Abstract
Purpose
In properly selected patients with breast cancer, nipple-sparing mastectomy (NSM) is generally considered safe by oncologic standards. We examined two groups of patients who underwent direct-to-implant (DTI) reconstruction after NSM, comparing complications encountered, revision rates, and aesthetic outcomes. The patients were stratified based on type of surgical incision and assigned to inframammary fold (IMF) and non-IMF groups.
Methods
We investigated 141 patients (145 breasts) subjected to NSM and immediate DTI reconstruction between 2013 and 2016. A total of 62 breasts (in 58 patients) were surgically removed via IMF incisions, with the other 83 breasts (in 83 patients) removed by non-IMF means.
Results
Complications associated with IMF (n=62) and non-IMF (n=83) incisions were as follows: skin necrosis (IMF, 9; non-IMF, 18); hematoma (IMF, 3; non-IMF, 4); seroma (IMF, 8; non-IMF, 4); mild capsular contracture (IMF, 4; non-IMF, 7); and tumor recurrence (IMF, 2; non-IMF, 8). Surgical revisions were counted as duplicates (IMF, 18; non-IMF, 38). Aesthetic outcomes following IMF incisions were rated as very good (44.2%), good (23.1%), fair (23.1%), or poor (9.6%).
Conclusion
IMF incision enables complete preservation of the nipple-areolar complex, yielding superior aesthetic results in immediate DTI breast reconstruction after NSM. The nature of incision used had no significant impact on postoperative complications or reoperation rates and had comparable oncologic safety to that of non-IMF incisions. IMF incisions produced the least visible scarring and did not affect breast shape. Most patients were satisfied with the aesthetic outcomes.
Improvements in breast reconstruction technique have significantly enhanced the quality of life for most patients with breast cancer. Therapeutic standards have evolved from radical intervention to skin-sparing mastectomy (SSM) or most recently nipple-sparing mastectomy (NSM), advancing reconstructive and aesthetic outcomes incrementally [12]. Although NSM has raised safety issues, this technique is generally considered an acceptable oncologic option in properly selected patients [345].
The demand for immediate breast reconstruction following conservative mastectomy has increased, with implant-based reconstruction most commonly pursued. According to Albornoz et al. [6], approximately 80% of all breast reconstructions performed in the United States involve prosthetic devices, and the vast majority are implanted immediately, placed at the time of mastectomy. When performing immediate implantbased breast reconstruction, it is still debatable whether a single-stage, direct-to-implant (DTI) strategy or a two-stage (tissue expander/implant) solution is best.
NSM with immediate prosthetic reconstruction may be performed using either a one-stage or two-stage method. Proponents of the one-stage DTI approach emphasize its low revision rate, fewer operations, reduced overall cost, and excellent patient outcomes [789]. Those supporting the two-stage method cite the opportunity to recontour and select an ideal device at second stage, reduced capsular contracture risk in the setting of postmastectomy radiation, lower unplanned revision rate, as well as excellent patient outcomes [1011].
When performing NSM, the choice of incision may be problematic, given that numerous variations have reportedly shown merit. Alternatives include radial, transverse periareolar, lateral, inferolateral, double concentric periareolar, vertical infra-areolar, and inframammary fold (IMF) incisions, with or without an axillary extension [12131415]. Endara et al. [16] have comprehensively addressed the process of choosing an appropriate incision for NSM. A radial or IMF incision is known to minimize scarring.
Although no incision is ideal, some certainly should be avoided. Endara et al. [16] found similar rates of nipple necrosis for radial and IMF incisions (8.8% and 9.1%, respectively), whereas this rate was much higher (17.8%) for periareolar/circumareolar incision lines. The highest incidence of nipple necrosis (81.8%) was recorded for a transareolar approach [16]. Using IMF incisions, the Beth Israel Deaconess group achieved an 82.0% nipple-areolar complex (NAC) survival rate in 17 breasts [17]. Blechman et al. [18] also reported a 94.0% NAC survival rate; in our series, the NAC survival rate was 95.1%.
Both oncologic and plastic surgeons would nevertheless agree on the convenience of a non-IMF incision in performing NSM. Moreover, a clearer intraoperative view is enabled. The drawback is a potential for visible scarring and nipple deformity or positional changes as likely consequences of scar contractures.
In this study, we examined two groups of patients who underwent DTI reconstruction after NSM, comparing complications encountered, revision rates, and aesthetic outcomes. The patients were stratified by the nature of the surgical incision, assigned to IMF and non-IMF groups.
We investigated 141 patients (145 breasts) subjected to NSM and immediate DTI reconstruction at Gangnam Severance Hospital, Seoul, Korea between 2013 and 2016. A total of 62 breasts (in 58 patients) were operated upon using IMF incisions, with the remaining 83 breasts (in 83 patients) involving non-IMF incisions. Detailed patient chart reviews were conducted retrospectively. The Yonsei University College of Medicine Institutional Review Board approved this study (number: 2017-08352014-001), and all participating patients granted informed consent.
