AD shows a wide heterogeneity of etiological factors
89, which underscore its chronic and often indocile course. As the multi-faceted nature of its pathophysiology become further unraveled, a gradual, shift in disease paradigm has been set in motion; the incumbent paradigm is the model of perturbed equilibrium in the adaptive arm of the immune system
1011, in which a largely Th
2-process, coupled with dysregulated T
reg subpopulation
12 triggers a cascade of closely-intertwined immunological interactions. The breached-barrier model has given its way to one of cellular/adaptive immunity and the constituent molecular mediators (with a distinct emphasis on cytokines and chemokines), which are presently considered “bolts and screws” of the machinery. These mediators may also have a direct bearing on characteristic symptoms; studies have revealed that in AD-associated intractable pruritus-where the receptors involved are often not entirely histaminic
13 -proinflammatory cytokines are prime target suspects that instigate it
1415. Naturally brought along with this paradigmatic shift was revamping in management scheme; systemic immunomodulators and biologics have taken a center-stage as an important arsenal against the immunological leg of AD patho-mechanism
16. The authors, having experienced an apparent, skin barrier-reparative effect with an HS water after 4 weeks of bathing in a tub
17, were prompted with the question of whether HS water brings upon beneficial effects on cellular and molecular level with certain immune-mediating action. Our results point to the evidence that
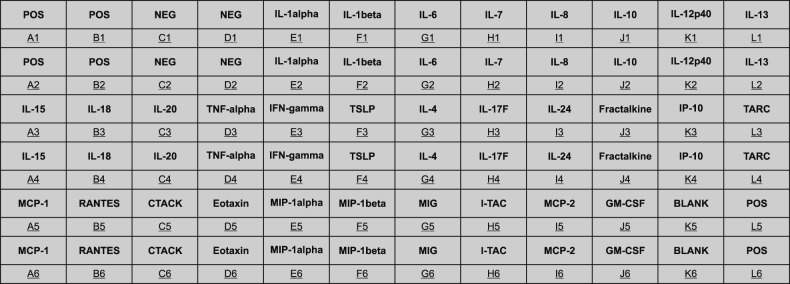
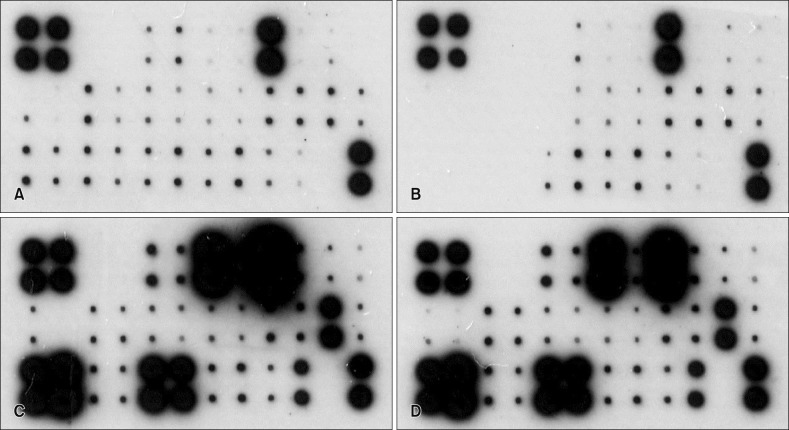
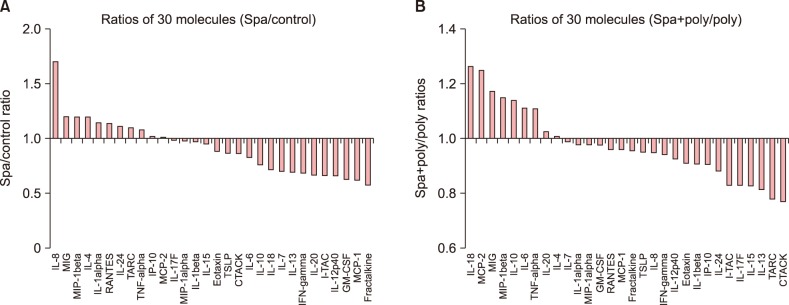
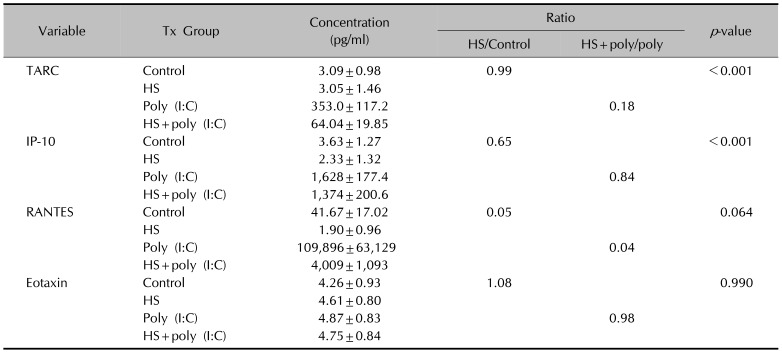
Magumsan HS water was shown to have on overall suppressed expression of the key KC-derived immune mediators, which. The expression level of IL-1α, IL-6, IL-8, TNF-α, and GM-CSF are attenuated in the presence of an NaCl/CaCl HS water
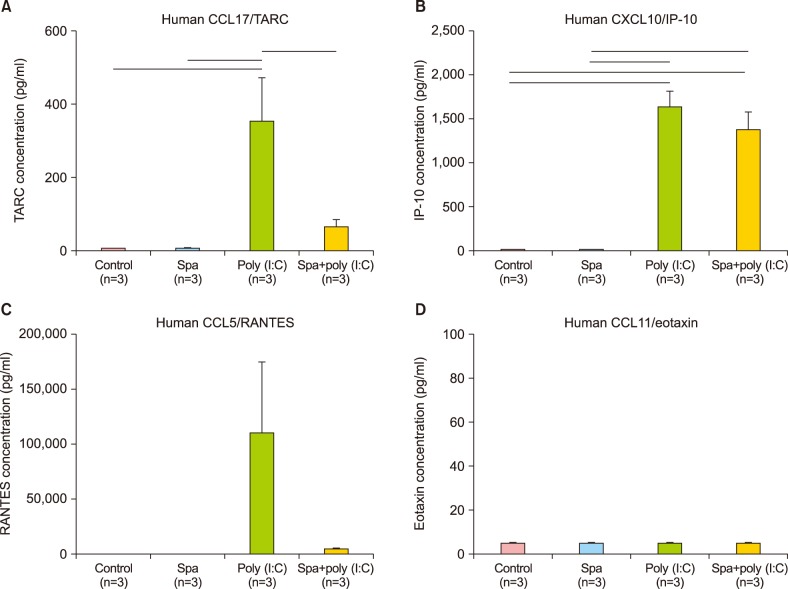
in vitro. In the poly (I:C)-stimulated, HS-treated group, reduction in relative level of expression was seen with IL-8, IL-15, IL-17F, IL-24, fractalkine, IP-10, TARC, MCP-1, RANTES, CTACK, and I-TAC. IL-8 is a KC-derived cytokine that acts as a chemoattractant for neutrophils during the Th
