Introduction
Mature cystic teratomas (MCTs), commonly called dermoid cysts, account for approximately 20% of ovarian tumors and are one of the most common benign ovarian neoplasms [
1]. MCTs can occur in any age group. In particular, in younger women (teenagers to 20s), it has the highest incidence rate among various ovarian tumors [
2]. Pathologically, teratomas are classified into 3 groups: mature, immature, and monodermal (e.g., struma ovarii, carcinoid tumors, and neural tumors) [
3].
Most MCTs are asymptomatic, but may present with mass effects (e.g., abdominal distension, frequency), acute abdominal pain due to torsion, or rupture. Among MCTs, torsion is the most common complication with an incidence of 3.2–16% [
2]. In addition, rupture (1.2–3.8%) [
4], and malignant transformation (1–2%) [
5], may sometimes be adherent to the small and large bowel, causing intestinal obstruction or fistula formation [
6]. Particularly in women of reproductive age, ovarian torsion is of great clinical importance because of the possibility of an oophorectomy due to ovarian necrosis.
When an adnexa totally or partially rotates around its own vascular axis, adnexal torsion occurs [
7]. Adnexal torsion stimulates the venous and lymphatic blockage of ovarian parenchyma and includes complications such as massive congestion and hemorrhagic infarction followed by an arterial blockage that can cause gangrene and hemorrhagic necrosis [
8]. Adnexal torsion is generally unilateral [
9], and a high neutrophil to lymphocyte (N/L) ratio (>3) has been identified as a valuable marker in cases where it is difficult to diagnose adnexal torsion [
7]. A recent study reported that the combined measurement of cancer antigen 125 (CA125), cancer antigen 19-9 (CA19-9), and the N/L ratio provided an efficient method for the diagnosis of MCTs with torsion [
10].
Previous studies on torsion with MCTs have been limited and usually treated as a complication of MCTs. Besides, previous research on MCTs has focused on sonographic features, malignant transformation, and clinical presentation according to age [
11]. Therefore, this study sought to investigate the various factors affecting MCTs with torsion, and the factors were analyzed to see whether they appeared as a result of the torsion. Finally, the purpose of this study was to determine whether these factors can provide a more accurate and specific preoperative diagnosis of MCTs with torsion.
Materials and methods
Our study included patients with histologically proven MCTs, treated with surgery at the Gangnam Severance Hospital between July 2006 and May 2017. In total, 384 patients with MCTs surgery were classified as the torsion group (24 patients) or control group (360 patients). The presence, direction, degree, and necrosis of torsion were confirmed via surgical findings. Incidental or co-operation cases in appendicitis or cholecystitis surgery were excluded, and cases with malignancy-related or chronic systemic disease and cases accompanied with ovarian rupture or ectopic pregnancy were also excluded because white blood cell (WBC), neutrophil, and C-reactive protein (CRP) levels could have been affected.
In this study, gynecologic history, operation history, body mass index, body temperature, tumor markers (CA125, CA19-9) and laboratory tests such as WBC count, neutrophil count, lymphocyte count, N/L ratio, mean platelet volume, and CRP were measured. All patients underwent ultrasonography (US), and computed tomography (CT) or magnetic resonance imaging (MRI) were performed to rule out the possibility of malignancy or surgical abdomen including torsion in patients with acute abdominal pain. Tumor size was determined based on surgical records and preoperative images (US, CT, and MRI).
Statistics
Data were analyzed with SAS version 9.3 (SAS Institute, Cary, NC, USA). Continuous variables were calculated as mean ± standard deviation and compared with the use of Student's t-test. Categorical variables were calculated as rate (%) and compared using χ2 or Fisher's exact test as appropriate. A P-value < 0.05 was considered statistically significant.
Results
The clinical characteristics of patients are presented in
Table 1. Patients in the torsion group were significantly younger, with an average age of 26.8 ± 10.8 compared with 32.2 ± 11.6 (
P=0.009) in the control group. One (4.2%) patient in the torsion group had a previous abdominopelvic surgery history, compared with 26.1% of the control patients (
P=0.016). The incidental cases in the chief complaint were significantly different among 2 groups (torsion group, 4.2%; control group, 62.8%;
P<0.001). Twenty-three (95.8%) of the patients in the torsion group had abdominal pain, compared with 17.8% of the control patients (
P<0.001). WBC count, neutrophil count, and N/L ratio were also significantly higher in the torsion group than in the control group. No significant difference was observed among groups for tumor markers such as CA125 and CA19-9, mean platelet volume, and CRP. The patient as younger, without previous abdominopelvic surgery history, with obvious abdominal pain, the higher the WBC count, neutrophil count and the N/L ratio was close to the characteristics of torsion.
Table 1
Description of the patient characteristics between the torsion and control groups

