Abstract
Background
Alopecia areata (AA) is an autoimmune skin disease difficult to manage and treat. The pathogenesis of AA features a T-cell-associated autoimmune process, and systemic immunosuppressive therapy is prescribed widely for AA.
Objective
To evaluate the efficacy and tolerance of systemic low-dose methotrexate (LD-MTX) therapy in treatment of recalcitrant AA multiplex.
Methods
In a retrospective, non-controlled study, we evaluated 29 patients with recalcitrant AA treated with LD-MTX and assessed the therapeutic response according to severity of disease, disease duration, cumulative dose of MTX, and drug safety.
Results
MTX was administered twice weekly, and the mean maximum weekly dose was 14.48 mg. The response was A5 (regrowth=100.0%) in 14 (48.3%) patients and A4 (regrowth of 75%~90%) in 12 (41.4%) patients. Three patients had poor response to LD-MTX treatment (A2: n=2 [6.9%], A1: n=1 [3.4%]). All three of the patients showing a poor response had disease durations exceeding 24 months. Relapse was observed in 31% of patients with more than 75% regrowth. Common side-effects were elevated liver enzyme levels and gastrointestinal discomfort.
Alopecia areata (AA) is an autoimmune disorder causing non-scarring hair loss. The estimated worldwide prevalence of AA is 0.1%~0.2%, and varies with ethnicity and geographic location1. The natural history of AA also varies, depending on the disease severity. Some hairless patches can be cured with minimal treatment, but severe AA, especially alopecia totalis and alopecia universalis (AU), is difficult to manage. Conventional treatments include systemic corticosteroid and local steroid injections234. Immunomodulators such as cyclosporine and azathioprine are also used567. However, some cases of AA remain intractable or experience frequent recurrence necessitating novel treatment options.
Methotrexate (MTX) was originally developed as a chemotherapeutic agent and acts as an antimetabolite and antifolate. MTX is used to treat many autoimmune disorders, including rheumatoid arthritis, dermatomyositis, lupus, and Crohn's disease8. In dermatology, MTX is often used to treat psoriasis and chronic eczema9. Weekly administration of 7.5~20 mg of methotrexate (low-dose MTX, LD-MTX) is considered generally safe and tolerable10. In this study, we evaluated the efficacy and tolerability of LD-MTX in treatment of AA multiplex who previously failed to respond to several conventional topical and/or systemic treatment.
This retrospective study reviewed the charts of 29 patients who were diagnosed with AA and treated with LD-MTX, from January 2008 to August 2014 in the Dermatology Clinic of Chungnam National University Hospital (IRB no. CNUH 2015-12-007). We defined recalcitrant AA as no response to more than 3 types of treatments with disease duration more than 6 months. The initial weekly dosage of MTX was 10 mg and increased by 2.5 mg every two weeks. During LD-MTX treatment, folic acid was supplied to all patients and complete blood cell counts and liver and kidney function tests were done at baseline, 1 month and every 3 months thereafter during the follow-up period. The duration of follow-up period after stopping LD-MTX treatment ranged from 12 to 20 months.
At baseline, we evaluated the severity of AA according to the Severity of Alopecia Tool (SALT)11. We also collected information on disease duration, previous treatments, MTX doses, treatment duration, relapse, disease-free duration and adverse events. The clinical response to hair regrowth was classified as follows: A0, no change or further loss; A1, 1%~24%; A2, 25%~49%; A3, 50%~74%; A4, 75%~99%; A5, 100%12.
Patient demographics are summarized in Table 1. The median age was 40.31 (range, 16~65) years; 16 patients were male and 13 were female. The mean disease duration was 29.03±32.61 months. The patients previously received triamcinolone local injection (100.0%), topical corticosteroid (100.0%), methylprednisolone pulse therapy (24.1%), narrow-band ultraviolet B treatment (44.8%) or systemic steroid therapy (31.0%).
The initial dose of MTX was 10 mg/week, and the mean maximum dose was 14.48 (range, 10~17.5) mg/week. The total cumulative dose was 350.86 (range, 165~990) mg and the mean treatment duration was 28.1 (range, 12~74) weeks.
Twenty-six of twenty-nine patients had clinical improvement after LD-MTX treatment. There were 14 patients (48.3%) in A5 and 12 patients (41.4%) in A4 (Fig. 1). However, 3 patients had poor response (A1: 3.4% [n=1], A2: 6.9% [n=2]). Regarding the severity of alopecia, most of S2 patients had clinical improvement (A5: 47.8% [n=11], A4: 43.5% [n=10]) and all 5 patients of S3 and S4 groups showed clinical improvement. However, 2 patients in S2 and 1 patient in S5 had no clinical improvement during LD-MTX treatment (Table 2).
Considering the disease duration, all patients who had less than 24 months disease duration showed significant clinical improvement (A5: n=9 [31.0%], A4: n=10 [34.5%]). Of the 10 patients with disease durations exceeding 24 months, 3 (30.0%) patients had no clinical improvement to LD-MTX treatment. We used the Mann-Whitney test to compare the response to therapy according to duration before treatment. The outcome was significantly different between the 6~24 months group and 24 months or more group (p=0.013). The outcome did not differ significantly between 12 months or less group and 13~24 months group (p=1.000).
