Amputation of affected phalanx was performed for the surgical management of SUM
in situ in the past, but several recent literatures advocated the phalangeal bone conserving surgery in early stage
91011. Therefore, early detection and diagnosis of SUM is important because it can improve quality of life as well as 5-year survival rate. Our study provides a clinical screening tool for SUM
in situ. In 1989, Saida and Ohshima
3 described 5 clinical features of SUM
in situ: noticed after middle age; pigmented band broader than 6 mm; brownish band with shades of tan to black; Hutchinson's sign; and slight degree of nail deformity. Levit et al.
6 reviewed this and other literatures to propose the “ABCDEF” mnemonic for SUM: “A” stands for age, ranged from 20 to 90 years in Asian or African-American race; “B” stands for pigmented black-brown band with breadth wider than 3 mm; “C” stands for rapid change in nail band or lack of change in nail dystrophy despite appropriate treatment; “D” stands for digit, namely, most commonly in thumb of dominant hand; “E” stands for extension of pigmentation on periungual skin; “F” stands for family or personal history of dysplastic nevus syndrome or melanoma. Although this mnemonic seems to be useful, items need to be added or corrected for SUM
in situ. First, all patients were adults (i.e., older than 18 years). Previous ABCDEF rule specified the range of age, but we simplified “A” as “adult age”. All of 18 cases of SUM
in situ in our previous study were adults as well
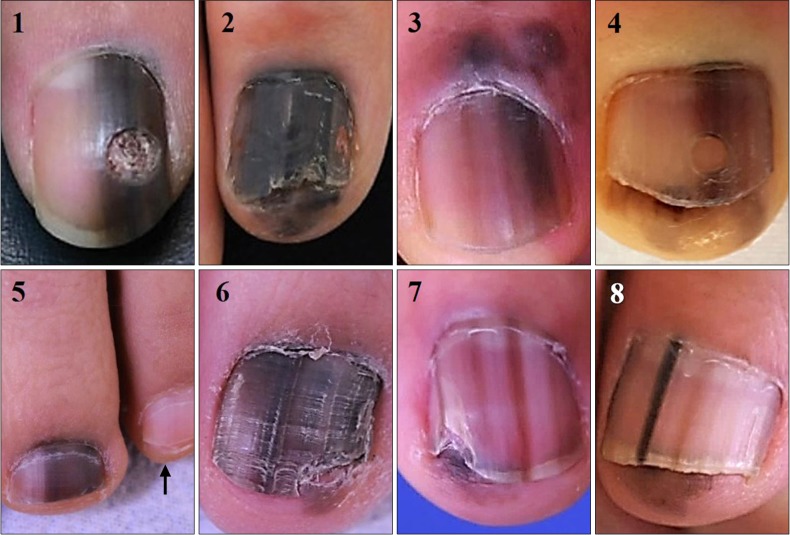
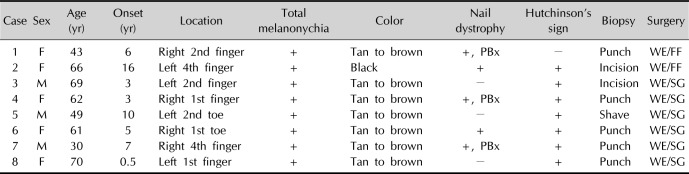
4. In addition, all of our cases had faint to dark background pigmentation in entire nail plate. Cases 1, 3, 4, 5, and 8 might be confused with partial melanonychia, but background pigmentation adjacent to the dark band was identified by comparing with unaffected opposite finger/toe or adjacent fingers/toes (
Fig. 1, arrow in case 5). Thus, we suggest that “dark brown bands in brown background” is a characteristic of SUM
in situ, adding background pigmentation to “B” of previous ABCDEF rule. We recommend clinical photograph should include all 10 fingers or toes for correct description of lesion. Background pigmentation can differentiate benign nail pigmentation disorder from SUM
in situ as well. In our previous study, 18 SUM
in situ were included. Fourteen out of 18 (77.8%) SUM
in situ had entirely pigmented nail plate
4. On the other hand, in our clinicopathological analysis of 28 nail matrix nevus (NMN) cases (not published), all except for 2 total melanonychia cases exhibited sharp band margin and normal colored nail plate adjacent to the lesion. Two total melanonychia cases were children. Moreover, in another study on NMN, there were adult cases with band breadth wider than two thirds of affected nail
12. Therefore, background pigmentation is more appropriate clinical criterion than specified band breadth for SUM
in situ. The information on change in nail band or treatment response of nail dystrophy at initial visit may be incorrect, because we have to rely on patient's memory. In addition, nail dystrophy is not frequently accompanied with SUM
in situ. Only about 25% of our case and 16.7% of our previous cases had nail dystrophy. In tertiary center setting, patients occasionally underwent nail unit biopsy already, and we cannot be sure that the nail dystrophy was occurred by melanoma or biopsy. Therefore, change in nail band or nail deformity may not be an appropriate item for clinical checklist when we initially examine the lesion suspected as SUM
in situ. “D” and “F” in the rule are only ancillary items. SUM should be suspected if a patient is adult and has melanonychia striata with background pigmentation whether “D” and “F” are satisfied or not. Moreover, more than half of our cases had lesion on digits other than thumbs or big toes and none of our cases had family or personal history of melanoma or dysplastic nevus syndrome. Levit et al.
6 commented that periungual pigmentation is usually late presenting sign of SUM, and not suitable for early detection of SUM although they included Hutchinson's sign in the ABCDEF rule. However, nearly 90% of our case showed Hutchinson's sign in spite of SUM
in situ. Eleven of 18 (61.1%) SUM
in situ in our previous study had Hutchinson's sign on periungual skin, but only 1 pediatric NMN had hyponychial pigmentation
4. Hyponychial pigmentation in 1 NMN case can be one of the atypical features found in pediatric NMNs
12. Periungual extension of pigmentation can happen before dermal invasion because radial growth phase usually precedes vertical growth except for nodular melanomas. In a previous case series of 9 acral lentiginous melanoma
in situ, mean duration of radial growth phase was 9.4 years
13. Three of them were SUM
in situ, and Hutchinson's sign was identified in two cases. We suggest “color in periungual skin” or “C” as the third rule. Finally, all 26 cases in this and our previous investigation had solitary lesion, namely, only 1 digit was affected. Multiple melanonychia is usually caused by benign disorders that can manifest as nail pigmentation, such as Laugier-Hunziker syndrome
1415 or Peutz-Jeghers syndrome
16. Although there was one case report on multiple primary SUM in 3 fingers, it was very exceptional and 3 lesions appeared sequentially, not at the same time
17. Therefore, SUM
in situ involves only one digit except for very rare cases. We suggest “single digit lesion” or “D” as the last rule.
The limitation of our study is small sample size, but all 8 cases satisfied ABCD rule. Furthermore, this strategy can be applied to our previous 18 SUM in situ.
Based on current data, we believe our nail ABCD rule is a simple, sensitive, and specific clinical tool for screening SUM in situ. For accurate documentation of lesion, we recommend clinical photography including whole 10 fingers or toes. Further refinement of this clinical criteria based on larger sample size is necessary to facilitate early recognition of SUM in situ.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download