Prior to 2016, mastectomy and breast reconstruction at our facility entailed non-IMF incisions. Since then, IMF incision has been the method of choice. NSM is also our surgical preference and is typically reserved for smaller tumors (<3 cm across) situated >3 cm from the NAC. If undertaking SSM or confronting likely postmastectomy radiotherapy for nodal positivity, a tissue expander (rather than DTI) would be inserted.
Preoperative markings were discussed and agreed upon by the oncologic and reconstructive teams. A lateral IMF incision was our preferred method. Incisions were made along the curvilinear skin crease, extending as far lateral as the 3 o'clock position for the left breast (9 o'clock for the right breast) and extending as far inferior as the 6 o'clock position. Sentinel lymph node biopsies or axillary dissections were also attempted through such incisions. If broader exposure was needed, counter-incisions of the axilla or along previous scars were made.
Acellular dermal matrices (DermACELL® [LifeNet Health, Virginia Beach, USA]; MegaDerm® [L&C BIO, Seoul, Korea]; or AlloDerm® [LifeCell Corp., Branchburg, USA]) were regularly used as inferolateral slings to expand inferior pockets and gain better IMF control. All permanent breast implants (Mentor Worldwide LLC, Santa Barbara, USA) were placed using dual plane technique (subpectoral and acellular dermal matrix positioning).
Skin incisions were routinely sutured to deep tissue (3–0 Vicryl; Ethicon, Bridgewater, USA), thereafter using 4–0 VLoc™ (Medtronic, Minneapolis, USA) and 5–0 Monosof (Medtronic) layer by layer. Two drains were placed at the time of mastectomy and removed after ~2–3 weeks. In the interim, patients were maintained on intravenous or oral antibiotics to protect against indigenous skin flora.
Until the day of operation and on postoperative days 1 and 2, methylprednisolone sodium succinate (125 mg Solu-Medrol®; Pfizer, New York, USA) was injected twice daily. This regimen served to promote skin flap perfusion and prevent postoperative edema.
As early as 2002, reports had surfaced on use of leukotriene inhibitors to treat capsular contractures [1920]. The benefits of montelukast (Singulair®; Merck, Kenilworth, USA) in softening breasts and mitigating capsular contractures have since been documented in a number of studies. Our facility allows once-daily montelukast administration, starting on postoperative day 7.
Complications were defined as necrosis of mastectomy skin and/or NAC, necessitating surgical intervention; seroma; capsular contracture; infection requiring intravenous antibiotics; implant loss; and tumor recurrence. Incidences of various reoperations in both groups were analyzed as well. To assess the impact of postoperative radiation on various complication and reoperation rates, subgroup analyses were performed, separating patients into postmastectomy radiation therapy (PMRT) and non-PMRT groups.
All patients were photographed pre- and postoperatively by the plastic surgeon. Aesthetic outcomes were gauged through subjective evaluation of certain factors, including NAC position, breast shape, and symmetry (Table 1), as suggested by Salibian et al. [21] Using a three-point scale, results were rated as very good, good, fair, or poor. Clinical examinations and reviews of photo documentation were carried out for this purpose by two plastic surgeons and one oncologic surgeon. Bilateral mastectomies in two patients were excluded. Subgroup analysis (PMRT vs. non-PMRT) was also conducted.
Complication and reoperation data were analyzed statistically. Descriptive statistics and measures of central tendency were used to describe absolute and mean results, respectively, with analysis of binary datasets via t-test and invoking chi-square analysis for proportional response comparisons. Fisher exact test was applied to small values. All computations relied on standard software (SPSS version 15.0.1 for Windows; SPSS Inc., Chicago, USA), setting significance at p<0.05.
We analyzed 145 NSM procedures (unilateral, 141; bilateral, 2). The IMF group accounted for 62 breasts in 58 patients (mean age, 46.4±5.9 years; mean body mass index [BMI], 22.7±2.3 kg/m2). Neoadjuvant and adjuvant chemotherapy was given to six and 28 patients, respectively; 47 patients received hormonal therapy; and 16 breasts were irradiated after reconstruction. The mean size of implanted prosthetics was 219.9±57.4 mL. In 62 DTI reconstructions, our oncologic breast surgeons performed sentinel lymph node biopsy in 46, axillary lymph node dissection in 13, and neither procedure in two.
The non-IMF group accounted for 83 breasts in 83 patients (mean age, 45.2±8.5 years; mean BMI, 22.6±3.2 kg/m2). Neoadjuvant and adjuvant chemotherapy was given to three and 25 patients, respectively; 65 patients received hormonal therapy; and 14 breasts were irradiated after reconstruction. The mean size of implanted prosthetics was 255.3±76.9 mL. In 83 DTI reconstructions, our oncologic breast surgeons performed sentinel lymph node biopsy in 74 and axillary lymph node dissection in nine (Table 2).
Procedural complications are summarized in Table 3. In the IMF group, skin necrosis (9/62, 14.5%), hematoma (3/62, 4.8%), seroma (8/62, 12.9%), capsular contracture (4/62, 6.5%) and infection (4/62, 6.5%) were encountered. Severe complications requiring breast implant removal were limited to three instances (4.8%). One of these patients acquired a postoperative infection and another developed severe capsular contracture after adjuvant radiotherapy. The third patient experienced extensive skin necrosis, which we debrided. The implant was then exchanged for a smaller one, anticipating a later reduction mammoplasty on the other side. There were two instances of tumor recurrence (3.2%), each adequately managed by oncologic surgeons.
