Abstract
ATP in quantity co-stored with neurotransmitters in the secretory vesicles of neurons, by being co-released with the neurotransmitters, takes an important role to modulate the stimulus-secretion response of neurotransmitters. Here, in this study, the modulatory effect of ATP was studied in Ca2+ channels of cultured rat adrenal chromaffin cells to investigate the physiological role of ATP in neurons. The Ca2+ channel current was recorded in a whole-cell patch clamp configuration, which was modulated by ATP. In 10 mM Ba2+ bath solution, ATP treatment (0.1 mM) decreased the Ba2+ current by an average of 36±6% (n=8), showing a dose-dependency within the range of 10−4∼10−1 mM. The current was recovered by ATP washout, demonstrating its reversible pattern. This current blockade effect of ATP was disinhibited by a large prepulse up to +80 mV, since the Ba2+ current increment was larger when treated with ATP (37±5%, n=11) compared to the control (25±3%, n=12, without ATP). The Ba2+ current was recorded with GTPγS, the non-hydrolyzable GTP analogue, to determine if the blocking effect of ATP was mediated by G-protein. The Ba2+ current decreased down to 45% of control with GTPγS. With a large prepulse (+80 mV), the current increment was 34±4% (n=19), which 25±3% (n=12) under control condition (without GTPγS). The Ba2+ current waveform was well fitted to a single-exponential curve for the control, while a double-exponential curve best fitted the current signal with ATP or GTPγS. In other words, a slow activation component appeared with ATP or GTPγS, which suggested that both ATP and GTPγS caused slower activation of Ca2+ channels via the same mechanism. The results suggest that ATP may block the Ca2+ channels by G-protein and this Ca2+ channel blocking effect of ATP is important in autocrine (or paracrine) inhibition of adrenaline secretion in chromaffin cell.
References
1. Augustine GJ, Neher E. Calcium requirments for secretion in bovine chromaffin cells. J Physiol. 450:247–271. 1992.
2. Boarder MR, Marriot D, Adams M. Stimulus secretion coupling in cultured chromaffin cells. Biochem Pharm. 56(1):163–167. 1987.

3. Albillos A, Artalejo AR, Lopez MG, Gandia L, Garcia AG, Carbone E. Calcium channel subtypes in cat chromaffin cells. J Physiol. 477:197–213. 1994.

5. Dunlap K, Fischbach GD. Neurotransmitters decrease the calcium conductance activated by depolarization of embryonic chick sensory neurons. J Physiol. 317:519–535. 1981.
6. Forscher P, Oxford GS. Modulation of calcium channels by norepinephrine in internally dialyzed avian sensory neurons. J Gen Physiol. 85:743–763. 1985.

7. Galvan M and Adams PR. Control of calcium current in rat sympathetic neurons by norepinephrine. Brain Res. 244:135–144. 1982.
8. Dolphin AC and Scott RH. Calcium channel currents and their inhibition by (−)-baclofen in rat sensory neurons: modulation by guanine nucleotides. J Physiol. 386:1–17. 1987.
9. Grassi F and Lux HD. Voltage-dependent GABA-induced modulation of calcium currents in chick sensory neurons. Neurosci Lett. 1015:113–119. 1989.
10. Menon-Johansson AS, Berrow N, Dolphin AC. G(o) transduces GABAB-receptor modulation by N-type calcium channels in cultured dorsal root ganglion neurons. Pflugers Arch. 425:335–343. 1993.
11. Harkins AB and Fox AP. Activation of purinergic receptors by ATP inhibits secretion in bovine adrenal chromaffin cells. Brain Res. 885:231–239. 2000.
12. Carbone E, Carabelli V, Casetti T, Baldelli P, Hernandez-Guijo JM, Giusta L. G-protein and cAMP-dependent Lchannel gating mechanism: a manifold system to control calcium entry in neurosecretory cells. Pflugers Arch. 442(6):801–813. 2001.
13. Diverse-Pierluissi M, Dunlap K, Westhead EW. Multiple actions of extracellular ATP on calcium currents in cultured bovine chromaffin cells. Proc Nat Acad Sci USA. 88:1261–1265. 1991.

14. Holz GG IV, Rane SG, Dunlap K. GTP-binding proteins mediate transmitter inhibition of voltage-dependent calcium channels. Nature. 319:670–672. 1986.

