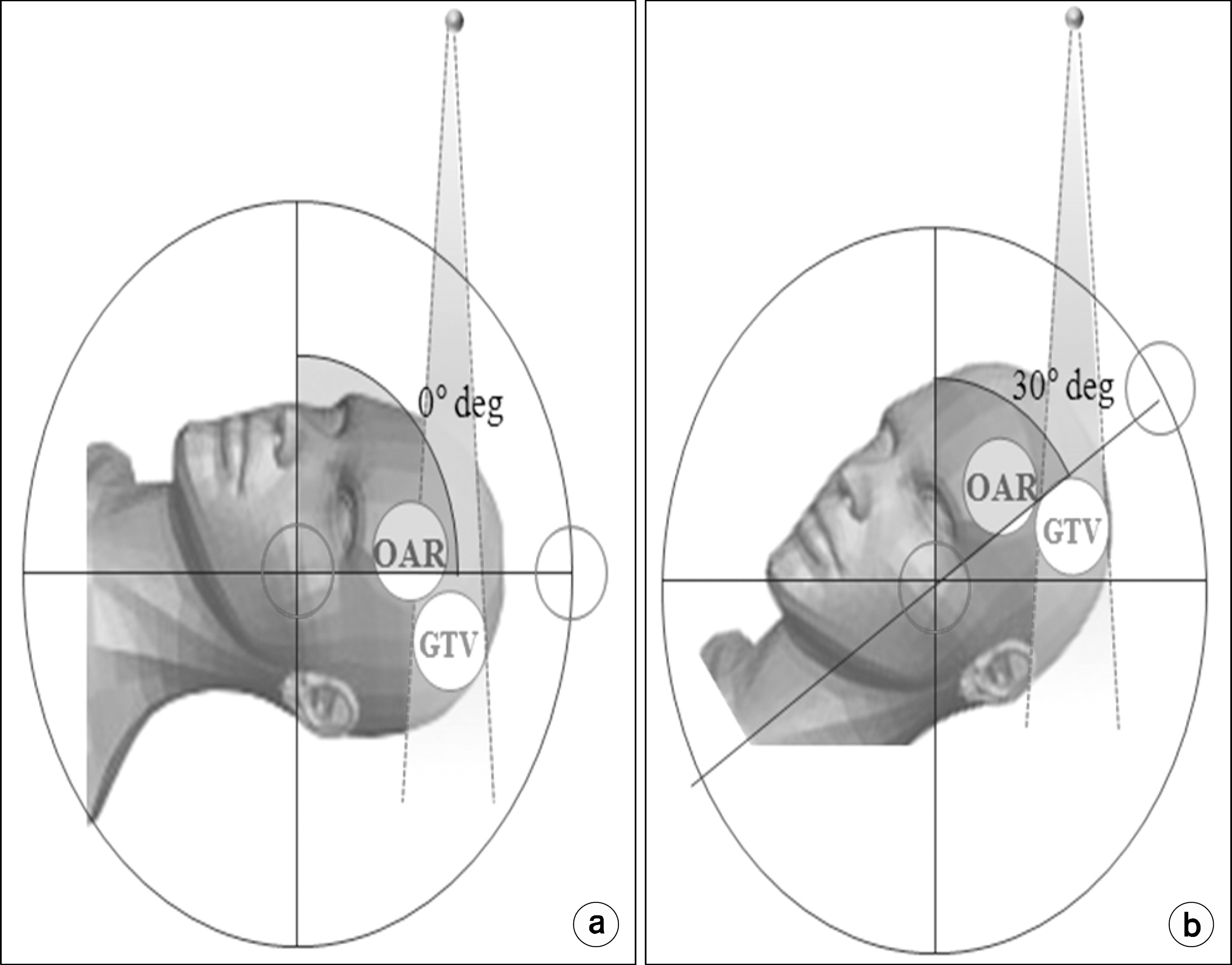
Since the head and neck region is densely located with organs at risk (OAR), OAR-sparing is an important issue in the treatment of head and neck cancers. This study— in which different treatment plans were performed varying the head tilt angle on brain tumor patients— investigates the optimal head elevation angle for sparing normal organs (e.g. the hippocampus) and further compares the dosimetric characteristics of different types of radiation equipment. we performed 3D conformal radiation therapy (3D-CRT), intensity-modulated radiation therapy (IMRT), and tomotherapy on 10 patients with brain tumors in the frontal lobe while varying the head tilt angle of patients to analyze the dosimetric characteristics of different therapy methods. In each treatment plan, 95% of the tumor volume was irradiated with a dose of 40 Gy in 10 fractions. The step and shoot technique with nine beams was used for IMRT, and the same prescription dose was delivered to the tumor volume for the 3D-CRT and tomotherapy plans. The homogeneity index, conformity index, and normal tissue complication probability (NTCP) were calculated. At a head elevation angle of 30 o, conformity of the isodose curve to the target increased on average by 53%, 8%, and 5.4%. In 3D-CRT, the maximum dose received by the brain stem decreased at 15 o, 30 o, and 40 o, compared to that observed at 0 o. The NTCP value of the hippocampus observed in each modality was the highest at a head and neck angle of 0 o and the lowest at 30 o. This study demonstrates that the elevation of the patients' head tilt angle in radiation therapy improves the target region's homogeneity of dose distribution by increasing the tumor control rate and conformity of the isodose curve to the target. Moreover, the study shows that the elevation of the head tilt angle lowers the NTCP by separating the tumor volume from the normal tissues, which helps spare OARs and reduce the delivered dose to the hippocampus.