In the non-IMF group, skin necrosis (18/83, 21.7%), hematoma (4/83, 4.8%), seroma (4/83, 4.8%), capsular contracture (7/83, 8.4%), infection (10/83, 12.0 %), implant removal (7/83, 8.4%), and tumor recurrence (8/83, 9.6%) were recorded. In the IMF (vs. non-IMF) group, capsular contracture (p=0.656), infection (p=0.259), implant loss (p=0.398), and tumor recurrence (p=0.132) rates tended to be lower, and the risk of skin necrosis (p=0.030) was significantly less. However, there were no significant between-group differences in terms of complications (Table 3, Figures 1 and 2).
We also investigated reoperations for secondary corrections, which were counted as duplicates. Results are shown in Table 4. In the IMF group, there were six skin flap revisions (9.7%), three hematoma evacuations (4.8%), two capsular contracture corrections (3.2%), three contralateral augmentation mammoplasties (4.8%), three implant extractions (4.8%), and one implant exchange (1.6%). In the non-IMF group, there were 13 skin flap revisions (15.7%), four hematoma evacuations (4.8%), one capsular contracture correction (1.2%), three scar revisions (3.6%), three contralateral augmentation mammoplasties (3.6%), four implant extractions (4.8%), and 10 implant exchanges (12.0%). The rate of implant exchange owing to size mismatch was significantly lower in the IMF (vs. non-IMF) group (p=0.019).
In subgroup analysis, a higher incidence of complications was evident in the PMRT (vs. non-PMRT) group across all fields, including skin necrosis (17/30, 56.7%), hematoma (4/30, 13.3%), seroma (9/30, 30.0%), capsular contracture (9/30, 30.0%), infection (10/30, 33.3%), implant loss (8/30, 26.7%) and tumor recurrence (7/30, 23.3%). Again, skin necrosis in the IMF (vs. non-IMF) group was significantly less (p=0.024), but no other significant differences emerged (Table 5).
PMRT had no apparent impact on reoperation outcomes, and there were no significant differences according to type of incision (Table 6).
To evaluate aesthetic outcomes, we compared pre- and postoperative photos. In the IMF group (n=58), two patients (four mastectomies) were excluded, and postoperative follow-up photos were lacking in four patients. In the non-IMF group (n=83), 28 patients also lacked follow-up photos. Ultimately, 107 patients (IMF, 52; non-IMF, 55) were eligible for review and comparison (Table 7).
Aesthetic outcomes in the IMF group were rated as follows: very good, 44.2% (23/52); good, 23.1% (12/52); fair, 23.1% (12/52); and poor, 9.6% (5/52). Of the 12 patients with fair results, six had received postoperative radiotherapy and two had experienced intractable seromas, leading to deep wound infections. The other four patients were free of comorbidities. One of the five patients with poor results showed breast asymmetry, having undergone implant removal for postoperative infection. Skin flap necrosis occurring in another three patients required surgical revision, and a capsular contracture necessitated capsulotomy and implant exchange.
Aesthetic outcomes in the non-IMF incision group were rated as follows: very good, 16.4% (9/55); good, 32.7% (18/55); fair, 29.1% (16/55); and poor, 21.8% (12/55). Of the 16 patients with fair results, most developed capsular contracture or implant malposition. In those rated as poor, some showed capsular contracture or mismatched implant size. There were also instances of implant malposition or skin necrosis needing reoperation. Outcomes rated as very good were significantly more numerous in the IMF (vs. non-IMF) group (p=0.002).
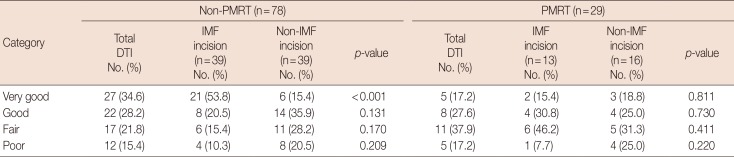
In subgroup analysis, the aesthetic superiority of the IMF incision was upheld in the non-PMRT group (n=78), with significantly more patients rated very good (p<0.001) and fewer rated as poor (p=0.209). However, in breast reconstructions performed after PMRT (n=29), there were no significant between-group differences (IMF vs. non-IMF) (Table 8).
Presently, NSM is a valid surgical option for treating breast cancer in selected patients. The combination of NSM and immediate breast reconstruction has been gaining traction as a preferred surgical strategy [222324], Furthermore, NSM techniques now include removal of glandular tissue from the nipple. This is a critical point of differentiation from prior subcutaneous mastectomy methods, where significant residual breast tissue was retained within and beneath the nipple. Benediktsson and Perbeck [25] have subsequently confirmed the oncologic safety of NSM, showing survival rates comparable to those of conventional mastectomy, given a 13-year median follow-up period.
One- and two-stage prosthetic reconstructions have been performed at a number of institutions using an array of incisions, including periareolar, inferior radial, inframammary, lateral, lateral radial, and inferolateral variations. The selection of a one- or two-stage approach in the setting of NSM is a complex process. Two-stage reconstruction yields better results, improving symmetry and implant position. Moreover, this strategy allows greater control over final breast volume and shape. However, cost is clearly a factor in that surgery is carried out at least twice. On the other hand, one-stage prosthetic reconstruction has few complications and confers relatively good aesthetic outcomes. At our facility, two-stage prosthetic reconstruction is elected if skin flap volume is scant or postoperative radiotherapy is anticipated.