2-dominated acute inflammation phase of AD
18. IL-15 is renowned for its role in psoriasis, where the proinflammatory mediator is released from KC to influence survival and turnover of CD8
+ cells, which in turn feeds back to the KC to enhance inflammation
19. IL-24 is secreted from KC and facilitates expression of secretion of pro-inflammatory mediators IL-8, PGE
2, and MMP-1, in a positive feedback regulation of epidermal inflammation in response to external and internal sources
20. On the other hand, there was an up-regulation of IL-10, an anti-inflammatory cytokine, and six other proinflammatory cytokines and chemokines (IL-6, IL-20, TNF-α, MIP-1β, MCP-2, and GM-CSF). Taken together, the expression profile results reaffirm our previous findings that the HS water exerts significant curbing effects on proinflammatory mediators from KC and simultaneously contribute to holding of inflammation in check through facilitating secretion of T
reg-related mediators. Even in the face of this shift of weight from barrier to immunity, one factor that remains a crucial common denominator is the role played by KCs. KCs make up the brick and mortar of the outermost layer of barricade against constant physical, chemical, and bacterial barrage from the external environment. On another level, these cells are an important command post for the immunological interplay between major elements of innate and cellular immunity
2122. A vast reservoir for innate immune mediator is one of the first responders at the scene to neutralize the threats of possible outside intrusion or noxious stimulus from within. In the case of the latter, these chemical alarm signals often take the shape of pattern recognition receptors, typified by TLR and nucleotide-binding oligomerization domain-like receptors. Our preliminary investigations led to the conviction that TLR 3 agonist is the most capable of inducing secretion of various mediator molecules. Hence, it was used categorically for recreating the
in vivo environment
circa KC during the Th
2-driven inflammation.