References
1. Lee SW, Back GM, Yi BY, et al. Preliminaryresultsofa phaseI-IIstudyofsimultaneousmodulatedacceleratedradio-therapyfornondisseminatednasopharyngealcarcinoma. Int.J. Radiat.Oncol.Biol.Phys. 65:152–160. 2006.
2. Shiau AC, Lai PL, Liang JA, et al. Dosimetricverification ofsurfaceandsuperficialdosesforheadandneckIMRTwith differentPTVshrinkagemargins.Med.Phys. 38:1435–1443. 2011.
3. Pezner RD, Archambeau JO. Braintoleranceunit: Ameth-odtoestimateriskofradiationbraininjuryforvariousdose schedules.Int.J.Radiat.Oncol.Biol.Phys. 7:397–402. 1981.
4. Milano MT, Constine LS, et al. NormalTissueTolerance Dose Metrics for Radiation The rapyof Major Organs. Semin. Radiat.Oncol. 17:131–140. 2007.
5. Sahgal A, Barani LJ, Jr JN, et al. PrescriptionDose GuidelineBasedonPhysicalCriterionforMultipleMetastatic BrainTumorsTreatedWithStereotacticRadiosurgery.Int.J. Radiat.Oncol.Biol.Phys. 78:605–608. 2010.
6. Jeraj R, Mackie TR, Balog J, et al. Radiationcharacter-isticsofhelicaltomotherapy.Med.Phys. 31:396–404. 2004.
7. Kan MW, Cheung JY, Leung LH, et al. Theaccuracyof dosecalculationsbyanisotropicanalyticalalgorithmsforstereo-tacticradiotherapyinnasopharyngealcarcinoma.Phys.Med. Biol. 56:397–413. 2011.
8. Park SH, Park HC, Oh DH, et al. Multi-institutional ComparisonofIntensitymodulatedradiationTherapy(IMRT) PlanningstrategiesandPlanningResultsforNasopharyngeal Cancer.J.Korean.Med.Sci. 24:pp.248–255. 2009.
10. Marsh JC, Godbole R, Diaz AZ, et al. Sparing of the hippocampus, limbic circuitandneural stem cell compartment during partial brain radio therapy for glioma:: Adosimetricfeasibility study.J.Med.Imag.Radiat.On. 55:pp.442–449. 2011.
