Abstract
Background
Growth factors play important roles in wound healing. However, the evidence for the effects of growth factors on post-thyroidectomy scars is limited.
Objective
We performed a prospective study to assess the preventive and therapeutic effect of a multi-growth factor (MGF)-containing cream on post-thyroidectomy scars.
Methods
Twenty-one patients with thyroidectomy scars applied MGF cream twice a day. We assessed the changes in erythema, pigmentation, skin elasticity, and skin hydration status using the erythema index, melanin index, cutometer, and corneometer, respectively. In addition, Vancouver scar scale (VSS) and patient satisfaction were assessed at 10 days after surgery (baseline), 2 weeks, 6 weeks, and 12 weeks after baseline.
Results
The mean total VSS scores were significantly lower at 6 weeks (3.24±1.51 vs. 1.91±1.38) and 12 weeks (3.24±1.51 vs. 1.71±1.59) compared to the baseline. The degree of pigmentation was significantly lower at 12 weeks compared to the baseline, and the skin elasticity, and the skin hydration status were significantly higher at 12 weeks compared to the baseline. Over 85% of the patients were satisfied with the use of MGF cream without any adverse effect.
Thyroid cancer is the most common type of cancer diagnosed in South Korea. More than 40,000 people in the country were diagnosed with the disease in 20111. Thyroid cancer is also increasing dramatically in United States. Over 40,000 new cases of thyroid cancer were diagnosed in the United States in 2010 and the average annual percentage changes in incidence are 5.4% in men and 6.5% in women, suggesting that this cancer will be the fourth leading cancer diagnosed by 2030 in United States2. In the patients diagnosed with thyroid cancer, completeness of surgical resection is an important determinant of outcome. Thus the most of the patients are treated with thyroidectomy with central compartment neck dissection (CCND) or modified radical neck dissection, although small tumors that have not spread outside the thyroid gland may be treated by lobectomy3.
Surgical treatments inevitably remain scars, and thyroid surgery especially makes scar on neck. Scar on neck easily shows hypertrophy due to its inevitable many movements and stretching4. And it is easily noticed by the people due to its location and consequently causes many cosmetic problems. Thus it is increasingly important to prevent or minimize scar changes in patients who underwent thyroidectomy.
Wound healing is a complex process involving many cells, cytokines, and growth factors. Growth factors, such as epidermal growth factor (EGF), and fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), play important roles in wound healing4. However, the evidence for the effects of growth factors on post-thyroidectomy scars is limited. In this study, we performed a prospective study to assess the preventive and therapeutic effect of a multi-growth factor (MGF)-containing cream on post-thyroidectomy scars.
Patients with thyroid carcinoma who underwent open total thyroidectomy with CCND between January 2015 and July 2015 were enrolled in this study. A 5- to 7-cm cervical collar incision was made, and subplatysmal flap dissection was performed from the sternal notch inferiorly to the thyroid cartilage superiorly and laterally to both medial borders of the sternocleidomastoid muscle.
This study was approved by the Institutional Review Board of Severance Hospital, Yonsei University (IRB no. 4-2014-0892), and written and informed consent was obtained from each patient. Exclusion criteria included a history of keloid scarring and any other scar preventive cosmetic use in the 3 months prior to treatment.
The patients applied MGF cream twice a day. The MGF cream contained multiple growth factors, such as PDGF, VEGF, EGF, and FGF with humectant and emollient (PDGF and VEGF, EGF and FGF, 1.5% weight of mixture).
All patients were assessed the changes in post-thyroidectomy scar status after applying MGF cream at 10 days after surgery (baseline), 2 weeks, 6 weeks, and 12 weeks after baseline. During the follow-up period, patients were prohibited from using any oral or topical medication, excepting the creams used in this study.
Photographs were obtained using identical digital camera settings, lighting conditions, and patient positioning at the first visit before applying MGF cream, as well as at the second visit (2 weeks after baseline), third visit (6 weeks after baseline), and fourth visit (12 weeks after baseline).
Melanin indices and erythema indices were measured to assess the degree of pigmentation and erythema with a Mexameter® MX 18 connected to the Multiprobe Adapter System (Courage Khazaka Electronic GmbH, Köln, Germany) and at each follow-up visit. The degree of skin elasticity was measured with a Cutometer® dual MPA 580 (Courage Khazaka Electronic GmbH) and the degree of skin hydration was measured with a Corneometer® CM825 (Courage Khazaka Electronic GmbH) at every follow-up visit. Patients maintained comfortable supine positon during the measurements and the assessment was done at the laboratory which maintaining constant temperature and humidity. The probe was always held perpendicular and maintained firm contact to the scar tissue to be assessed. At every visit, physicians graded the post-thyroidectomy scar status using the Vancouver scar scale (VSS)5, which includes pigmentation (0, normal; 1, hypopigmented; 2, mixed pigmentation; 3, hyperpigmented), vascularity (0, normal; 1, pink; 2, red; 3, purple), pliability (0, normal; 1, supple; 2, yielding; 3, firm; 4, banding; 5, contracture), and height (0, flat; 1, ≤2 mm; 2, 2~5 mm; 3, ≥5 mm). The score for each parameter was assessed separately, and then all 4 parameters were combined.
At final visit, patients also scored their overall rates of satisfaction (0=unsatisfied, 1=slightly satisfied, 2=satisfied, 3=very satisfied), and adverse effects associated with the cream including erythema, tingling, scaling, and itching were assessed.
Data were compared using the one-way ANOVA test and the individual differences were assessed using a Tukey test if the ANOVA was significant. All data are presented in the form of mean±standard deviation. All statistical analyses were performed using IBM SPSS software ver. 23.0 (IBM Co., Armonk, NY, USA), with p<0.05 considered statistically significant in all cases.
A total of twenty-one East Asian patients who underwent open total thyroidectomy with CCND were enrolled in this study, and were composed of three males and eighteen females. The mean age of subjects was 42.19 years (range: 28~56 years) and the mean body mass index was 23.52±4.00.
Degree of erythema and pigmentation was evaluated at each visit using the erythema index and the melanin index. We found no significant differences in erythema index between the visits. However, the melanin index at 12 weeks was significantly decreased compared to the baseline (165.5±48.25 vs. 128.6±29.41; p<0.05).
The relative elasticity of the surgical scar was assessed at each visit. The cutometer at 6 weeks (0.56±0.16 vs. 0.72±0.14; p<0.05) and 12 weeks (0.56±0.16 vs. 0.76±0.10; p<0.05) were significantly increased compared to the baseline, and the cutometer at 12 weeks (0.62±0.18 vs. 0.76±0.10; p<0.05) was significantly increased compared to the cutometer at 2 weeks.
