Abstract
Bowen's disease usually manifests as a slowly enlarging erythematous scaly patch or plaque. An uncommon variant of Bowen's disease showing a verrucous appearance has been reported and a distinct variant with a prominent clear cell change on histopathology, in addition to a verrucous surface change, was also reported. We describe novel form of Bowen's disease having a cerebriform appearance and showing histopathologically a significant clear cell change and propose that the clinical term “lobulated Bowen's disease” would be compatible for the description of this unique clinical variant. From a histopathological point of view, the precise definition and etiopathogenesis of the clear cell change in Bowen's disease should be elucidated.
Bowen's disease is a squamous cell carcinoma in situ of the skin which usually manifests as a slowly enlarging erythematous scaly patch or plaque. An uncommon variant of Bowen's disease showing a verrucous appearance, so-called “papillated Bowen's disease,” has been reported12 and a distinct variant with a prominent clear cell change on histopathology, in addition to a verrucous surface change, was reported as “warty and clear cell Bowen's disease (WCCBD)”34.
We describe another unique form of Bowen's disease having a cerebriform appearance and showing histopathologically a significant clear cell change and we propose the term “lobulated Bowen's disease” for this special variant.
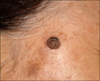
A 76-year-old woman presented with a 1-year history of an asymptomatic solitary nodule on the right temple. The lesion had enlarged slowly over time and no significant scaling or bleeding had occurred. Physical examination showed a 1×1 cm sized well-demarcated brownish walnut-shaped nodule on the right temple (Fig. 1).
Histopathological examination of punch biopsy specimens taken from central and marginal area revealed marked acanthosis with elongation and thickening of rete ridges and full-thickness atypia and disarray of keratinocytes with a significant clear cell change in the epidermis (Fig. 2A, B). The basement membrane remained intact and the upper dermis showed a moderate amount of inflammatory infiltrate. The clear cells occupied about 50% of the tumor population and showed positive staining for periodic acid-Schiff (PAS); however, negative staining for PAS with diastase (D-PAS), suggesting that the material contained in the clear cells was glycogen (Fig. 2C, D). A diagnosis of Bowen's disease with a clear cell change was made. The lesion was completely excised with clear resection margins and no recurrence was noted during a 6-month follow-up.
Several clinicopathological variants of Bowen's disease have been reported; however, Bowen's disease showing a cerebriform or walnut-like appearance has never been reported in the literature. The clinical feature of our case differs from that of papillated Bowen's disease in that the surface was not verrucous or hyperkeratotic, moreover, there was no papillomatosis on a histopathological examination12. Unusual cases of melanocytic nevi having a similar clinical appearance have been reported as “lobulated melanocytic nevi”56. We think the term “lobulated” also fit to describe the distinctive appearance of our case. To compare the clinicopathological differences between so-called “papillated” and “lobulated” Bowen's disease, we briefly describe another confirmed case of Bowen's disease showing a verrucous appearance developed on the right fifth finger of a 45-year-old man in our clinic (Fig. 3).
Although Bowen's disease sometimes shows a focal clear cell change on a histopathological examination, a prom inent clear cell change is not a common feature of Bowen's disease. Al-Arashi and Byers7 analyzed the overall features of Bowen's disease with a clear cell change and classified it by the percentage of clear cells and about 5% of Bowen's disease showed a clear cell change over 10%. The authors especially defined Bowen's disease with a clear cell change over 80% as “clear cell Bowen's disease (CCBD)” and, according to the authors, our case can be defined as Bowen's disease with a moderate clear cell change. The origin of clear cells in Bowen's disease remains controversial. Initial studies suggest that the clear cells are the result of degenerative changes in the cytoplasm89. Subsequent studies show that the clear cell change results from glycogen deposition, which can be identified by positive staining for PAS and negative staining for D-PAS and indicates a differentiation toward outer root sheath710. Some authors believe that the clear cell change is due to the cytopathic effects of human papillomavirus (HPV) infection on the basis of the presence of HPV DNA in the tumor cells111213. It might differ depending on how the clear cell is defined. In our case, the clear cell change was due to glycogen deposition and HPV DNA was not detected in the tumor cells by polymerase chain reaction test.
Among a lot of clear cell neoplasms of the skin, the differential diagnosis of Bowen's disease with a clear cell change should especially include pagetoid Bowen's disease, extramammary Paget's disease (EMPD), clear cell acanthoma, superficial spreading melanoma in situ, sebaceous carcinoma and trichilemmal carcinoma. Pagetoid Bowen's disease is often confused with CCBD; however, pagetoid cells in pagetoid Bowen's disease have pale-staining cytoplasm, not totally clear cytoplasm, and they show a pagetoid growth pattern, unlike clear cells in CCBD. Paget cells in EMPD also show a pagetoid growth pattern and stain positive for both PAS and D-PAS. Furthermore, in EMPD, flattened normal basal cells lying between Paget cells and the underlying dermis can be easily observed. In clear cell acanthoma, the area consisting of abundant glycogenated clear cells is sharply demarcated with the adjacent normal epidermis, unlike CCBD14. Superficial spreading melanoma in situ can be distinguished from CCBD by a pagetoid growth pattern of tumor cells and several melanocytic markers15. Sebaceous carcinoma is basically a dermal tumor and the tumor cells contain lipid globules, which can be identified by lipid stainings. Although trichilemmal carcinoma also consists of atypical cells with abundant glycogenated clear cytoplasm, the atypical cells form solid, lobular or trabecular growth patterns with foci of pilar-type keratinization and with peripheral palisading16.
In conclusion, we present a distinctive type of Bowen's disease showing a walnut-like appearance and a clear cell change and we propose that the clinical term “lobulated Bowen's disease” would be compatible for the description of this unique clinical variant. From a histopathological point of view, the precise definition and etiopathogenesis of the clear cell change in Bowen's disease should be elucidated.
Figures and Tables
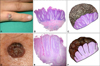
Fig. 2
(A, B) Marked acanthosis with elongation and thickening of rete ridges and full-thickness atypia and disarray of keratinocytes with a significant clear cell change in the epidermis (H&E; A: ×100, B: ×400). The clear cells shows positive staining for (C) periodic acid-Schiff (PAS) (×200) and negative staining for (D) PAS with diastase (D-PAS) (×200).
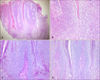
Fig. 3
(A~C) “Papillated” Bowen's disease. Papillomatous change on histopathology corresponds to clinically verrucous appearance (B: H&E, ×100). (D~F) “Lobulated” Bowen's disease. Marked acanthosis with elongation and thickening of rete ridges on histopathology is represented by clinically cerebriform or walnut-like appearance (E: H&E, ×100). (C, F) Original photographs were modified for schematic illustration.
References
1. Sun JD, Barr RJ. Papillated Bowen disease, a distinct variant. Am J Dermatopathol. 2006; 28:395–398.