As Endara et al. [16] have maintained, choosing the right incision for NSM facilitates both therapeutic and reconstructive efforts, preserving nipple-areola blood flow and producing an aesthetically favorable scar. Our oncologic surgeons generally relied upon non-IMF incisions until 2016. Thereafter, IMF incisions were introduced, supported by ideal incision criteria (referenced earlier). Since this transition, patient satisfaction has improved, and surgeons are pleased with the results, hoping to even further improve postoperative aesthetics.
Herein, we examined patients undergoing one-stage DTI breast reconstruction via non-IMF incisions and those reconstructed through IMF incisions, comparing complications. Skin necrosis in the IMF (n=9, 14.5%) vs. non-IMF (n=18, 21.7%) group was significantly less prevalent (p=0.030). There were nine instances of IMF-related skin necrosis in early 2016, suggesting a learning curve for this this new method. By preserving 3rd and 4th intercostal perforators to fortify the blood supply, our oncologic surgeon successfully enhanced remnant skin flap thickness and viability, and skin necrosis was seldom seen later that year. Nevertheless, skin necrosis did remain more problematic in non-IMF (vs. IMF) incisions, especially at the lower pole of the breast. One potential explanation is the gravitational pressure of implants against the lower reaches of skin flaps.
In the evolution of NSM procedures, the advantage of immediate breast reconstruction has brought to bear the importance of skin flap thickness in maintaining flap viability. Using 4 to 5 mm flaps, some sources [26] have confined necrosis to ~17%; through even thicker (10-mm) flaps, others have achieved rates <5% [2728]. Although thicker remnant flaps may indeed limit necrosis, no standard method is yet available for calibration, so consistency may be difficult to ensure. On average, our team generates 8-mm skin flaps. We are convinced that such relatively thick flaps may contribute to the lower rates of flap necrosis presently achieved. This also explains the lesser mean implant size in our IMF group, compared with the non-IMF group. The thin skin flaps formerly left behind were needed to cover larger sized implants. However, current (i.e., less aggressive) mastectomy trends leave relatively thick skin flaps, restricting the size of breast implants.
Rates of hematoma and seroma did not differ significantly by incision type, nor did the rate of capsular contractures, albeit somewhat premature. The follow-up period of ~1 year in the IMF group is possibly insufficient to reasonably assess contracture rates. Extended follow-up monitoring in future studies is truly needed. Rates of infection, implant loss, and tumor recurrence similarly showed no between-group differences.
In a subgroup analysis comparing postoperative complications in PMRT and non-PMRT groups, the various complications encountered did not differ significantly. As for reoperation outcomes, implant extraction was significantly more likely in the non-IMF (vs. IMF) group. This may be related to the higher rate of skin necrosis shown by the non-IMF group. Our data further indicated that PMRT had no impact on reoperation outcomes, regardless of incision type.
A multiplicity of criteria has been developed for assessing the aesthetic outcomes of NSM, including patient self-assessment, nipple sensitivity, postoperative scarring, nipple placement, breast contour, and overall symmetry [172930]. We used three criteria in our evaluations: NAC position, breast shape, and symmetry. IMF incisions concealed postoperative scarring and also resulted in fewer patient complaints.
Bilateral reconstruction (IMF group: four mastectomies) was excluded in our analysis of aesthetic outcome, owing to an inherent advantage in terms of symmetry. Accordingly, 35 patients (67.3%) in the IMF group were rated as having very good or good results, surpassing the corresponding rate (49.1%) in the non-IMF group. In the IMF (vs. non-IMF) group, significantly more patients were rated as very good (p=0.002).
Of note, we found no significant differences in aesthetic outcomes of IMF and non-IMF incisions after PMRT, regardless of aesthetic rating achieved. It may be that PMRT effects offset any aesthetic benefit conferred by IMF incisions. However, the aesthetic superiority of IMF (vs. non-IMF) incisions was upheld in non-PMRT subgroup analysis. Finally, because aesthetic outcomes were evaluated by our staff plastic surgeons, bias cannot be ruled out.
In conclusion, the use of IMF incisions for DTI breast reconstruction after NSM allows complete preservation of the NAC, with superior aesthetic results. We found that related postoperative complications, secondary corrective operations, and oncologic safety all compared favorably with non-IMF incisions. Moreover, IMF incisions produced the least visible scars and did not affect breast shape. Most patients expressed satisfaction with their aesthetic outcomes.
References
1. Didier F, Radice D, Gandini S, Bedolis R, Rotmensz N, Maldifassi A, et al. Does nipple preservation in mastectomy improve satisfaction with cosmetic results, psychological adjustment, body image and sexuality? Breast Cancer Res Treat. 2009; 118:623–633. PMID: 19003526.