15. Kajikawa Y, Saitoh N, Takahashi T. GTP-binding protein beta gamma subunits mediate presynaptic calcium current inhibition by GABA(B) receptor. Proc Nat Acad Sci USA. 98:8054–8058. 2001.
16. Akaike A, Mine Y, Sasa M, Takaori S. Voltage and current clamp studies of M-and Nicotinic excitation of the rat chromaffin cells. J Pharmacol Exp Ther. 255:333–339. 1990.
17. Hamil OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high resolution current recording from cells and cell free membrane patches. Pflugers Arch. 391:85–100. 1981.
18. Marchetti C, Robello M. Guanosine-5'-O-(3-thiotriphosphate) modifies kinetics of voltage-dependent calcium current in chick sensory neurons. Biophys J. 56:1267–1272. 1989.

19. Artalejo CR, Rossie S, Perlman RL, Fox AP. Voltage dependent phosphorylation may recruit Ca2+ current facilitation in chromaffin cells. Nature. 358:63–66. 1992.
20. Dolphin AC. Facilitation of Ca2+ current in excitable cells. Trends Neurosci. 19(1):35–43. 1996.

21. R. Eckert and J.E. Chad: Inactivation of calcium channels: Prog. Biophysi. Mol. Biol. 44:215–267. 1984.
22. Powell AD, Teschemacher AG, Seward EP. P2Y purinoceptors inhibit exocytosis in adrenal chromaffin cells via modulation of voltage-operated calcium channels. J Neurosci. 20(2):606–616. 2000.

23. Dubyak GR, el-Moatassim C. Signal transduction via P2-purinergic receptors for extracellular ATP and other nucleotides. Am J Physiol. 265:C577–606. 1993.

24. Carabelli V, Hernández-Guijo JM, Baldelli P, Carbone E. Direct autocrine inhibition and cAMP-dependent potentiation of single L-type Ca2+ channels in bovine chromaffin cells. J Physiol. 532:(. (Pt 1):):. 73–90. 2001.
Fig. 1.
