Abstract
Neurogenic weakness, ataxia and retinitis pigmentosa (NARP) syndrome is a rare maternally inherited mitochondrial disorder. Radiologic findings in NARP syndrome are varied; they include cerebral and cerebellar atrophy, basal ganglia abnormalities, and on rare occasions, leukoencephalopathy. This article describes an extremely rare case of NARP syndrome mimicking mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS).
Neurogenic weakness, ataxia, and retinitis pigmentosa (NARP) syndrome is caused by point mutations m.8993T>C or m.8993T>G in the mtDNA ATPase 6 gene. It often presents itself as a neurodegenerative disorder of the retina as well as the central and peripheral nervous system. The signs and symptoms of NARP include: sensorimotor axonal polyneuropathy, ataxia, retinitis pigmentosa, sensorineural hearing loss, seizures, and cognitive impairment (1). As of yet, strict diagnostic criteria for NARP syndrome has not been defined. The diagnosis is based on clinical features and the identification of a mtDNA pathogenic variant through molecular genetic testing (2). The radiological features of NARP syndrome are reported as cerebral and cerebellar atrophy, basal ganglia atrophy, and cystic/cavitary T2 hyperintensity of the bilateral basal ganglia and brain stem (34). This article examines an extremely rare case of NARP syndrome which manifested itself as diffuse non-vascular territory cortical involvement mimicking mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS).
A 43-year-old woman visited the emergency department with drowsy mentation, dyspnea, vomiting, and chills. She had been diagnosed with NARP syndrome 10 years prior at another hospital, based on clinical symptoms and a genetic study that detected the m.8993T>G mtDNA mutation. The woman had also been diagnosed with paranoid schizophrenia and a bilateral loss of vision. Upon her arrival, arterial blood gas analysis revealed a pO2 of 23.8 mmHg (normal, 80–90 mmHg), pCO2 of 27.1 mmHg (normal, 35–45 mmHg), pH of 7.304 (normal, 7.35–7.45), and HCO3 level of 13.4 mmol/L (normal, 23–29 mmol/L). A blood chemistry study showed a creatine kinase level of 1265 U/L (normal, 30–188 U/L), a lactate dehydrogenase level of 431 U/L (normal, 146–238 U/L), and a lactic acid level of 71 mg/dL (normal, 4.5–19.8 mg/dL). She was initially diagnosed as having respiratory failure, probably due to asphyxia or bronchopneumonia related to aspiration, and was subsequently intubated and connected to a ventilator. The patients low pO2 levels resolved immediately after ventilation was initiated.
A brain computed tomography (CT) scan revealed multifocal areas of low density in both the parietal and occipital lobes (Fig. 1a).
On day 2 of the patient's hospitalization, a diffusion-weighted magnetic resonance (DW-MR) image was obtained using a 1.5T system (Magnetom Avanto, Siemens AG, Erlangen, Germany). It showed diffusion restriction in both the parietal, occipital, and posterior temporal lobes, mainly comprising cortex and subcortical white matter. The DW-MR also showed severe cerebellar and brainstem atrophy. The bilateral basal ganglia and thalamus was not affected (Fig. 1b, c). Throughout the time imaging was performed, we were unaware of the patient's previous NARP syndrome diagnosis. Our differential diagnosis was acute stage MELAS; We also considered atypical hypoxic ischemic encephalopathy.
A follow-up blood test showed the patient had a lactic acid level of 34 mg/dL, and a pyruvic acid level of 0.141 mmol/L (normal, 0.034–0.102 mmol/L). In addition, a gene mutation test was performed and no MELAS associated genes were detected.
On day 20 of the patient's hospitalization, she underwent a follow-up magnetic resonance imaging (MRI) using a 3.0T system (Achieva, Philips Healthcare, Best, the Netherlands). T2-weighted imaging (T2-WI) and T2 fluid attenuated inversion recovery (T2-FLAIR) showed diffuse excessive high signal intensity of the cortex and subcortical white matter with gyral swelling in both frontal, parietal, temporal, and occipital lobes, which increased to some extent when compared to the previous DWI detection. Contrast enhanced T1-WI (CE T1-WI) showed diffuse gyral enhancement that was more prominent in newly developed lesions found on both fronto-parietal lobes. There was no visible abnormal signal intensity in the bilateral basal ganglia or thalamus (Fig. 2a, b, c, d).
Therefore, the patient was treated with conservative therapy and showed clinical improvement.
Patients' with the m.8993T>G mutation are divided into three groups; early onset Leigh's syndrome, late infancy syndrome of intermediate severity, and adult onset NARP syndrome (4). NARP syndrome patients show: proximal neurogenic muscle weakness with sensory neuropathy, ataxia, pigmentary retinopathy, seizures, learning difficulties, and dementia. The initial signs include particularly ataxia and learning difficulties, often manifested in early childhood. Some of the other signs include: short stature, sensorineural hearing loss, progressive external ophthalmoplegia, cardiac conduction defects, and mild anxiety disorder (4).