2. Didier F, Arnaboldi P, Gandini S, Maldifassi A, Goldhirsch A, Radice D, et al. Why do women accept to undergo a nipple sparing mastectomy or to reconstruct the nipple areola complex when nipple sparing mastectomy is not possible? Breast Cancer Res Treat. 2012; 132:1177–1184. PMID: 22350788.

3. Jensen JA, Orringer JS, Giuliano AE. Nipple-sparing mastectomy in 99 patients with a mean follow-up of 5 years. Ann Surg Oncol. 2011; 18:1665–1670. PMID: 21174155.
4. Jensen JA. Breast cancer: is nipple sparing mastectomy safe? Ann Surg. 2009; 250:657–658. PMID: 19710601.

5. Crile G Jr, Esselstyn CB Jr, Hermann RE, Hoerr SO. Partial mastectomy for carcinoma of the breast. Surg Gynecol Obstet. 1973; 136:929–933. PMID: 4349981.
6. Albornoz CR, Bach PB, Mehrara BJ, Disa JJ, Pusic AL, McCarthy CM, et al. A paradigm shift in U.S. breast reconstruction: increasing implant rates. Plast Reconstr Surg. 2013; 131:15–23. PMID: 23271515.
7. Salzberg CA, Ashikari AY, Koch RM, Chabner-Thompson E. An 8-year experience of direct-to-implant immediate breast reconstruction using human acellular dermal matrix (AlloDerm). Plast Reconstr Surg. 2011; 127:514–524. PMID: 21285756.