Peak current (IBa)-voltage (Vm) relationship recorded in adrenal chromaffin cell held at −80 mV activated by 150 ms depolarizing pulses applied in 10 mV steps. (a) Activation of Ba2+ currents. Upper and lower traces are voltage and current traces, respectively. (b) Voltage-current relationship of Ba2+ current from 25 cells.
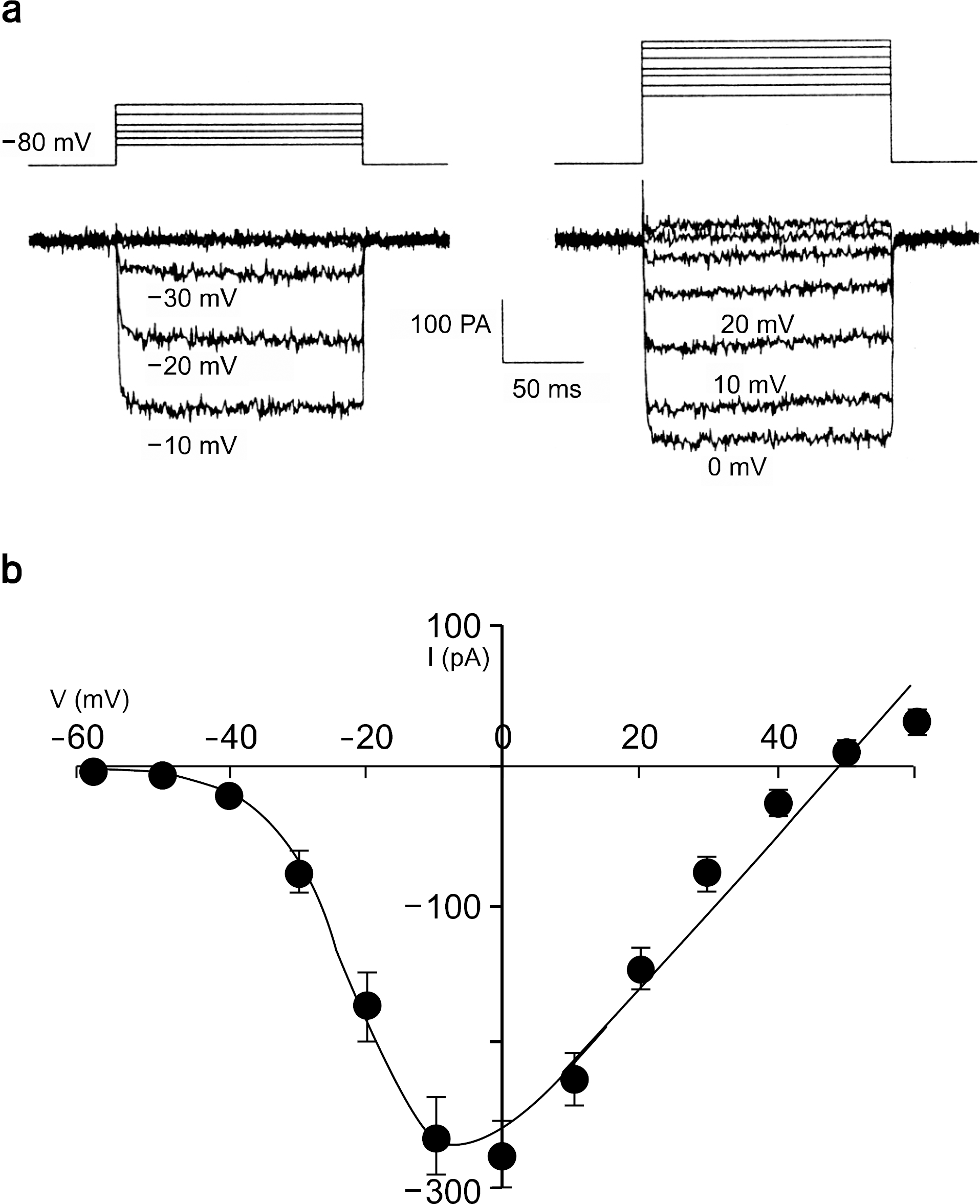
Fig. 2.
Effect of ATP on Barium current (IBa). (a) Upper trace is voltage protocol. Lower traces are control Ba2+ current trace (left), current trace when the external solution is switched to ATP-containing solution (middle), and current trace after wash-out of ATP (right), respectively. (b) Effect of ATP on Ba2+ current. Peak amplitude of control Ba2+ current was 281.37±53.53 pA while that with ATP was 168.05±28.6 pA, with wash-out of ATP was 243.45±70.7 pA (mean±S.E.M., n=8). ∗p-value<0.05. (c) Left: Current traces show Ba2+ currents recorded and concentration of ATP in one cell. Right: Time course of inhibitory effects of ATP on IBa. IBa was activated by 50 ms depolarization pulses from -80 mV to 0 mV applied at a frequency of 0.1 Hz. As indicated by the bars, ATP (0.1, 1, 10, 100 μM) was added to or removed (W/O, washed out) from the external solution. Concentration was successively changed from 0.1 μM to 100 μM. (d) Left: Current traces of control, and with and after washout of ATP. Curve fitting was performed and activation time constant (τ) was calculated in each trace. Right: Normalized current: normalization of raw current with respect to the final point. Upper trace (●) is current during ATP, middle (□), current after washout of ATP, and lower trace (■), control current.