The relative skin hydration status was also assessed at each visit, and the corneometer at 2 weeks (31.60±9.80 vs. 57.62±12.29; p<0.05), 6 weeks (31.60±9.80 vs. 60.40±11.06; p<0.05) and 12 weeks (31.60±9.80 vs. 54.71±9.71; p<0.05) were significantly increased compared to the baseline (Table 1, Fig. 1).
The severity of the post-thyroidectomy scars were assessed using VSS and is summarized in Table 2 and Fig. 2. The mean total VSS scores were significantly lower at 6 weeks (3.24±1.51 vs. 1.91±1.38; p<0.05) and 12 weeks (3.24±1.51 vs. 1.71±1.59; p<0.05) compared to the baseline. Photographs of representative patients are shown in Fig. 3.
In the subset analysis of VSS, the pigmentation score of the scar became significantly lower at 2 weeks (0.76±0.94 vs. 0.19±0.68; p<0.05), 6 weeks (0.76±0.94 vs. 0.05±0.22; p<0.05), and 12 weeks (0.76±0.94 vs. 0; p<0.05) compared to the baseline, and the vascularity score of the scar also became significantly lower at 6 weeks (1.14±0.36 vs. 0.81±0.40; p<0.05) and 12 weeks (1.14±0.36 vs. 0.76±0.44; p<0.05) compared to the baseline. None of the patients using MGF cream showed hypertrophy during the 12 weeks of follow-up period.
Twenty-one patients completed the study, and overall satisfaction score was assessed at last visit, after applying 12 weeks of MGF cream. Mean satisfactions score was 2.19±0.68, which means majority of the patients were satisfied with the use of MGF cream and the percentage of patients who were very satisfied or satisfied was over 85% (18/21) (Fig. 4). There was no adverse event or discomfort associated with the use of the MGF-containing cream.
Hypertrophic scar and keloid are skin abnormalities that are characterized by excessive deposition of collagen in the dermis and subcutaneous tissues resulting from unbalanced tissue homeostasis during normal wound healing, which can sometimes cause cosmetic and functional impairment such as disfigurement, contractures, pruritus and pain6.
Although there have been many options for treating hypertrophic scars such as intralesional corticosteroids, intralesional 5-fluorouracil, cryotherapy, laser therapy and surgical excision, scar is irreversible and none of these treatments can turn back the scar to previous state without leaving a trace789. Thus it is important to prevent or minimize scar formation rather than treating developed scar late. Early and effective treatment of scarring can prevent severe scar change and the scarring that often follows. To minimize scarring, optimal environment for wound healing including proper circulation, nutrition and immune status is essential4. We try to protect wound from infection and support optimal moisture using a variety of dressing materials, silicone gel or sheet in case of necessity, however, these methods alone are not sufficient in effect410.
A number of growth factors, such as transforming growth factor (TGF)-β, TGF-α, FGF, VEGF, PDGF, and EGF, play important roles in wound healing through autocrine, paracrine, and endocrine processes11. These growth factors control many key cellular activities involved in the normal tissue repair process, including cell division, cell migration, angiogenesis, and synthesis of extracellular matrix components4.
PDGF and EGF promote the wound healing cascade by activating and attracting neutrophils, macrophages, endothelial cells, and fibroblasts1213. Some growth factors, such as FGF, VEGF, and PDGF, act as angiogenic factors that stimulate endothelial cells and mediate neovascularization and vessel repair at the wound site. They are also important modulators of cell growth and differentiation141516.
In particular, EGF is secreted from platelets, keratinocytes, and macrophages during the wound healing process and activates tyrosine kinase by binding to the EGF receptor present in keratinocytes and fibroblasts17, which has been proven in numerous studies to promote wound healing by shortening the healing time and increasing the tissue tensile strength by stimulating keratinocyte proliferation and acting on fibroblasts and smooth muscle cells1819.
Further, there have been several studies that have examined the benefits of exogenous growth factor application for wound healing and scar. It was demonstrated that recombinant human EGF significantly reduces cutaneous scars by suppressing inflammatory reactions, decreasing TGF-β1 expression, and suppressing excessive collagen formation in a murine full-thickness wound healing model20. In addition, recombinant human PDGF has been approved by the US Food and Drug Administration as a topical growth factor for use on chronic diabetic neuropathic foot ulcers. It promotes the chemotactic recruitment and proliferation of cells involved in wound repair and increases the probability that well-perfused, properly debrided diabetic foot ulcers will heal completely and in shorter times2122.
To date, silicone-based products such as silicone sheets and gels are widely used for scar prevention. A meta-analysis from Cochrane group showed that silicone gel sheeting reduced the incidence of hypertrophic scarring in high-risk individuals compared with no treatment (response rate: 0.46; 95% confidence interval, 0.21~0.98)23. The mechanism of action of silicone therapy has not been fully elucidated but occlusion and hydration of the stratum corneum with subsequent cytokine-mediated signaling from keratinocytes to dermal fibroblasts are now universally accepted as the major mechanisms24. In vitro study has shown that the production of basic FGFs (bFGFs) can be increased by silicone products25. And in vivo study using immunohistochemistry has also showed that the expression of bFGF was significantly higher in the scar tissue following the use of silicone gel sheeting compared with the untreated scar tissue26. The MGF cream contained many growth factors including FGF, so that it can be considered to share a common mechanism of action with the silicone gel sheeting in scar prevention.
In our study, over 85% of the patients were satisfied with the use of MGF cream without showing any adverse effect. And the VSS scores and the results of the objective assessments demonstrated the improvement of overall scar quality. VSS scores were improved with the use of MGF cream especially in pigmentation and vascularity subsets. In objective assessments, the degree of pigmentation, the relative elasticity, and the skin hydration status showed improvement with the use of MGF cream.
However, this study was performed only in the study group. Thus it is difficult to rule out the placebo effects, and this absence of the control group could be a limitation in this study. Nevertheless, the MGF cream might have additive or supportive effect for scar formation after thyroidectomy according to the results in this study. However, this should be clarified with further randomized controlled studies in the future, and studies comparing the efficacy between silicone gel sheeting and MGF cream or a combination of these products in scar management can be needed.
Figures and Tables
Fig. 1
(A~D) Data from the objective assessments of the patients at every visit. V1: visit 1 (baseline), V2: visit 2 (2 weeks after applying multi-growth factor [MGF] cream), V3: visit 3 (6 weeks after applying MGF cream), V4: visit 4 (12 weeks after applying MGF cream). *Signifies statistically significant difference (p<0.05).