The duration of follow-up after stopping the LD-MTX ranged from 12 to 20 months (15.26±2.80). Relapse occurred in 31.0% (n=9) of patients with >75% regrowth and the mean disease-free duration was 11.9 months (6~20 months).
The treatments were well tolerated by most patients but liver enzyme levels became elevated in three patients (10.3%), although no symptoms were evident. MTX therapy was stopped immediately and the patients were referred to a hepatologist. Three patients (10.3%) experienced gastrointestinal discomfort. It was transient and the symptoms disappeared after stopping the LD-MTX treatment. One patient developed lymphopenia and another showed reactivation of pulmonary tuberculosis (Table 3).
In this study, we showed that LD-MTX is a useful treatment option for intractable AA multiplex. This result is similar to those of previous studies. Chartaux and Joly13 reported complete regrowth in 57% of the patients treated with MTX alone. Droitcourt et al.14 also reported a 50% complete response using combination therapy with a high-dose pulse corticosteroid and MTX, although it was less effective in children. Oral dexamethasone afforded a similar response rate (37%) in AA patients, although relapse was frequent (74%) and adverse events were more common (30%)15. There are several reports using MTX in AA with systemic corticosteroids to increase the therapeutic efficacy131416. Systemic corticosteroids is effective in treating AA but side-effects during long-term therapy and relapse after dose reduction or withdrawal have restricted their use17. To avoid the side-effects caused by corticosteroids, we evaluated the efficacy and safety of LD-MTX alone in AA resistant to several conventional therapies.
Using the SALT score, most of the S1 to S4 patients had clinical improvement with LD-MTX treatment (26 of 28 patients [92.86%]). Despite the high efficacy of LD-MTX in this study, one S5 patient with AU had no clinical improvement. For AU patients, there is no satisfactory treatment option. Regarding the disease duration, all patients with durations less than 24 months had significant clinical improvement, while patients with disease duration exceeding 24 months showed no response to LD-MTX treatment. Similar to our results, other studies also have suggested that disease duration is an important prognostic factor in AA718.
Studies reporting recurrence rate after MTX treatment in AA is highly variable. A previous study found an 80% relapse rate and another recent study reported 33% relapse rate in patients with more than 50% regrowth1319 In our study, 9 patients (31.0%) with >75% hair regrowth experienced recurrent of disease. The difference between recurrence rates may be due to differences in the severity of disease, disease duration, cumulative MTX dose and concomitant medication.
No consensus has been reached on the optimal LD-MTX treatment duration, maintenance dosage or concomitant use of MTX with glucocorticosteroid. In our series, the mean treatment duration was 28.1 (range, 12~74) weeks. One study recommended that MTX treatment be continued for 18~24 months12. Another reported hair regrowth after 18 months of MTX treatment14. The further study will be needed to evaluate the efficacy of longer treatment duration and maintenance treatment after LD-MTX.
Though the treatments were well tolerated by most patients, some patients experienced side-effects. The most common side-effect was an abnormal liver function. The patients had no symptoms, such as fatigue, anorexia, or poor general condition. The abnormalities were detected only on regular check-ups performed during LD-MTX treatment. The changes were transient; the enzyme levels normalized after stopping MTX treatment. Previous study found that about 9% of patients on LD-MTX experienced transient elevations in transaminase levels16. When MTX is prescribed, it is essential to perform regular check-ups, including complete blood cell counts and liver function tests. Both baseline and follow-up chest x-rays are also important to check for re-activation of pulmonary tuberculosis. If problems are detected early, the adverse events may be reversible.
In conclusion, this study identified clinical benefit and safety for LD-MTX alone as a treatment option for patients with intractable and recurring AA. During treatment, regular check-ups, including complete blood cell counts and liver enzyme tests, are necessary. Further prospective studies are required to establish the optimal treatment duration, minimum effective maintenance dose to maintain the remission and timing of drug discontinuation.
Figures and Tables
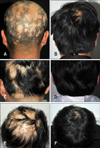 | Fig. 1Clinical response of low-dose methotrexate (LD-MTX) treatment. Baseline features of patients (A, C, E) and clinical response to LD-MTX treatment (B, D, F). A5 responses were seen in a (A, B) 44-year-old S4 male, (C, D) 47-year-old S2 male, and (E, F) 47-year-old S3 female. |
Table 1
Demographic characteristics of the patients (n=29)