11. Huang D, Xia P, Akazawa P, et al. Comparisonoftreatmentusingintensity-modulatedradiotherapyandthree-dimen-sionalconformalradiotherapyforparanasalsinuscarcinoma,Int. J.Radiat.Oncol.Biol.Phys.56,No. 1:pp.158–168. (. 2003.
12. Niroomand RA, Razavi R, Thobejane S, et al. Radiation doseperturbationattissue-titaniumdentalinterfacesinhead andneckcancerpatients.Int.J.Radiat.Oncol.Biol.Phys. 34:475–480. 1996.
13. Choi JY, Won YJ, Park JY, et al. Developmentofa ThermoplasticOralCompensatorforImprovingDoseUniformity inRadiationTherapyforHeadandNeckCancer.Progressin MedicalPhysics. 23:269–278. 2012.
14. Vesna Prokic, Nicole Wiedenmann, Fels F, et al. Whole BrainIrradiationWithHippocampalSparingandDoseEscalation onMultipleBrainMetastases: APlanningStudyonTreatment Concepts.Int.J.Radiat.Oncol.Biol.Phys.85,No. 1:pp.264–270. (. 2013.
15. Vinai Gondi, Tolakanahalli R, Mehta MP, et al. Hippo-campal-SparingWhole-BrainRadiotherapy: A “How-To” TechniqueUsingHelicalTomotherapyandLinearAccelerator-BasedIntensity-ModulatedRadiotherapy.Int.J.Radiat.Oncol. Biol.Phys. 78:,No.(4):pp.1244–1252. (. 2010.
16. Wang X, Zhang X, Dong L, et al. Effectivenessofnon-coplanarIMRTplanningusingaparallelizedmultiresolution beamangleoptimizationmethodforparanasalsinuscarcinoma. Int.J.Radiat.Oncol.Biol.Phys. 63:594–601. 2005.
17. Siglin J, Champ CE, Vakhnenko Y, et al. Optimizingpatientpositioningforintensitymodulatedradiationtherapyinhip-pocampal-sparingwholebrainradiationtherapy. Pract. Radiat. Oncol. 4:378–383. 2014.
18. Joseph KJ, Syme A, Small C, et al. Atreatmentplanning studycomparinghelicaltomotherapywithintensity-modulated radiotherapyforthetreatmentofanalcancer.Radiother.Oncol. 94:pp.60–66. 2010.
19. Lee FK, Yip CW, Cheung nkie Chun-hung, et al. Dosimetricdifferenceamongst3techniques:: TomoTherapy, sliding-windowintensity-modulatedradiotherapy(IMRT),and RapidArcradiotherapyinthetreatmentoflate-stagenasophar-yngealcarcinoma(NPC).Med.Dosim. 39:pp.44–49. 2014.
20. Alber M, Nusslin F. Optimizationofintensitymodulatedra-diotherapyunder constraintsforstaticanddynamicMLC delivery.Phys.Med.Biol. 46:3229–3239. 2001.
21. Chui H, Rangarajan A. Anewpointmatchingalgorithmfor non-rigidregistration.Comp.Vis.ImageUnd. 89:pp.114–141. 2003.
22. Semenenko VA. Lyman-Kutcher-BurmanNTCPmodel parametersforradiationpneumonitisandxerostomiabasedon combinedanalysisofpublishedclinicaldata.Phys.Med.Biol. 53:737–55. 2008.
23. G Luxton G, Keall PJ, King CR. Anewformulafornormal tissuecomplicationprobability(NTCP)asafunctionofequiv-alentuniformdose(EUD).Phys.Med.Biol. 53:23–36. 2008.