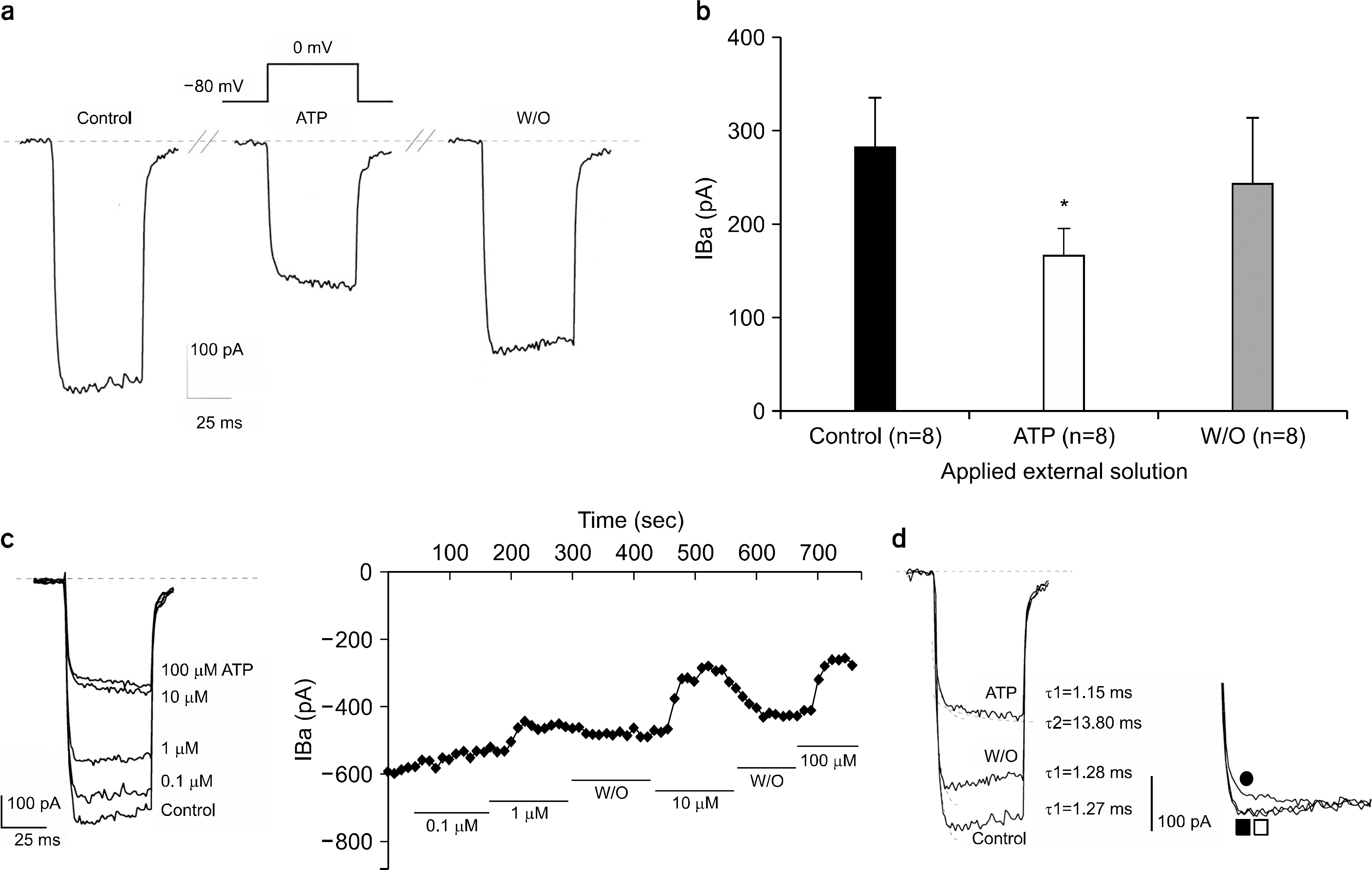
Fig. 3.
Effect of GTPγS (50 μM) on Ba2+ current. (a) Effect of GTPγS on gating kinetics of Ca2+ channel. Current trace of control condition vs intracellular GTPγS. Curve fitting was performed and activation time constant was calculated for each trace as shown. (b) Averaged results are shown (mean±S.E.M.) with the number of cells given in parentheses. Peak amplitude of IBa was 261.65±38.84 pA (n=11) while that with GTPγS was 118.78±17.59 pA (n=19). ∗p-value<0.05.
