Dear Editor:
Minerals and essential elements are important components of nutrition1. These elements play crucial roles in the normal functioning of the immune system and antioxidant mechanisms which are related to the pathogenesis of atopic dermatitis (AD)2. Previous studies have hypothesized that AD is associated with a non-specific decrease concerning trace metals34. Furthermore, we previously reported that zinc (Zn) supplementation led to clinical improvement in AD patients with low hair Zn levels5. However, there is little data on other hair mineral levels in AD. Therefore, the aim of this study was to analyze the concentrations of trace elements in hair and to evaluate their relevance to disease severity in children with AD.
A total of 66 children (37 boys, 29 girls; mean age, 5.88 years; range, 1~14 years) with confirmed diagnoses of mild to moderate AD (eczema area and severity index [EASI] scores <26) were enrolled. A sex- and age-matched control group consisted of 25 children (15 boys, 10 girls; mean age, 6.12 years; range, 2~12 years) without dermatological disorders. The study protocol was approved by the ethics committee at Hanyang University Seoul Hospital (IRB no. 2011-R-34).
Participants were asked not to chemically process their hair for at least 8 weeks prior to mineral analysis. Mineral measurements were performed using a microwave temperature-controlled digestion technique and Perkin-Elmer Mass Spectrometer (SciexElan 6100; Perkin-Elmer Corporation, Foster City, CA, USA)6. The mineral concentrations are expressed as mg% (mg/100 g of hair). The reference ranges determined by US Trace Elements Inc. (TEI), which has been derived comprehensively from numerous data and widely used in the several studies, were used in this study.
The EASI score, trans-epidermal water loss (TEWL) using a Tewameter TM210® (Courage & Khazaka, Cologne, Germany), and visual analogue scales (VAS) for pruritus and sleep disturbance were assessed.
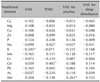
Fifteen nutritional elements and seven toxic elements were analyzed. The nutritional elements included calcium (Ca), magnesium (Mg), sodium (Na), potassium (K), copper (Cu), Zn, phosphate (P), iron (Fe), manganese (Mn), chromium (Cr), selenium (Se), boron (B), cobalt (Co), molybdenum (Mo), and sulfur (S). The toxic elements included uranium (U), arsenic (As), beryllium (Be), mercury (Hg), cadmium (Cd), lead (Pb), and aluminum (Al).
Among the nutritional elements, Ca, Mg, Cu, Zn, P levels were statistically lower and Na, K, Mn, Cr, and Mo levels were statistically higher in the AD patients compared to those in the control group. However, the mean levels of all these minerals were within the TEI reference ranges, except for K, which was higher than the reference range. Among the toxic minerals, Cd, Pb and As were significantly higher in AD patients than in control patients. In both groups, the mean levels of Cd and Pb were below the lower reference limit. Similarly, the mean level of As was within the reference range in the AD patients (Table 1). Next, we analyzed the significance of the relationship between numerical value of minerals that showed difference between two groups and the parameters of clinical severity and found a statistical significance for a few minerals. The increased levels of K in AD patients were negatively correlated with EASI scores. The Cd level was positively correlated with TEWL, but the mean level of Cd in AD patients was lower than the normal reference range. Although there was a statistical significance between Mo level and VAS for pruritus, they showed a weak correlation (Table 2). Based on the clinical parameters used, we did not find that these minerals were clinically relevant to AD.
There is growing interest in the role of minerals and micronutrients in AD. There have been several studies on mineral concentrations in AD patients, but the reported results have been contradictory. Zn is the most widely studied mineral that has clinical relevance in AD patients34. In this study, we found that the hair Zn level was significantly lower in AD patients as in our previous study5. Ca is involved in cellular physiology and is a major component of bone and teeth mineralization. Mg is an essential mineral that plays a critical role in the immune response. The lower levels of Ca and Mg in AD patients may reflect an avoidance of dairy products and nuts. Cu is an essential trace element in antioxidant systems and DNA synthesis7. Hon et al.4 suggested that the Cu/Zn ratio were increased in AD patients and positively correlated with the clinical severity348. However, in this study, there was no difference in the Cu/Zn ratio between the two groups or correlation with the clinical severity. This discrepancy between studies may result from differences in nutritional habits, age, or study methodology. K was the only element whose level exceeded the upper limit of the reference range in AD patients. However, since essential minerals interact with one another, there could be an imbalance in AD patients9.
There seems to be low risk of environmental hazards from heavy metal intoxication in Korean children with AD, because levels of toxic elements which were significantly higher in AD patients than in normal controls were below or within the reference ranges. Regardless, caution must be taken to prevent intoxication with such elements in AD patients. To our knowledge, no previous studies have evaluated the hair levels of various mineral levels in AD patients and established a relationship between the clinical severity and the mineral levels. Despite some differences between the two groups with regard to mineral levels, we did not find meaningful clinical relevance between the severity of AD and the micronutrient levels.
Although we did not evaluate the subjects' diets, the levels of trace elements may have differed due to nutritional imbalances in atopic patients. Nevertheless, most mineral levels were within the reference ranges. This finding indicates that atopic children did not have severe mineral deficiencies. Based on our findings, mildly reduced mineral levels are likely insufficient to produce clinical changes10. Large supplementation trials are needed to assess the clinical efficacy of replacing depleted minerals in AD patients. Indiscriminate commercial exploitation of mineral supplement product should be sublated before the confirmation of therapeutic effects of mineral supplementation.
Figures and Tables
Table 1
Comparison of hair mineral contents in atopic dermatitis (AD) patients and controls

Table 2
The correlation between clinical parameters and hair mineral levels in AD patients
References
1. Wang IJ, Guo YL, Weng HJ, Hsieh WS, Chuang YL, Lin SJ, et al. Environmental risk factors for early infantile atopic dermatitis. Pediatr Allergy Immunol. 2007; 18:441–447.

2. Puertollano MA, Puertollano E, de Cienfuegos GÁ, de Pablo MA. Dietary antioxidants: immunity and host defense. Curr Top Med Chem. 2011; 11:1752–1766.

3. David TJ, Wells FE, Sharpe TC, Gibbs AC, Devlin J. Serum levels of trace metals in children with atopic eczema. Br J Dermatol. 1990; 122:485–489.

4. Hon KL, Wang SS, Hung EC, Lam HS, Lui HH, Chow CM, et al. Serum levels of heavy metals in childhood eczema and skin diseases: friends or foes. Pediatr Allergy Immunol. 2010; 21:831–836.

5. Kim JE, Yoo SR, Jeong MG, Ko JY, Ro YS. Hair zinc levels and the efficacy of oral zinc supplementation in patients with atopic dermatitis. Acta Derm Venereol. 2014; 94:558–562.

6. Miekeley N, de Fortes Carvalho LM, Porto da Silveira CL, Lima MB. Elemental anomalies in hair as indicators of endocrinologic pathologies and deficiencies in calcium and bone metabolism. J Trace Elem Med Biol. 2001; 15:46–55.

8. Tasaki M, Hanada K, Hashimoto I. Analyses of serum copper and zinc levels and copper/zinc ratios in skin diseases. J Dermatol. 1993; 20:21–24.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download