ACKNOWLEDGMENT
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2015R1A2A2A01004664).
References
1. Safavi K. Prevalence of alopecia areata in the first national health and nutrition examination survey. Arch Dermatol. 1992; 128:702.

2. Chang KH, Rojhirunsakool S, Goldberg LJ. Treatment of severe alopecia areata with intralesional steroid injections. J Drugs Dermatol. 2009; 8:909–912.
3. Kar BR, Handa S, Dogra S, Kumar B. Placebo-controlled oral pulse prednisolone therapy in alopecia areata. J Am Acad Dermatol. 2005; 52:287–290.

4. Luggen P, Hunziker T. High-dose intravenous corticosteroid pulse therapy in alopecia areata: own experience compared with the literature. J Dtsch Dermatol Ges. 2008; 6:375–378.

5. Goddard CJ, August PJ, Whorwell PJ. Alopecia totalis in a patient with Crohn's disease and its treatment with azathioprine. Postgrad Med J. 1989; 65:188–189.

6. Farshi S, Mansouri P, Safar F, Khiabanloo SR. Could azathioprine be considered as a therapeutic alternative in the treatment of alopecia areata? A pilot study. Int J Dermatol. 2010; 49:1188–1193.

7. Açıkgöz G, Calıkan E, Tunca M, Yeniay Y, Akar A. The effect of oral cyclosporine in the treatment of severe alopecia areata. Cutan Ocul Toxicol. 2014; 33:247–252.

8. Cipriani P, Ruscitti P, Carubbi F, Liakouli V, Giacomelli R. Methotrexate: an old new drug in autoimmune disease. Expert Rev Clin Immunol. 2014; 10:1519–1530.

9. Haustein UF, Rytter M. Methotrexate in psoriasis: 26 years' experience with low-dose long-term treatment. J Eur Acad Dermatol Venereol. 2000; 14:382–388.

10. Spring P, Gschwind I, Gilliet M. Prurigo nodularis: retrospective study of 13 cases managed with methotrexate. Clin Exp Dermatol. 2014; 39:468–473.

11. Olsen EA, Hordinsky MK, Price VH, Roberts JL, Shapiro J, Canfield D, et al. Alopecia areata investigational assessment guidelines--part II. national alopecia areata foundation. J Am Acad Dermatol. 2004; 51:440–447.
12. Royer M, Bodemer C, Vabres P, Pajot C, Barbarot S, Paul C, et al. Efficacy and tolerability of methotrexate in severe childhood alopecia areata. Br J Dermatol. 2011; 165:407–410.

13. Chartaux E, Joly P. Long-term follow-up of the efficacy of methotrexate alone or in combination with low doses of oral corticosteroids in the treatment of alopecia areata totalis or universalis. Ann Dermatol Venereol. 2010; 137:507–513.
14. Droitcourt C, Milpied B, Ezzedine K, Hubiche T, Belin E, Akpadjan F, et al. Interest of high-dose pulse corticosteroid therapy combined with methotrexate for severe alopecia areata: a retrospective case series. Dermatology. 2012; 224:369–373.

15. Kurosawa M, Nakagawa S, Mizuashi M, Sasaki Y, Kawamura M, Saito M, et al. A comparison of the efficacy, relapse rate and side effects among three modalities of systemic corticosteroid therapy for alopecia areata. Dermatology. 2006; 212:361–365.

16. Joly P. The use of methotrexate alone or in combination with low doses of oral corticosteroids in the treatment of alopecia totalis or universalis. J Am Acad Dermatol. 2006; 55:632–636.

17. Yeo IK, Ko EJ, No YA, Lim ES, Park KY, Li K, et al. Comparison of high-dose corticosteroid pulse therapy and combination therapy using oral cyclosporine with low-dose corticosteroid in severe alopecia areata. Ann Dermatol. 2015; 27:676–681.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download