Abstract
Purpose
It is known that carriage rates of Staphylococcus aureus (S. aureus) are highest in newborns and that the asymptomatic carriage of methicillin-resistant Staphylococcus aureus (MRSA) is associated with invasive MRSA infection with the colonizing strain. This study was carried out to investigate the carriage rates of MRSA in neonates with neonatal jaundice.
Methods
We reviewed the medical records of 545 neonates admitted with neonatal jaundice to neonatal intensive care units between January 2006 and December 2010. Nasal and inguinal swab specimens had been taken from them and cultured for the isolation of S. aureus. Antimicrobial susceptibility tests had been done for such isolates to determine methicillin-resistance.
Results
Out of 545 neonates, 318 (58.3%) were colonized with S. aureus and 214 (39.3%) were colonized with MRSA. Results of the antibiogram analysis showed that 65.7% of MRSA isolates were likely to be community-associated (CA) MRSA.
Conclusion
Based on the MRSA carriage rate of 39.3%, a surveillance program for MRSA colonization is considered necessary in neonates transferred from other clinics or hospitals. Out of MRSA isolates, 65.7% were likely to be CA-MRSA. This suggests that CA-MRSA strains were already present in obstetric clinic environments where the neonates were born. It is thought that MRSA surveillance programs in these environments are also necessary. (Korean J Pediatr Infect Dis 2011;18:143–153)
References
1). Wertheim HF, Melles DC, Vos MC, Leeuwen WV, Bel-kum AV, Verbrugh HA, et al. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect Dis. 2005; 5:751–62.

2). Choi EH. Clinical manifestation and treatment of methi-cillin-resistant Staphylococcus aureus infections in children. Korean J Pediatr Infect Dis. 2009; 16:1–5.

3). EIFF CV, Becker K, Machka K, Stammer H, Peters G. Nasal carriage as a source of Staphylococcus aureus bacteremia. N Engl J Med. 2001; 344:11–16.
4). Huang YC, Chou YH, Su LH, Lien RI, Lin TY. Methi-cillin-resistant Staphylococcus aureus colonization and its association with infection among infants hospitalized in neonatal intensive care units. Pediatrics. 2006; 118:469–74.

5). Patel M. Community-associated meticillin-resistant Staphylococcus aureus infections. Drugs. 2009; 69:693–716.

6). Naimi TS, LeDell KH, Como-Sabetti K, Borchardt SM, Boxrud DJ, Etienne J, et al. Comparison of community-and health care-associated Staphylococcus aureus infection. JAMA. 2003; 290:2976–84.
7). Popovich KJ, Weinstein RA, Hota B. Are community-associated methicillin-resistant Staphylococcus aureus (MRSA) strains replacing traditional nosocomial MRSA strains? Clin Infect Dis. 2008; 46:787–94.

8). Popovich K, Hota B, Rice T, Aroutcheva A, Weinstein RA. Phenotypic prediction rule for community-associated methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 2007; 45:2293–5.
9). Millar BC, Loughrey A, Elborn JS, Moore JE. Proposed definitions of community-associated meticillin-resistant Staphylococcus aureus (CA-MRSA). J Hosp Infect. 2007; 67:109–13.

10). Gregory ML, Eichenwald EC, Puopolo KM. Seven-year experience with a surveillance program to reduce methi-cillin-resistant Staphylococcus aureus colonization in a neonatal intensive care unit. Pediatrics. 2009; 123:e790–6.

11). Kim DH, Kim SM, Park JY, Cho EY, Choi CH. Characterization of methicillin-resistant Staphylococcus aureus nasal colonization among neonatal unit staffs and infection control measures. Korean J Pediatr Infect Dis. 2009; 16:131–41.
12). Matouskova I, Janout V. Current knowledge of methi-cillin-resistant Staphylococcus aureus and community-associated methicillin-resistant Staphylococcus aureus. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2008; 152:191–202.