2. Suárez-Vilela D, Izquierdo-García F, Domínguez-Iglesias F, Méndez-Álvarez JR. Combined papillated Bowen disease and clear cell atypical fibroxanthoma. Case Rep Dermatol. 2010; 2:69–75.

3. Hernández-Pérez E, Figueroa DE. Warty and clear cell Bowen's disease. Int J Dermatol. 2005; 44:586–587.

4. Sezer E, Yuksek J. Warty and clear-cell Bowen’s disease successfully treated with photodynamic treatment. Photodermatol Photoimmunol Photomed. 2010; 26:48–50.

5. Cho KH, Lee AY, Suh DH, Lee YS, Koh JK. Lobulated intradermal nevus. Report of three cases. J Am Acad Dermatol. 1991; 24:74–77.
6. Kim DH, Park HS, Paik SH, Jeon HC, Cho KH. Four cases of lobulated intradermal nevus: a sign of aging melanocytic nevus. Ann Dermatol. 2011; 23:115–118.

7. Al-Arashi MY, Byers HR. Cutaneous clear cell squamous cell carcinoma in situ: clinical, histological and immunohistochemical characterization. J Cutan Pathol. 2007; 34:226–233.

8. Kuo T. Clear cell carcinoma of the skin. A variant of the squamous cell carcinoma that simulates sebaceous carcinoma. Am J Surg Pathol. 1980; 4:573–583.

9. Requena L, Sánchez M, Requena I, Alegre V, Sánchez Yus E. Clear cell squamous cell carcinoma. A histologic, immunohistologic, and ultrastructural study. J Dermatol Surg Oncol. 1991; 17:656–660.

10. Corbalán-Vélez R, Ruiz-Macia JA, Brufau C, López-Lozano JM, Martínez-Barba E, Carapeto FJ. Clear cells in cutaneous squamous cell carcinoma. Actas Dermosifiliogr. 2009; 100:307–316.

11. Kwon TJ, Ro JY, Mackay B. Clear-cell carcinoma: an ultrastructural study of 57 tumors from various sites. Ultrastruct Pathol. 1996; 20:519–527.

12. Cohen PR, Schulze KE, Rady PL, Tyring SK, He Q, Martinelli PT, et al. Coincidental consort clear cell cutaneous carcinoma: facial squamous cell carcinoma in situ containing human papillomavirus and cancer cells with clear cytoplasm in an octogenarian couple. South Med J. 2007; 100:525–530.

13. Cohen PR, Schulze KE, Rady PL, Tyring SK, He Q, Martinelli PT, et al. Detection of human papillomavirus in cutaneous clear cell squamous cell carcinoma in situ: viral-associated oncogenesis may contribute to the development of this pathologic variant of skin cancer. J Cutan Pathol. 2008; 35:513–514.

14. Kirkham N, Aljefri K. Tumors and cysts of the epidermis. In : Elder DE, Walter Lever, editors. Lever's histopathology of the skin. 11th ed. Philadelphia: Wolters Kluwer;2015.
15. Elder DE, Elenitsas R, Murphy GF, Xu X. Benign pigmented lesions and malignant melanoma. In : Elder DE, Walter Lever, editors. Lever's histopathology of the skin. 11th ed. Philadelphia: Wolters Kluwer;2015.
16. Stewart CL, Novoa RA, Seykora JT. Tumors of the epidermal appendages. In : Elder DE, Walter Lever, editors. Lever's histopathology of the skin. 11th ed. Philadelphia: Wolters Kluwer;2015.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download