|
Characteristics |
Torsion (n=24) |
Control (n=360) |
P-value |
|
Age (yr) |
26.9±10.8 |
33.2±11.6 |
0.009 |
|
Parity |
0.4±1.0 |
0.6±1.0 |
0.296 |
|
Menopause |
|
|
0.493 |
|
Yes |
1 (4.2) |
39 (10.8) |
|
No |
23 (95.8) |
321 (89.2) |
|
Previous abdominopelvic surgery |
|
|
0.016 |
|
Yes |
1 (4.2) |
94 (26.1) |
|
No |
23 (95.8) |
266 (73.9) |
|
Body mass index (kg/m2) |
21.8±5.3 |
22.2±3.9 |
0.737 |
|
Pregnancy status |
|
|
>0.999 |
|
Yes |
0 (0) |
9 (2.5) |
|
No |
24 (100) |
351 (97.5) |
|
Dysmenorrhea (VAS) |
2.0±2.3 |
1.9±2.1 |
0.890 |
|
Chief complaint |
|
|
<0.001 |
|
Incidental |
1 (4.2) |
226 (62.8) |
<0.001 |
|
Abdominal pain |
23 (95.8) |
64 (17.8) |
<0.001 |
|
Body temperature (°C) |
36.7±0.6 |
36.5±0.3 |
0.139 |
|
CA125 (U/mL) |
23.8±12.8 |
31.9±51.7 |
0.064 |
|
CA19-9 (U/mL) |
146.4±319.1 |
59.2±135.9 |
0.311 |
|
WBC count (103/µL) |
10.9±4.5 |
6.4±2.1 |
<0.001 |
|
Neutrophil count (103/µL) |
7.9±4.2 |
4.0±1.8 |
<0.001 |
|
Lymphocyte count (103/µL) |
1.9±1.0 |
1.9±0.6 |
0.878 |
|
N/L ratio |
6.2±4.9 |
2.3±1.6 |
<0.001 |
|
Mean platelet volume (fL) |
8.5±1.3 |
8.5±1.3 |
0.729 |
|
CRP (mg/L) |
22.6±59.3 |
9.9±18.3 |
0.371 |
The surgical procedures and findings of patients were as shown in
Table 2. Patients in the torsion group had a larger cyst size than those in the control group (8.9±3.2 cm vs. 6.9±3.4 cm,
P=0.005). Significant differences of emergency case numbers and unilateral salpingo-oophorectomy (USO) cases of surgical procedure were also observed among the 2 groups. Abdominal pain was not severe or subsided in all cases (7 cases) with elective operation of torsion. In addition, there were 1 incidental case, 3 cases in which the symptoms were too mild or unclear to predict torsion, and 3 cases in suspection that the pain would be weakened due to detorsion delayed the operation. Although the 3 cases were suspected as detorsion, however, it was found that the pain lost due to necrosis progress when the patient actually went into surgery. Hysterectomy was performed only in patients with intrauterine lesions. All 10 patients who underwent hysterectomy with bilateral salpingo-oophorectomy (BSO) in the control group were accompanied by intrauterine lesions of myoma (eight patients), adenomyosis (one patient), and endometrial cancer stage Ia (one patient). Seventeen patients including one in the torsion group performed only BSO. BSO with or without hysterectomy was performed only to patients in postmenopausal status considering the risk of a recurrent MCT or other problems such as ovarian cancer risk and patient request. The diagnosis of underlying endometriosis and pelvic adhesion was not significantly different among 2 groups. The larger the cyst size was more occurred the torsion. In the torsion group, the ratio of emergent operations due to severe pain and USO due to necrosis was higher than the control group. Underlying endometriosis and pelvic adhesion were predicted to affect the occurrence rate of torsion, but there was no significant difference in this study.
Table 2
Comparison of surgical procedures and findings between torsion and control group