13). Lee YJ, Kim HJ, Byun SY, Park SE, Park HJ. Risk factors associated with complicated methicillin-resistant Staphylococcus aureus bacteremia in neonates. Korean J Pediatr. 2010; 53:173–7.

14). Cimolai N. Staphylococcus aureus outbreaks among newborns: new frontiers in an old dilemma. Am J Perinatol. 2004; 20:125–36.
15). Peacock SJ, Justice A, Griffiths D, Silva GD, Kantzanou MN, Crook D, et al. Determinants of acquisition and carriage of Staphylococcus aureus in infancy. J Clin Microbiol. 2003; 41:5718–25.
16). Ko KS, Lee JY, Baek JY, Peck KR, Rhee JY, Kwon KT, et al. Characterization of Staphylococcus aureus nasal carriage from children attending an outpatient clinic in Seoul, Korea. Microb Drug Resist. 2008; 14:37–44.
17). Kim YM, Oh CE, Kim SH, Lee J, Choi EH, Lee HJ. Nasal carriage of Staphylococcus aureus from healthy children attending day care center. Korean J Pediatr Infect Dis. 2010; 17:9–15.

18). Shakya B, Shrestha S, Mitra T. Nasal carriage rate of methicillin resistant Staphylococcus aureus among at National Medical College Teaching Hospital, Birgunj, Nepal. Nepal Med Coll J. 2010; 12:26–9.
19). Alfaro C, Mascher-Denen M, Fergie J, Purcell K. Prevalence of methicillin-resistant Staphylococcus aureus nasal carriage in patients admitted to Driscoll Children's Hospital. Pediatr Infect Dis J. 2006; 25:459–61.

20). Nakamura MM, Rohling KL, Shashaty M, Lu H, Tang YW, Edwards KM. Prevalence of methicillin-resistant Staphylococcus aureus nasal carriage in the community pediatric population. Pediatr Infect Dis J. 2002; 21:917–22.

21). Creech CB, Kernodle DS, Alsentzer A, Wilson C, Edwards KM. Increasing rates of nasal carriage of methicillin-resistant Staphylococcus aureus in healthy children. Pediatr Infect Dis J. 2005; 24:617–21.

22). Kim ES, Song JS, Lee HJ, Choe PG, Park KH, Cho JH, et al. A survey of community-associated methicillin-resistant Staphylococcus aureus in Korea. J Antimicrob Chemother. 2007; 60:1108–14.

23). Ko KS, Park S, Peck KR, Shin EJ, Oh WS, Lee NY, et al. Molecular characterization of methicillin-resistant Staphylococcus aureus spread by neonates transferred from primary obstetrics clinics to a tertiary care hospital in Korea. Infect Control Hosp Epidemiol. 2006; 27:593–7.
24). Kim SM. Methicillin-resistant Staphylococcus aureus. Korean J Pediatr Infect Dis. 1997; 4:201–9.

25). Huang SS, Platt R. Risk of methicillin-resistant Staphylococcus aureus infection after previous infection or colonization. Clin Infect Dis. 2003; 36:281–5.
26). Davis KA, Stewart JJ, Crouch HK, Florez CE, Hospenthal DR. Methicillin-resistant Staphylococcus aureus (MRSA) nares colonization at hospital admission and its effect on subsequent MRSA infection. Clin Infect Dis. 2004; 39:776–82.

27). Centers for Disease Control and Prevention. Community-associated methicillin-resistant Staphylococcus aureus infection among healthy newborns Chicago and Los― Angeles County, 2004. MMWR Morb Mortal Wkly Rep. 2006; 55:329–32.
28). Diederen BM, Kluytmans JA. The emergence of infections with community-associated methicillin resistant Staphylococcus aureus. J Infect. 2006; 52:157–68.