Currently, the radiologic findings of NARP syndrome are rarely reported, and thus are not well recognized. Holt et al. (1) and Thorburn et al. (2) reported that NARP syndrome patients show cerebral and cerebellar atrophy. Rojo et al. (5) noted that NARP syndrome patients show cortical-subcortical atrophy and bilateral putamen hypodensity on CT. Gelfand et al. (3) expressed that NARP syndrome patients show cystic/cavitary T2 hyperintensity in the bilateral basal ganglia and cerebral and cerebellar atrophy. Uziel et al. (4) reported that NARP syndrome patients show cerebellar and cerebral atrophy as well as symmetric T2 hyperintensity of the basal ganglia and brainstem.
During our literature search we discovered two cases of NARP syndrome with MRI findings that mimicked those found in MELAS. Uziel et al. (4) noted that one patient having NARP syndrome displayed cerebellar atrophy and abnormal signal intensity in the basal ganglia, which is typical of NARP syndrome. However, the patient also showed abnormal signal intensity in the right occipital and parietal cortex as well as displaying subcortical white matter similar to the features associated with MELAS. Renard and labauge. (6) reported on a patient that displayed pontocerebellar atrophy, putaminal signal change, and bilateral occipital leukoencephalopathy. However, in our case, the patient did not display basal ganglia lesions. She showed only pontocerebellar atrophy and extremely diffuse infarct-like leukoencephalopathy, which first occurred in the posterior portion of the brain parenchyma and later showed up in the anterior portion of the brain parenchyma. These manifestations were more akin to MELAS than any other previously reported cases of NARP syndrome.
One of the characteristic findings of MELAS is symmetric or asymmetric infarct-like lesions in both cerebral hemispheres that are not confined to the vascular territories and the predominant posterior. Also, in some cases, patients with MELAS can show cerebellar atrophy, which can make a diagnosis more challenging (7).
In our case, the patient also had a clinical hypoxia. A hypoxic ischemic brain injury primarily affects gray matter structures, such as the basal ganglia, cerebellar hemisphere, and cerebral cortex, and it also involves the thalamus and hippocampus (8). But this case spared both the basal ganglia and thalamus; it was not well matched with hypoxic ischemic encephalopathy.
In conclusion, NARP syndrome is diagnosed through clinical and laboratory tests; radiologic findings are ancillary. Thus, rare cases of atypical radiologic findings may surface from NARP syndrome, which can cause a misdiagnosis. When clinicians encounter NARP syndrome or other mitochondrial diseases, they should carefully question atypical radiologic findings.
Figures and Tables
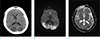 | Fig. 1(a) Low densities in both parietal and occipital lobes on a CT. (b, c) Diffusion restriction of both parietal, occipital, and posterior temporal lobes, mainly cortex and subcortical white matter. But, both basal ganglia and thalamus were not affected. |
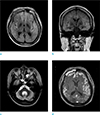 | Fig. 2(a, b) FLAIR axial and coronal scan showed diffuse hyperintensity in both frontal, parietal, temporal, and occipital lobes. Those lesions were increased to an extent when compared to previous diffusion weighted images. They also spared both the basal ganglia and thalamus. (c) T2-weighted image axial scan showed diffuse atrophied cerebellum. (d) Gd-enhanced T1-weighted image scan showed diffuse gyral enhancement in both cerebral hemispheres, more prominent in the newly developed lesions (both fronto-parietal lobes). |
References
1. Holt IJ, Harding AE, Petty RK, Morgan-Hughes JA. A new mitochondrial disease associated with mitochondrial DNA heteroplasmy. Am J Hum Genet. 1990; 46:428–433.
2. Thorburn DR, Rahman S. Mitochondrial DNA-associated Leigh syndrome and NARP. In : Pagon RA, Bird TD, Dolan CR, Stephens K, Adam MP, editors. GeneReviews™[Internet]. Seattle: University of Washington;1993–2013. last update: September 28, 2017. Accessed June 13, 2018. http://www.ncbi.nlm.nih.gov/books/NBK1173/.
3. Gelfand JM, Duncan JL, Racine CA, et al. Heterogeneous patterns of tissue injury in NARP syndrome. J Neurol. 2011; 258:440–448.

4. Uziel G, Moroni I, Lamantea E, et al. Mitochondrial disease associated with the T8993G mutation of the mitochondrial ATPase 6 gene: a clinical, biochemical, and molecular study in six families. J Neurol Neurosurg Psychiatry. 1997; 63:16–22.
5. Rojo A, Campos Y, Sanchez JM, et al. NARP-MILS syndrome caused by 8993 T>G mitochondrial DNA mutation: a clinical, genetic and neuropathological study. Acta Neuropathol. 2006; 111:610–616.
6. Renard D, Labauge P. Posterior leukoencephalopathy in NARP syndrome. Acta Neurol Belg. 2012; 112:417–418.

7. Scaglia F, Wong LJ, Vladutiu GD, Hunter JV. Predominant cerebellar volume loss as a neuroradiologic feature of pediatric respiratory chain defects. AJNR Am J Neuroradiol. 2005; 26:1675–1680.
8. Arbelaez A, Castillo M, Mukherji SK. Diffusion-weighted MR imaging of global cerebral anoxia. AJNR Am J Neuroradiol. 1999; 20:999–1007.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download