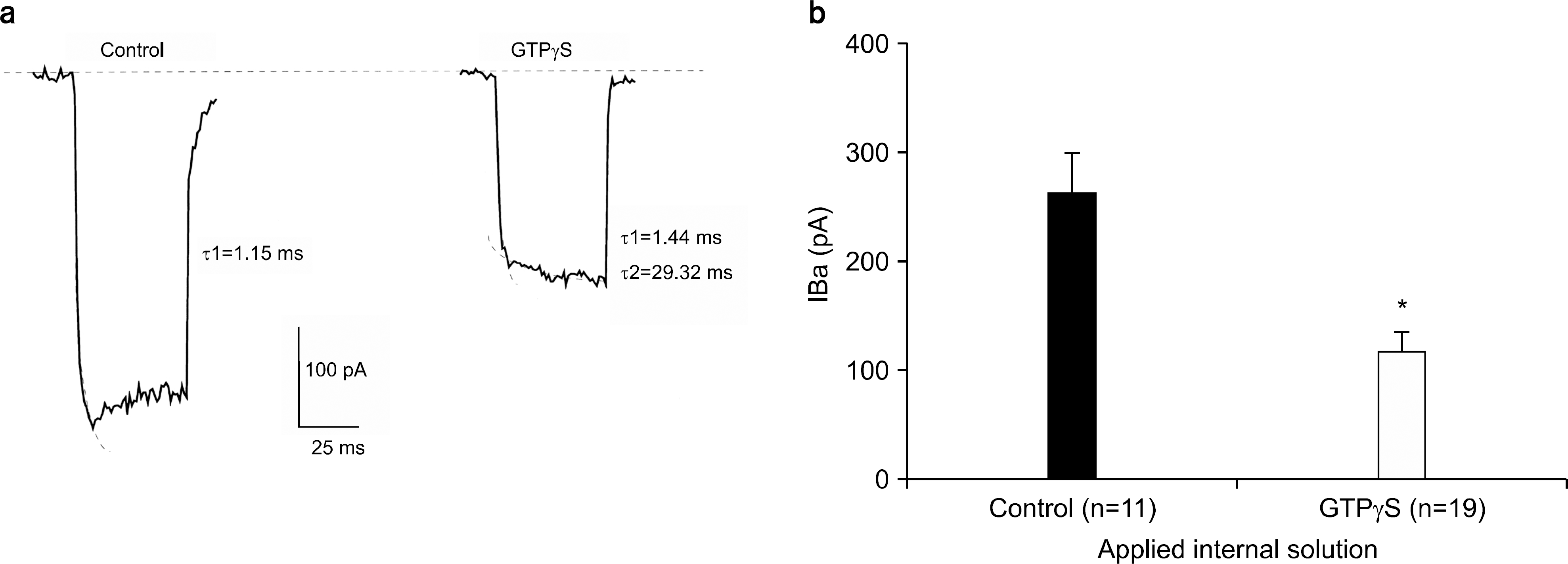
Fig. 4.
Prepulse induced facilitation recorded in different dialyzing conditions. (a) Left: Ba2+ currents were recorded without (●) and with (■) prepulse of 20 ms to +80 mV from Vh=−80 mV(upper trace) in control condition. Right: Records were obtained using the same pulse protocol but with extracellular ATP. Notice the pronounced inactivation on the facilitated currents that slowly relaxes to its control level. (b) Left: Ba2+ currents were recorded without (●) and with (■) prepulse of 30 ms to +80 mV from Vh=−80 mV(upper trace) in the application of intracellular GTPγS. Right: Records were obtained using same pulse protocol but with the application of intracellular GTPγS and additional extracellular ATP. There was no additional facilitation increment with GTPγS and ATP in comparison with GTPγS alone. (c) Prepulse induced facilitation in control condition was 25±3% (n=12) increase while that with extracellular ATP, with intracellular GTPγS, with intracellular GTPγS and extracellular ATP was 37±5% (n=11) increase, was 34±4% (n=19) increase, and 35±4% (n=8) increase, respectively. There was significant difference between control group and ATP-and or GTPγS- treated group. ∗p-value<0.05.
Fig. 5.
Schematic diagram of chromaffin cell stimulus-secretion coupling. The splanchnic nerve that innervates chromaffin cell release acetylcholine (Ach) that activates the nicotinic acetylcholine receptor (nAChR) that gates Na+, causing the membrane to depolarize (ΔV) sufficiently to trigger the action potentials to activate voltage-operated Ca2+ channels (VOCC). Entry of external Ca2+ increases the intracellular Ca2+ concentration ([Ca2+ i]) that triggers exocytosis and the release of chromaffin granule contents that contain adrenaline and ATP. The ATP may feedback to activate receptors that use the G protein to inhibit the VOCC. (Broken arrow represents the possibility).