8. Colwell AS, Damjanovic B, Zahedi B, Medford-Davis L, Hertl C, Austen WG Jr. Retrospective review of 331 consecutive immediate single-stage implant reconstructions with acellular dermal matrix: indications, complications, trends, and costs. Plast Reconstr Surg. 2011; 128:1170–1178. PMID: 22094736.
9. Colwell AS. Current strategies with 1-stage prosthetic breast reconstruction. Gland Surg. 2015; 4:111–115. PMID: 26005643.
10. Pusic AL, Cordeiro PG. Breast reconstruction with tissue expanders and implants: a practical guide to immediate and delayed reconstruction. Semin Plast Surg. 2004; 18:71–77. PMID: 20574485.

11. Spear SL, Seruya M, Rao SS, Rottman S, Stolle E, Cohen M, et al. Two-stage prosthetic breast reconstruction using AlloDerm including outcomes of different timings of radiotherapy. Plast Reconstr Surg. 2012; 130:1–9.

12. Regolo L, Ballardini B, Gallarotti E, Scoccia E, Zanini V. Nipple sparing mastectomy: an innovative skin incision for an alternative approach. Breast. 2008; 17:8–11. PMID: 17870535.

13. Boneti C, Yuen J, Santiago C, Diaz Z, Robertson Y, Korourian S, et al. Oncologic safety of nipple skin-sparing or total skin-sparing mastectomies with immediate reconstruction. J Am Coll Surg. 2011; 212:686–693. PMID: 21463813.

14. Colwell AS, Gadd M, Smith BL, Austen WG Jr. An inferolateral approach to nipple-sparing mastectomy: optimizing mastectomy and reconstruction. Ann Plast Surg. 2010; 65:140–143. PMID: 20606592.
15. Maxwell GP, Storm-Dickerson T, Whitworth P, Rubano C, Gabriel A. Advances in nipple-sparing mastectomy: oncological safety and incision selection. Aesthet Surg J. 2011; 31:310–319. PMID: 21385742.

16. Endara M, Chen D, Verma K, Nahabedian MY, Spear SL. Breast reconstruction following nipple-sparing mastectomy: a systematic review of the literature with pooled analysis. Plast Reconstr Surg. 2013; 132:1043–1054. PMID: 23924650.
17. Yueh JH, Houlihan MJ, Slavin SA, Lee BT, Pories SE, Morris DJ. Nipplesparing mastectomy: evaluation of patient satisfaction, aesthetic results, and sensation. Ann Plast Surg. 2009; 62:586–590. PMID: 19387167.
18. Blechman KM, Karp NS, Levovitz C, Guth AA, Axelrod DM, Shapiro RL, et al. The lateral inframammary fold incision for nipple-sparing mastectomy: outcomes from over 50 immediate implant-based breast reconstructions. Breast J. 2013; 19:31–40. PMID: 23252505.

19. Schlesinger SL, Ellenbogen R, Desvigne MN, Svehlak S, Heck R. Zafirlukast (Accolate): a new treatment for capsular contracture. Aesthet Surg J. 2002; 22:329–336. PMID: 19331987.

20. Schlesinger SL, Desvigne MN, Ellenbogen R, Svehlak S, Heck R. Results of using zafirlukast (Accolate) and montelukast (Singulair) for treatment of capsular contracture. Aesthet Surg J. 2003; 23:101–102.
21. Salibian AH, Harness JK, Mowlds DS. Inframammary approach to nipple-areola-sparing mastectomy. Plast Reconstr Surg. 2013; 132:700e–708e.

22. Rusby JE, Smith BL, Gui GP. Nipple-sparing mastectomy. Br J Surg. 2010; 97:305–316. PMID: 20101646.

23. de Alcantara Filho P, Capko D, Barry JM, Morrow M, Pusic A, Sacchini VS. Nipple-sparing mastectomy for breast cancer and risk-reducing surgery: the Memorial Sloan-Kettering Cancer Center experience. Ann Surg Oncol. 2011; 18:3117–3122. PMID: 21847697.

24. Spear SL, Willey SC, Feldman ED, Cocilovo C, Sidawy M, Al-Attar A, et al. Nipple-sparing mastectomy for prophylactic and therapeutic indications. Plast Reconstr Surg. 2011; 128:1005–1014. PMID: 21738086.

25. Benediktsson KP, Perbeck L. Survival in breast cancer after nipplesparing subcutaneous mastectomy and immediate reconstruction with implants: a prospective trial with 13 years median follow-up in 216 patients. Eur J Surg Oncol. 2008; 34:143–148. PMID: 17709228.

26. Verheyden CN. Nipple-sparing total mastectomy of large breasts: the role of tissue expansion. Plast Reconstr Surg. 1998; 101:1494–1500. PMID: 9583478.

27. Newman LA, Kuerer HM, Hunt KK, Kroll SS, Ames FC, Ross MI, et al. Presentation, treatment, and outcome of local recurrence after skin-sparing mastectomy and immediate breast reconstruction. Ann Surg Oncol. 1998; 5:620–626. PMID: 9831111.