|
Factor in surgery |
Torsion (n=24) |
Control (n=360) |
P-value |
|
Cyst size (cm) |
8.9±3.2 |
6.9±3.4 |
0.005 |
|
Surgery type |
|
|
<0.001 |
|
Elective |
7 (29.2) |
352 (97.8) |
|
Emergent |
17 (70.8) |
8 (2.2) |
|
Surgical procedure |
|
|
0.059 |
|
Cystectomy |
14 (58.3) |
273 (75.8) |
0.056 |
|
USO |
9 (37.5) |
61 (16.9) |
0.024 |
|
BSO |
1 (4.2) |
16 (4.4) |
>0.999 |
|
BSO+hysterectomy |
0 (0) |
10 (2.8) |
>0.999 |
|
Underlying endometriosis |
|
|
>0.999 |
|
Yes |
3 (12.5) |
47 (13.1) |
|
No |
21 (87.5) |
313 (86.9) |
|
Pelvic adhesion |
|
|
0.927 |
|
Yes |
6 (25) |
87 (24.2) |
|
No |
18 (75) |
273 (75.8) |
In this study, among the variables that are significant in the analysis of clinical characteristics and surgical procedures, the logistic regression model was constructed with 6 variables (age, WBC count, neutrophil count, N/L ratio, cyst size, and previous abdominopelvic surgery history). The surgery type (elective/emergent) and chief complaint were excluded. The association between 6 variables and torsion is presented in
Table 3. In the univariate analysis, all 6 variables were significantly associated with torsion. In the multivariate analysis (Backward method), 5 variables (all except for previous abdominopelvic surgery) were significantly associated with torsion.
Table 3
Logistic regression model of age, white blood cell count, neutrophil count, neutrophil to lymphocyte ratio, cyst size, and previous abdominal/pelvic surgery with torsion on mature cystic teratomas

|
Factor in logistic regression model |
Univariate analysis |
Multivariate analysis (Backward method) |
|
OR (95% CI) |
P-value |
OR (95% CI) |
P-value |
|
Age (yr) |
0.939 (0.895–0.985) |
0.010 |
0.944 (0.895–0.996) |
0.035 |
|
WBC count (103/µL) |
1.046 (1.030–1.062) |
<0.001 |
1.128 (1.048–1.214) |
0.001 |
|
Neutrophil count (103/µL) |
1.047 (1.031–1.062) |
<0.001 |
0.882 (0.803–0.969) |
0.009 |
|
N/L ratio |
1.481 (1.300–1.687) |
<0.001 |
1.849 (1.330–2.569) |
<0.001 |
|
Cyst size (cm) |
1.134 (1.034–1.244) |
0.008 |
1.147 (1.014–1.298) |
0.029 |
|
Previous abdominal/pelvic surgery |
0.123 (0.016–0.924) |
0.042 |
|
|
The area under the curve (AUC), cut-off value, sensitivity, and specificity of continuous variables were shown in
Table 4. The cut-off values of variables have been made based on the highest value of Youden's index (Sensitivity + Specificity − 1), and the results of age, WBC count, neutrophil count, N/L ratio, and cyst size were 30, 7,740, 6,500, 3.35, and 6.4, respectively. The combined values were derived from a multivariate logistic regression of 5 variables, from which a prediction calculation formula was created. The prediction model equation is as follows.
Table 4
Diagnostic values of affecting variables for mature cystic teratoma with torsion

|
Variables |
AUC (95% CI) |
P-value |
Cut-off value |
Sensitivity (%) |
Specificity (%) |
|
Age (yr) |
0.657 (0.547–0.768) |
0.005 |
30 |
75.0 |
54.2 |
|
WBC count (103/µL) |
0.838 (0.748–0.927) |
<0.001 |
7,740 |
79.2 |
79.4 |
|
Neutrophil count (103/µL) |
0.806 (0.706–0.906) |
<0.001 |
6,500 |
62.5 |
91.9 |
|
N/L ratio |
0.725 (0.582–0.867) |
0.002 |
3.35 |
62.5 |
86.4 |
|
Cyst size (cm) |
0.705 (0.607–0.804) |
<0.001 |
6.4 |
79.2 |
53.6 |
|
Combinationa)
|
0.898 (0.833–0.962) |
<0.001 |
0.084 |
75.0 |
90.8 |
The AUC obtained by combining the 5 variables showed very high correlation (AUC, 0.898;
P<0.001) and the cut-off value for the combination was 0.084. In this study, 18 of the 24 patients were able to diagnose torsion using this cut-off value, and the sensitivity was 75%.
Fig. 1 showed the receiver operating characteristic curves obtained from each of the 5 variables and their combinations.
Fig. 1
Receiver operating characteristic curves affecting variables to diagnose the mature cystic teratoma with torsion.
WBC, white blood cell; N/L ratio, neutrophil to lymphocyte ratio.
a)This consists of the age, WBC count, neutrophil count, N/L ratio, and cyst size.