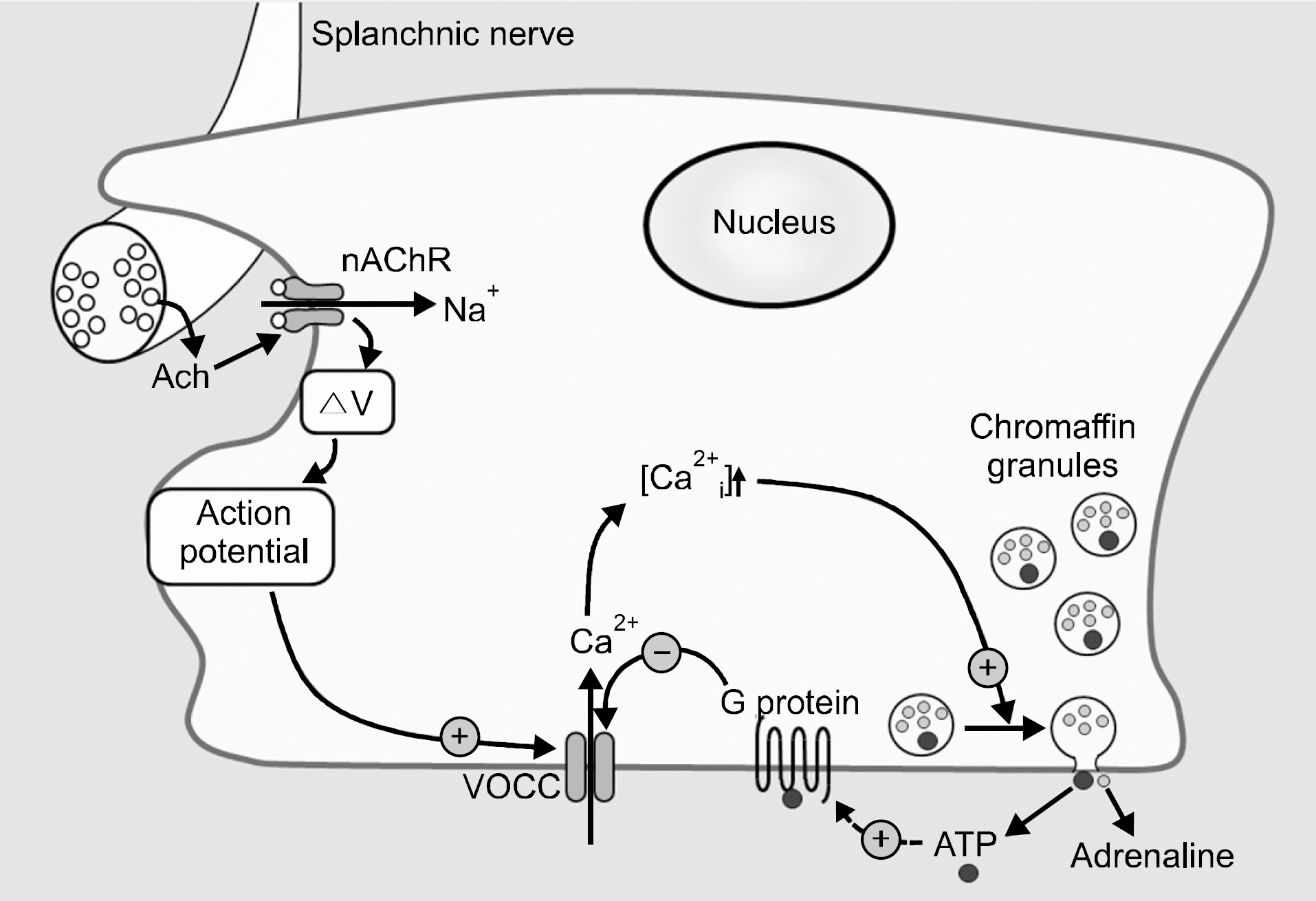
Table 1.
Activation time constants of Ca2+ channel when external solutions were switched to different condition on the same chromaffin cells. Each value is presented as mean±SEM.
| Condition of External Solution (N=8) | Activation Time Constant | |
|---|---|---|
| τ1 (ms) | τ2 (ms) | |
| Control | 1.24±0.15 | |
| ATP added | 1.17±0.22 | 13.5±2.75 |
| ATP wash-out | 1.26±0.17 | |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download