Akiyama et al.
23 suggested that trace metal ions (e.g., Mn
++ and I
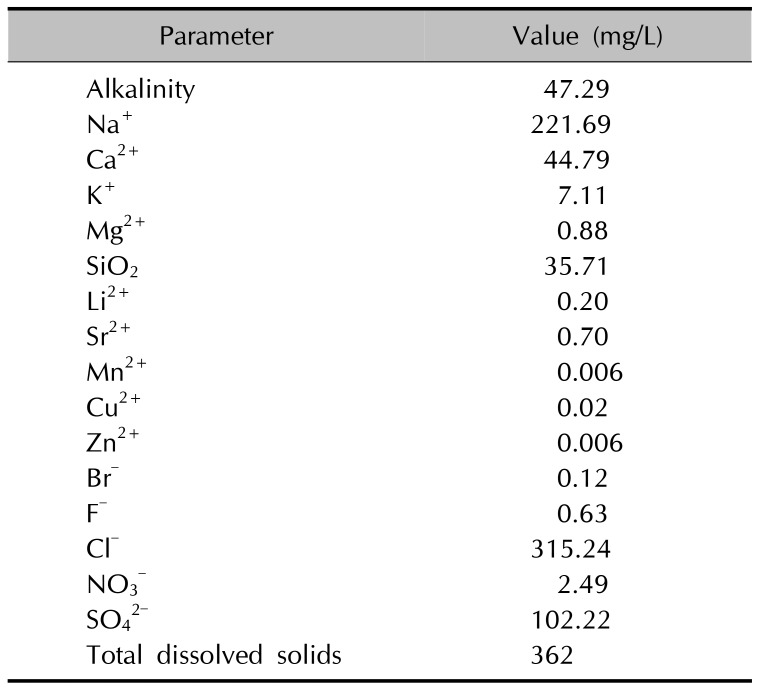
−) are crucial for the bactericidal property of HS water and its ability for keeping hydrogen concentration low. On par with the notion, the most predominant cation and anion in the present investigation were Na
++ and Cl
−, respectively (i.e., an NaCl type HS water), which were responsible for the improvement of cutaneousd barrier homeostasis in
Magumsan HS. The concentration of CaCl
2 in
Magumsan HS was 44 mg/L, and authors had attempted to produce a solution of CaCl
2 of the same concentration (for the purposes of comparison); however, the actual osmolarity was significantly deviant from the expected value, which forced us to abandon its use. Upon a consultation with a membrane physiologist, the authors have reckoned that the experimental value is of far more relevance, which led us to use only HS water for osmolarity, which would pose a serious flaw in accurate assessment of its effects on direct cell contact. Early on in our experiment, we added 4 M NaCl determination. Osmolarity of the water was determined to be 74 mOsm/kg initially along with 1 M sucrose as to bring the DMEM media equimolar to normal cell osmolarity. Afterwards, the resulting mixture of the
Magumsan water and DMEM powder was determined to be 350 mOsm/kg, which was deemed suitable.
The authors believe that our endeavor in the present investigation has left us with an alternative methodology for assessing potential immune-modulating actions of Korean HS water with varying spectrum of constituent ionic substances; this compendium represents a possible standard protocol for further sophisticated studies involving HS water. Our microarray results provided a window through which the immunological impact of an NaCl type HS water on skin overall can be grasped, and we are convinced that our work would serve as a primer for other prospective investigative trials that would hopefully fill in the missing puzzles between the apparent beneficial effects of HS water and its elaborate working mechanism. Future studies on detailed molecular machinery of immune-inflammatory regulation of HS water and varying degrees of effects based on differing mineral composition and concentrations would enable us to venture further into the territory that are left largely unchartered.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download