The clinical features of torsion group were as presented in
Table 5. The mean time from onset to surgery was 34.7 hours and the twist direction in the torsion group was clockwise in 8 patients, counter-clockwise in 8 patients, and unknown in 8 patients. The preoperative CT or MRI torsion diagnosis rate was 66.7%. The incidence of torsion by age was highest for those in their twenties (50%).
Table 5
Clinical features of the torsion group

|
Variables |
Value |
|
Time from onset (hr) |
34.7±32.2 (2–96) |
|
Twist direction |
|
|
Clockwise |
8 (33.3) |
|
Counter clockwise |
8 (33.3) |
|
Unknown |
8 (33.3) |
|
Torsion degree |
540±247.2 (180–1,080) |
|
Necrosis |
|
|
Yes |
9 (37.5) |
|
No |
15 (62.5) |
|
Preoperative CT or MRIa)
|
|
|
Torsion diagnosis |
14 (66.7) |
|
Others |
7 (33.3) |
|
Age subgroup (yr) |
|
|
<10 |
1 (4.2) |
|
10–19 |
5 (20.8) |
|
20–29 |
12 (50) |
|
30–39 |
4 (16.7) |
|
40–49 |
1 (4.2) |
|
≥50 |
1 (4.2) |
The correlation between ovarian necrosis and consumed time from abdominal pain onset to surgery in MCTs torsion group was presented in
Fig. 2. There was a statistical difference between the time from acute abdominal pain onset to surgery and existence of ovarian necrosis (
P<0.001). When the operation occurred at 62.7±26.7 hours later after abdominal pain onset in torsion group of MCTs, there were ovarian tissue necrosis. In this study, surgical procedures (cystectomy/USO/BSO) according to surgical type (elective/emergent) showed similar values in the torsion group.
Fig. 2
Comparison between ovarian tissue necrosis and consumed time from acute abdominal pain onset to surgery.
There was a significant difference in the total consumed time from acute abdominal pain onset to surgery (time from onset). When surgery occurred in early time, ovarian necrosis did not occur. Otherwise, ovarian necrosis was existed in late time (15.4±18.8 and 62.7±26.7 hours, respectively).
a)P<0.001.