24. Kutcher GJ, Burman C, Brewster L, et al. Histogramre-ductionmethodforcalculatingcomplicationprobabilitiesfor three-dimensionaltreatmentplanningevaluations.Int.J.Radiat. Oncol.Biol.Phys. 21:137–146. 1991.
25. Makrs LB, Yorke ED, Jackson A, et al. UseofNormal TissueComplicationProbabilityModelsintheClinic.Int.J. Radiat.Oncol.Biol.Phys. 76:S10–S19. 2010.
26. Emami B, Lyman J, Brown A, et al. Toleranceofnormal tissuetotherapeuticirradiation.Int.J.Radiat.Oncol.Biol.Phys. 21:109–122. 1991.
Fig. 2.
Delineation of the hippocampus in MRI (Orange Line: Hippocampus, Light green Line: Hippocampus 3 mm expansion).

Fig. 3.
Dose distribution among the 3 different treatment techniques (a) 3D-CRT, (b) Linac-IMRT and (c) Tomotherapy.
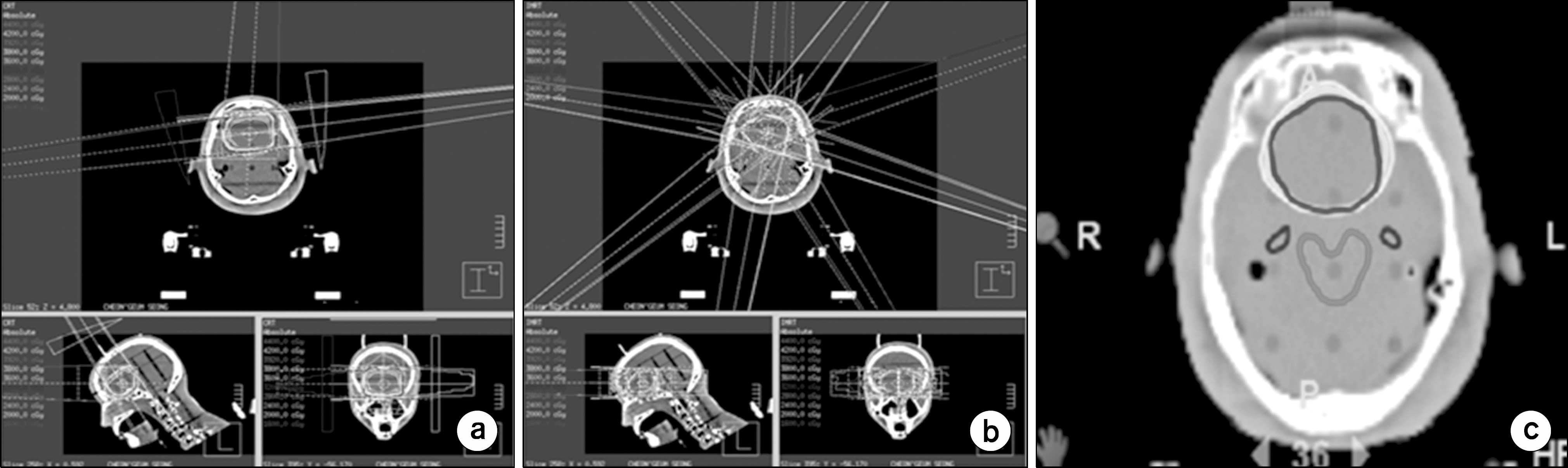
Fig. 4.
DVH (Dose Volume Histogram) among the 3 different treatment techniques. (a) 0 degree, (b) 30 degree.