Fig. 2
(A~E) Vancouver scar scale (VSS) score of the patients at every visit. V1: visit 1 (baseline), V2: visit 2 (2 weeks after applying multi-growth factor [MGF] cream), V3: visit 3 (6 weeks after applying MGF cream), V4: visit 4 (12 weeks after applying MGF cream). *Signifies statistically significant difference (p <0.05).

Fig. 3
Photographs of a 48-year-old male patient taken at each visit. (A) Baseline (Vancouver scar scale [VSS] vascularity 1, pigmentation 1, pliability 1, height 0), (B) 2 weeks after applying multi-growth factor (MGF) cream (VSS vascularity 0, pigmentation 0, pliability 1, height 0), (C) 6 weeks after applying MGF cream (VSS vascularity 0, pigmentation 0, pliability 0, height 0), (D) 12 weeks after applying MGF cream (VSS vascularity 0, pigmentation 0, pliability 0, height 0). The post-thyroidectomy scar showed improvement after applying MGF cream.
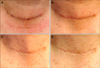
Table 1
Objective assessment data of the patients at every visit

Table 2
Vancouver scar scale score of the patients at every visit

References
1. Ahn HS, Kim HJ, Welch HG. Korea's thyroid-cancer “epidemic”--screening and overdiagnosis. N Engl J Med. 2014; 371:1765–1767.

2. Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014; 74:2913–2921.

3. American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer. Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009; 19:1167–1214.

4. Fonder MA, Lazarus GS, Cowan DA, Aronson-Cook B, Kohli AR, Mamelak AJ. Treating the chronic wound: a practical approach to the care of nonhealing wounds and wound care dressings. J Am Acad Dermatol. 2008; 58:185–206.

5. Sullivan T, Smith J, Kermode J, McIver E, Courtemanche DJ. Rating the burn scar. J Burn Care Rehabil. 1990; 11:256–260.

6. Tuan TL, Nichter LS. The molecular basis of keloid and hypertrophic scar formation. Mol Med Today. 1998; 4:19–24.

7. Lumenta DB, Siepmann E, Kamolz LP. Internet-based survey on current practice for evaluation, prevention, and treatment of scars, hypertrophic scars, and keloids. Wound Repair Regen. 2014; 22:483–491.

8. Berman B, Bieley HC. Adjunct therapies to surgical management of keloids. Dermatol Surg. 1996; 22:126–130.

9. Ledon JA, Savas J, Franca K, Chacon A, Nouri K. Intralesional treatment for keloids and hypertrophic scars: a review. Dermatol Surg. 2013; 39:1745–1757.

11. Diegelmann RF, Evans MC. Wound healing: an overview of acute, fibrotic and delayed healing. Front Biosci. 2004; 9:283–289.

12. Broughton G 2nd, Janis JE, Attinger CE. The basic science of wound healing. Plast Reconstr Surg. 2006; 117:7 Suppl. 12S–34S.

13. Velnar T, Bailey T, Smrkolj V. The wound healing process: an overview of the cellular and molecular mechanisms. J Int Med Res. 2009; 37:1528–1542.

14. Dejana E, Languino LR, Polentarutti N, Balconi G, Ryckewaert JJ, Larrieu MJ, et al. Interaction between fibrinogen and cultured endothelial cells. Induction of migration and specific binding. J Clin Invest. 1985; 75:11–18.

15. Grotendorst GR, Soma Y, Takehara K, Charette M. EGF and TGF-alpha are potent chemoattractants for endothelial cells and EGF-like peptides are present at sites of tissue regeneration. J Cell Physiol. 1989; 139:617–623.

16. Terranova VP, DiFlorio R, Lyall RM, Hic S, Friesel R, Maciag T. Human endothelial cells are chemotactic to endothelial cell growth factor and heparin. J Cell Biol. 1985; 101:2330–2334.

17. Nanney LB. Epidermal and dermal effects of epidermal growth factor during wound repair. J Invest Dermatol. 1990; 94:624–629.

18. Brown GL, Curtsinger L 3rd, Brightwell JR, Ackerman DM, Tobin GR, Polk HC Jr, et al. Enhancement of epidermal regeneration by biosynthetic epidermal growth factor. J Exp Med. 1986; 163:1319–1324.

19. Epstein JB, Gorsky M, Guglietta A, Le N, Sonis ST. The correlation between epidermal growth factor levels in saliva and the severity of oral mucositis during oropharyngeal radiation therapy. Cancer. 2000; 89:2258–2265.

20. Kim YS, Lew DH, Tark KC, Rah DK, Hong JP. Effect of recombinant human epidermal growth factor against cutaneous scar formation in murine full-thickness wound healing. J Korean Med Sci. 2010; 25:589–596.

21. Steed DL. Clinical evaluation of recombinant human platelet-derived growth factor for the treatment of lower extremity diabetic ulcers. Diabetic Ulcer Study Group. J Vasc Surg. 1995; 21:71–78. discussion 79-81.
22. Steed DL. Clinical evaluation of recombinant human platelet-derived growth factor for the treatment of lower extremity ulcers. Plast Reconstr Surg. 2006; 117:7 Suppl. 143S–149S. discussion 150S-151S.

23. O'Brien L, Pandit A. Silicon gel sheeting for preventing and treating hypertrophic and keloid scars. Cochrane Database Syst Rev. 2006; (1):CD003826.
24. Meaume S, Le Pillouer-Prost A, Richert B, Roseeuw D, Vadoud J. Management of scars: updated practical guidelines and use of silicones. Eur J Dermatol. 2014; 24:435–443.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download