29). Vandenesch F, Naimi T, Enright MC, Lina G, Nimmo GR, Heffernan H, et al. Community-acquired methicil-lin-resistant Staphylococcus aureus carrying Panton-Valentine leukocidin genes: worldwide emergence. Emerg Infect Dis. 2003; 9:978–84.
30). Regev-Yochay G, Rubinstein E, Barzilai A, Carmeli Y, Kuint J, Etienne J, et al. Methicillin-resistant Staphylococcus aureus in neonatal intensive care unit. Emerg Infect Dis. 2005; 11:453–6.
31). Fortunov RM, Hulten KG, Hammerman WA, Mason EO Jr, Kaplan SL. Community-acquired Staphylococcus aureus infections in term and near-term previously healthy neonates. Pediatrics. 2006; 118:874–81.

32). Choe YJ, Lee SY, Sung JY, Yang MA, Lee JH, Oh CE, et al. A review of Staphylococcus aureus infections in children with an emphasis on community-associated methicillin-resistant S. aureus infections. Korean J Pediatr Infect Dis. 2009; 16:150–61.

33). Seybold U, Halvosa JS, White N, Voris V, Ray SM, Blumberg HM. Emergence of and risk factors for methi-cillin-resistant Staphylococcus aureus of community origin in intensive care nurseries. Pediatrics. 2008; 122:1039–46.

34). Al-Tawfig JA. Father-to-infant transmission of community-acquired methicillin-resistant Staphylococcus aureus in a neonatal intensive care unit. Infect Control Hosp Epidemiol. 2006; 27:636–7.
35). Chen KT, Huard RC, Della-Latta P, Saiman L. Prevalence of methicillin-sensitive and methicillin-resistant Staphylococcus aureus in pregnant women. Obstet Gynecol. 2006; 108:482–7.