28. Kroll SS, Ames F, Singletary SE, Schusterman MA. The oncologic risks of skin preservation at mastectomy when combined with immediate reconstruction of the breast. Surg Gynecol Obstet. 1991; 172:17–20.
29. Djohan R, Gage E, Gatherwright J, Pavri S, Firouz J, Bernard S, et al. Patient satisfaction following nipple-sparing mastectomy and immediate breast reconstruction: an 8-year outcome study. Plast Reconstr Surg. 2010; 125:818–829. PMID: 20195110.
30. Mosahebi A, Ramakrishnan V, Gittos M, Collier J. Aesthetic outcome of different techniques of reconstruction following nipple-areola-preserving envelope mastectomy with immediate reconstruction. Plast Reconstr Surg. 2007; 119:796–803. PMID: 17312480.

Figure 1
Representative reconstructive outcome: a 43-year-old woman with ductal carcinoma in situ of left breast. (A) Preoperative view. (B) Symmetric nipple-areolar complex position and breast shape in immediate postoperative state.
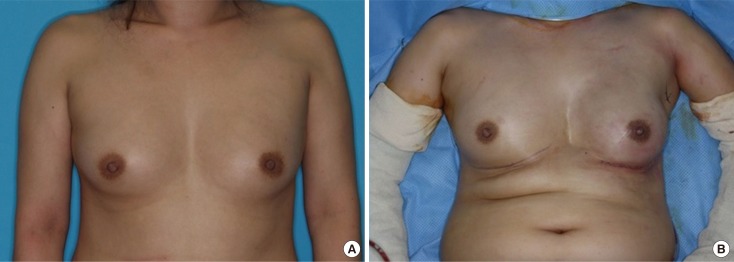
Figure 2
Representative reconstructive outcome: a 40-year-old woman with mucinous carcinoma of left breast. (A) Preoperative status, prior to nipple-sparing mastectomy (via inframammary incision) removing 464 g of tissue. (B) Outcome 6 months after immediate direct-to-implant reconstruction.
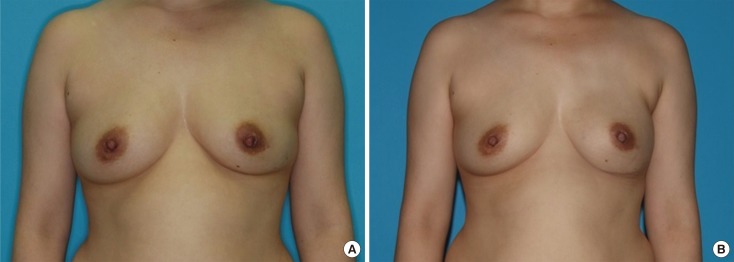
Table 1
Aesthetic outcome categories