Discussion
This study investigates the various factors that affect torsion in MCTs, the most common clinical ovarian tumor. In addition, various factors were analyzed to see whether it appeared as a result of torsion. Finally, the purpose of this study was to determine whether these factors can provide a more accurate and specific preoperative diagnosis of MCTs with torsion.
The incidence of the MCTs with torsion was known to be 3.2–16% [
2], and the torsion rate in this study was also 6.3%. Previous studies have demonstrated that the torsion rate increases significantly with age (teens) and for a larger cyst size. In this study, the torsion group was significantly younger than the control group, but the highest incidence was in patients in their 20s. The cyst size in the torsion group ranged 4–18 cm and the age of patients ranged 8–62 years, with 87.5% of total torsions occurring in the teenage years to the thirties.
Adnexal torsion is generally unilateral and more often on the right side than left (at a ratio of 3:2), this may be a result of the colon occupying the left or of hypermobility of the cecum and distal ileum [
9]. In our study, the direction of the torsion was right:left (58.3%:41.7%), which is consistent with previous studies, and neither recurrence nor bilaterality had occurred. In a previous study, a high N/L ratio (>3) was identified in 24 (88.9%) out of 27 in a torsion group, it was demonstrated that it seems a valuable marker in cases where it was difficult to diagnose adnexal torsion [
7]. In this study, only 14 out of 24 MCTs torsion patients showed a high N/L ratio, and it was determined to be less sensitive as a single indicator. It may be difficult to make a direct comparison between the results of this study conducted by MCTs torsion and previous studies that included all types of torsion.
We tried to investigate the relationship between pelvic adhesion and MCTs torsion due to previous abdominopelvic surgery or endometriosis, but the linkage was unclear. However, abdominopelvic surgical history was found to affect MCTs torsion as a single factor (
Table 3).
CA19-9 is known to be an important marker in the diagnosis of MCT [
12]. When combined with CA125, discrimination from ovarian cancer was possible if CA125 was at a normal level but CA19-9 was elevated [
13]. One case study reported an abnormal elevated CA19-9 (1,983 U/mL), which may be related to torsion in the ovary [
14]. In addition, previous studies about the indicator have referred to it as related to the extent of necrosis [
14,
15]. In our study, the patient with highest CA19-9 (1,209 U/mL) underwent a left salpingo-oophorectomy due to necrosis associated with torsion despite being 27 years of age.
Since the CA19-9 serum levels are correlated with larger tumor size [
12,
16], it may be necessary to study correlation after adjusting for the size factor. The cyst size was 16 cm in the above-mentioned case report [
14], and that of a patient who has the highest CA19-9 in this study was 13 cm. In the present study, 3 cases were performed in postoperative CA19-9 follow-up, one case in the torsion group (from 211.9 U/mL to 4.5 U/mL) and 2 cases in the control group (from 136.9 U/mL to 12.9 U/mL and from 871.3 U/mL to 3.7 U/mL), respectively. All 3 abnormal CA19-9 levels were returned to normal levels after surgery. The CA19-9 levels between the 2 groups were not statistically significant, but the mean values of both groups were higher than normal (
Table 1,
P=0.311). Using linear regression analysis, as with previous studies, there was a positive correlation between MCTs cyst size and CA19-9 (
P=0.004, n=167).
US is the first and most commonly used examination for the diagnosis of torsion in an emergency setting. However, US findings of adnexal torsion including a cystic, solid, or complex mass, thickening of the wall, and cystic hemorrhage are nonspecific [
17]. When using a color Doppler, if you see a twisted vascular pedicle (whirlpool sign), there is often a strong suggestion of this diagnosis, but the whirlpool sign is not clearly visible and it is often not observed. [
18]. Moreover, normal color Doppler findings may not exclude a diagnosis of torsion [
14]. Therefore, when torsion is suspected, a CT or MRI is often used to increase the accuracy of the diagnosis and to rule out the other acute abdomen [
17]. A twisted vascular pedicle or thickened tube is the most specific finding, and uterine deviation on the twisted side can be observed in almost all cases. The common CT and MR findings in adnexal torsion include tube thickening (84% of cases) [
17]. For abdominal pain patients coming to the emergency room, because of the procedural system in our hospital, the abdominopelvic computed tomography (APCT) will be taken first after the determination of pregnancy state. For the next step, there will be a consultation to the obstetrics and gynecology department if there is a gynecologic problem. When an ovarian cyst impression with or without torsion was found in APCT readings, US including color Doppler was always performed. It was performed throughout the torsion group, and 11 out of 24 patients (45.8%) were diagnosed torsion on US readings before surgery. A previous study reported that the accuracy of the preoperative diagnosis of ovarian torsion remains low (positive predictive value=46.1%) [
19], and the CT or MRI sensitivity in this study was 66.7%. In this study, 21 out of 24 torsion groups had CT or MRI scans, except for patients with obvious diagnosis of torsion (n=1) or underwent elective operation through outpatient because of their mild abdominal pain (n=2). In a published study on the diagnosis of adnexal torsion, scoring was performed by scoring for lumbar or abdominal pain, pain duration <8 hours, vomiting, absence of leucorrhea and metrorrhagia, and ovarian cyst size greater than 5 cm according to ultrasonography [
20]. However, there is a problem in this case that some indicators may be subjective and depend on the patient.
The strength of this study was the extraction of objective indexes such as age, WBC count, neutrophil count, N/L ratio, and cyst size, and to obtain their cut-off values. The predisposition of torsion in each cut-off value can be summarized in the case of age below 30 years, WBC count above 7,740/μL, neutrophil count above 6,500/μL, N/L ratio above 3.35, and cyst size above 6.4 cm. And these combination values are derived based on the equations presented in the result. Second, the AUC has a very high prediction rate of 0.898 when combined with these 5 factors, and the sensitivity is higher than that of CT or MRI (75% vs. 66.7%). Therefore, when a patient is admitted with acute abdominal pain, even without a CT scan, the diagnosis can be made with equal or higher probability. Also, the combined cut-off value (0.084) presented in this paper was applied to 7 cases with elective operation because the preoperative torsion diagnosis was not clear. As a result, the combination values of the 7 cases were 0.012, 0.027, 0.121, 0.176, 0.485, 0.789, and 0.804, respectively. The 5 cases out of 7 elective surgery cases satisfied the cut-off value of torsion. Therefore, the emergency surgery should not be delayed. Unclear or not severe abdominal symptoms are might be correlated with loss of symptoms due to ovarian tissue necrosis. Finally, if follow-up is performed with MCTs without symptoms, age and cyst size factors can be used to consult about the risk of torsion and to help determine the appropriate timing of surgery.
The limitation of this paper is that the number of torsion groups is too small compared to the control group. The ratio of torsion among all MCTs derived from this journal is 6.3%, and its absolute ratio is low, making it difficult to generalize the conclusion with this manuscript. However, despite the small sample size, statistically significant differences were obtained as presented. Since there are few systematic analyses focusing on the risk factors of MCTs torsion so far, if multi-center data are accumulated based on this study, it will provide a basis for generalization.
In conclusion, the combination of 5 parameters: age, WBC count, neutrophil count, N/L ratio, and cyst size, creates a very useful method for the diagnosis of MCTs torsion.











 PDF
PDF ePub
ePub Citation
Citation Print
Print



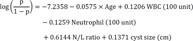
 XML Download
XML Download