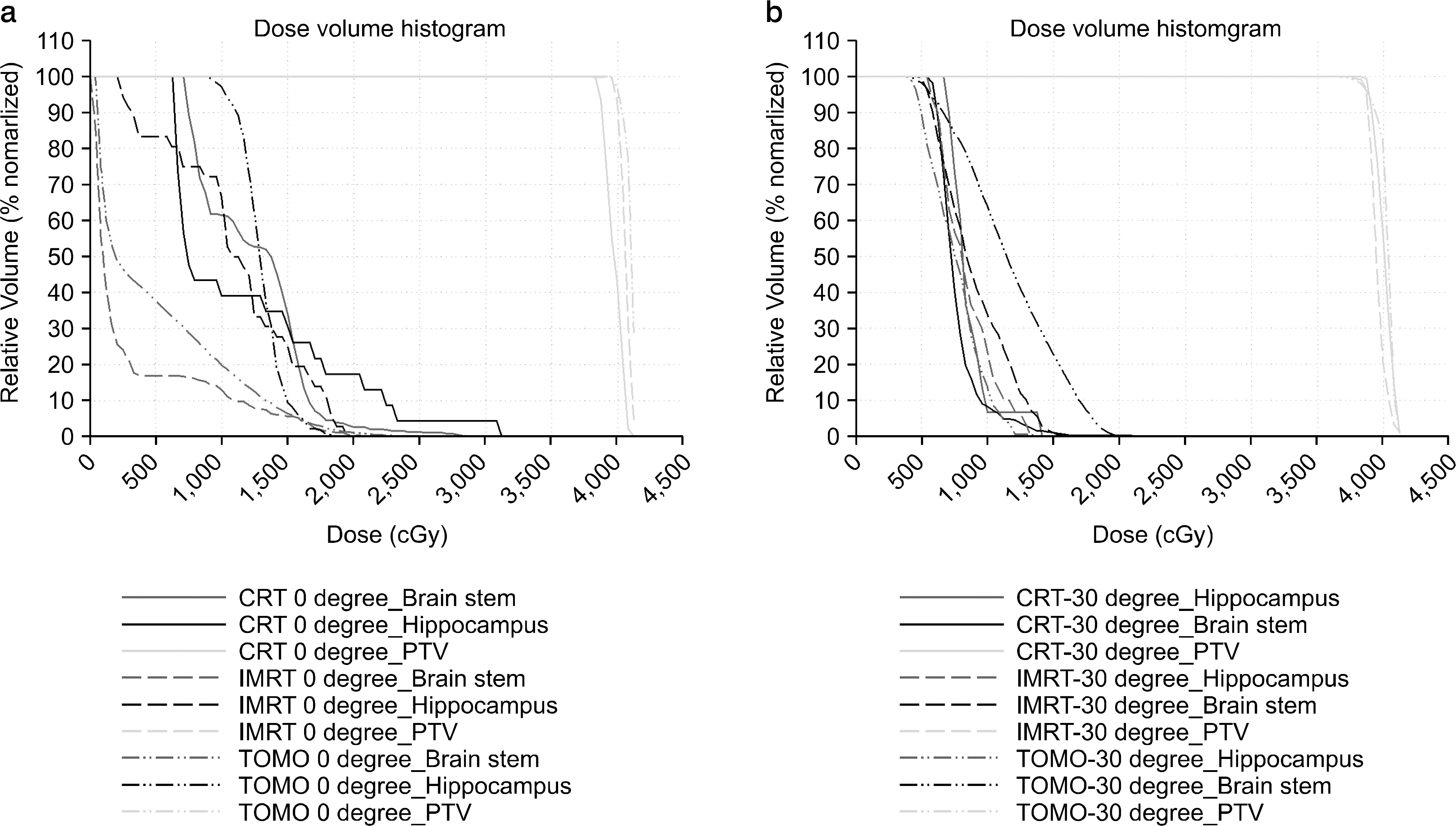
Table 1.
Characteristics of 10 patients participated in the study.
Table 2.
Comparison of the dose received by tumor in different radiation therapies with varied head elevation angles.
Table 3.
Comparison of the dose received by normal organs in different radiation therapies with varied head elevation angles.
Table 4.
Comparison of the dose received by the hippocampus with varied head elevation angles.
Table 5.
The NTCP value of normal organs in three different radiation therapies.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download