36). Thompson DJ, Gezon HM, Rogers KD, Yee RB, Hatch TF. Excess risk of staphylococcal infection and disease in newborn males. Am J Epidemiol. 1966; 84:314–28.
37). Singh K, Gavin PJ, Vescio T, Thomson RB, Deddish RB, Fisher A, et al. Microbiologic surveillance using nasal cultures alone is sufficient for detection of methicillin-resistant Staphylococcus aureus isolates in neonates. J Clin Microbiol. 2003; 41:2755–7.
Fig. 1.
Annual Proportion of Methicillin-resistant Sta-phylococcus aureus. MRSA, methicillin-resistant Sta-phylococcus aureus; CA-MRSA, community-associated MRSA; HA-MRSA, healthcare-associated MRSA.
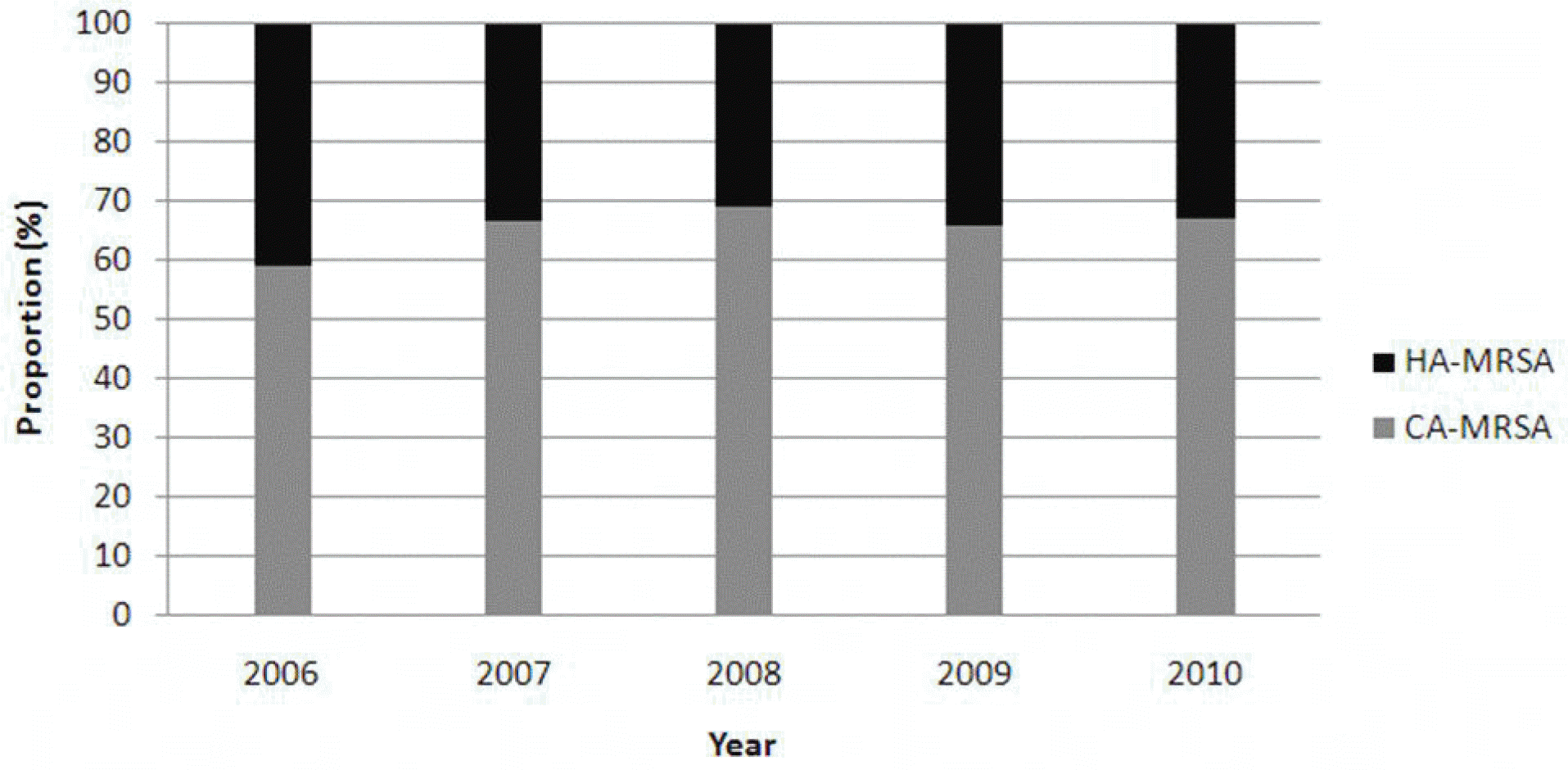
Table 1.
Carriage Rates ofStaphylococcus aureus by Culture Sites
Table 2.
Annual Carriage Rate of Staphylococcus aureus
Table 3.
Antibiotic Susceptibility Profiles of Community-Associated and Healthcare-Associated Methicillin-Resistant Staphylococcus aureus Isolates
Table 4.
Methicillin-resistant Staphylococcus aureus (MRSA) Antibiograms
Table 5.
Comparison of Carriage Rate of Staphylococcus aureus according to Delivery Type
| Number of neonates (%) | P value* | ||
|---|---|---|---|
| Vaginal delivery (n=394) | Cesarian section (n=151) | ||
| Total S. aureus | 236 (59.9) | 82 (54.3) | 0.236 |
| MRSA | 167 (42.4) | 47 (31.1) | 0.016 |
| CA-MRSA | 110 (27.9) | 32 (21.2) | 0.125 |
| HA-MRSA | 58 (14.7) | 16 (10.6) | 0.189 |
| MSSA | 75 (19.0) | 38 (25.2) | 0.114 |
Table 6.
Comparison of Carriage Rate of Staphylococcus aureus according to Gender
| Number of neonates (%) | P value* | ||
|---|---|---|---|
| Male (n=292) | Female (n=253) | ||
| Total S. aureus | 178 (61.0) | 140 (55.3) | 0.184 |
| MRSA | 120 (41.1) | 94 (37.2) | 0.347 |
| CA-MRSA | 76 (26.0) | 66 (26.1) | 0.523 |
| HA-MRSA | 45 (15.4) | 29 (11.5) | 0.482 |
| MSSA | 64 (21.9) | 49 (19.4) | 0.464 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download