Table 2
Comparative demographics for direct-to-implant breast reconstructions performed with IMF incisions and non-IMF incisions
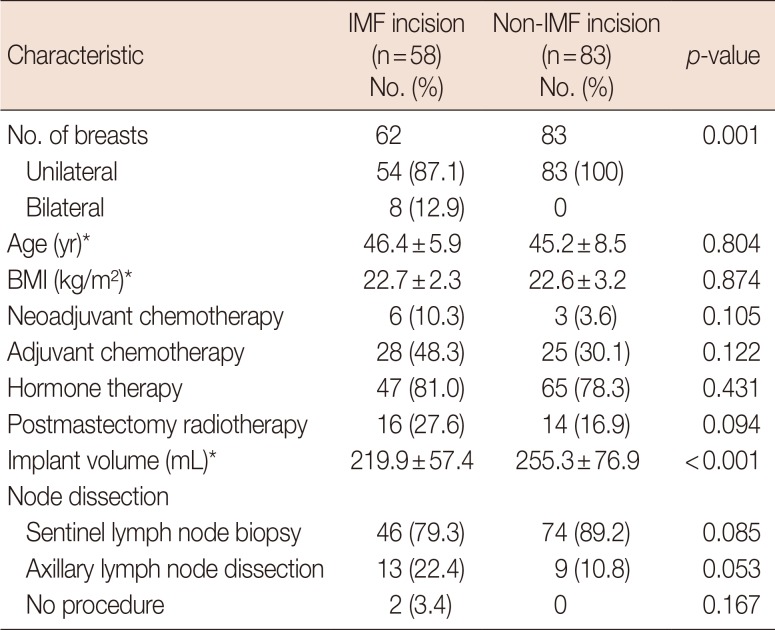
Table 3
Postoperative complications in patients undergoing DTI breast reconstruction following conservative mastectomy through IMF incision and non-IMF incision
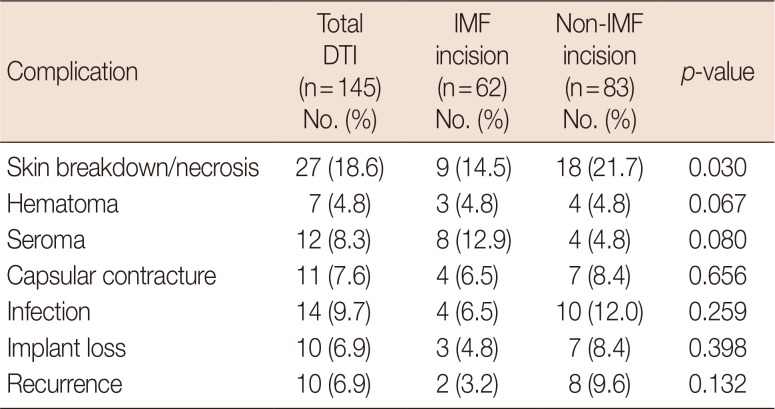
Table 4
Reoperation outcomes in patients undergoing DTI breast reconstruction following conservative mastectomy through IMF incision and non-IMF incision
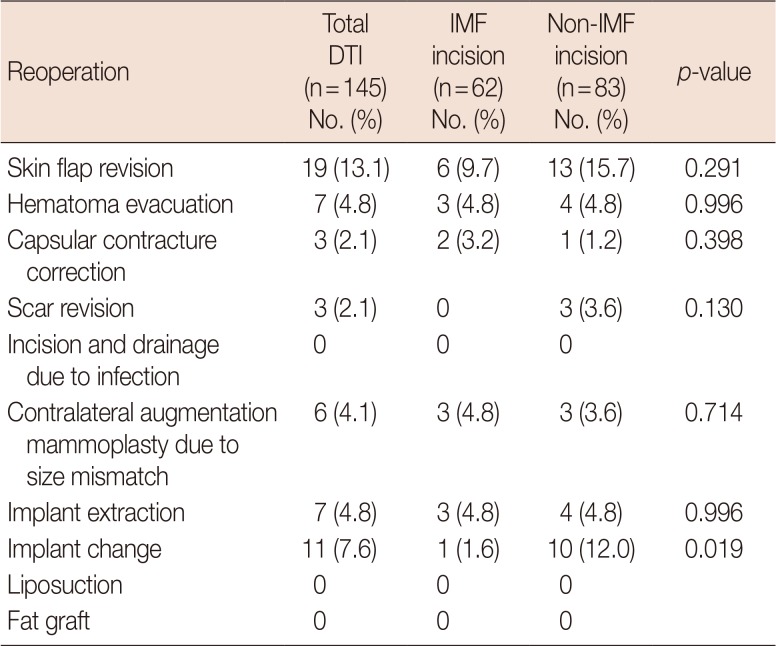
Table 5
Subgroup analysis of postoperative complications based on PMRT
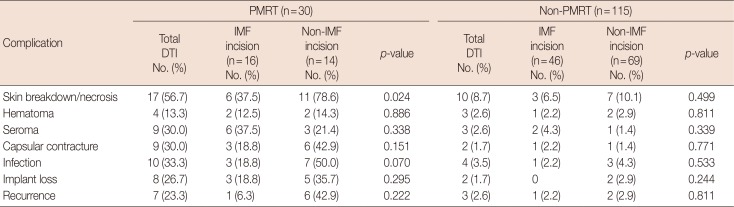
Table 6
Subgroup analysis of reoperation outcomes based on PMRT
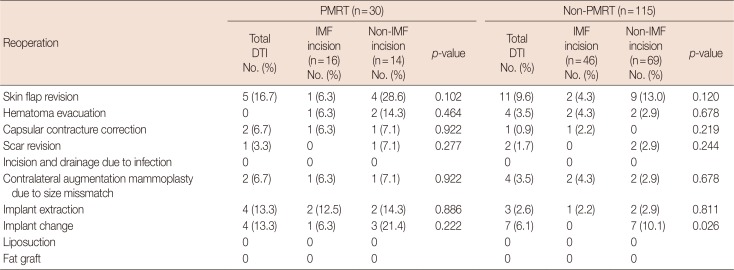
Table 7
Aesthetic outcomes of the reconstructions
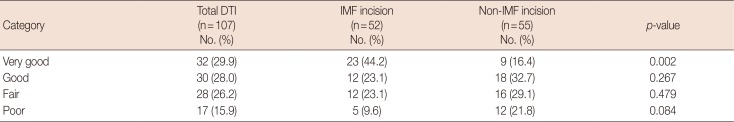
Table 8
Comparisons of the aesthetic outcomes of the breast reconstruction after PMRT status




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download