Abstract
Background
Bee venom (BV) has been widely investigated for potential medical uses. Recent inadvertent uses of BV based products have shown to mitigate signs of fungal infections. However, the component mediating the antifungal effect has not been identified.
Objective
This investigation compares bee venom in its whole and partial forms to evaluate the possible component responsible for the antifungal effect.
Methods
Forty-eight plates inoculated with Trichophyton rubrum were allocated into four groups. The groups were treated with raw BV (RBV), melittin, apamin and BV based mist (BBM) respectively and each group was further allocated accordingly to three different concentrations. The areas were measured every other day for 14 days to evaluate the kinetic changes of the colonies.
Results
The interactions of ratio differences over interval were confirmed in groups treated with RBV and BBM. In RBV, the level of differences were achieved in groups treated with 10 mg/100 µl (p=0.026) and 40 mg/100 µl (p=0.000). The mean difference of ratio in groups treated with RBV was evident in day 3 and day 5. The groups that were treated with melittin or apamin did not show any significant interaction. In BBM groups, the significant levels of ratio differences over time intervals were achieved in groups treated with 200 µl/100 µl (p=0.000) and 300 µl/100 µl (p=0.030).
Bee venom (BV) extracted from Apis mellifera L. has been utilized for centuries as a suitable pain killer and an anti-inflammatory agent for various chronic diseases12345. A number of recent studies further state the anti-mutagenic, anti-nociceptive, radio-protective, anti-tumorous and antibiotic properties of the BV5678. Extensive research has been carried out to evaluate the effects of BV and the components of BV in that manner have been elucidated. Bioactive substances including melittin, apamin, mast cell degranulating peptides with histamine, serotonine, dopamine, norepinephrine and a number of enzymes listing phospholipase, hyaluronidase and histidine decarboxylase791011. Various pathways including inhibition of toll like receptors and translocation of nuclear factor kappa B (NF-κB) and activator protein-1 signaling are suggested mechanism to the noticeable anti-inflammatory effects of BV1213. Two major components of BV, melittin and phospholipase A2, are generally thought to play an important role in the induction of irritation and allergic reaction associated with the bee stings7. Melittin, a 26 amino acid polypeptide, has been known to have antibacterial effects789101112. Recently, melittin-loaded perfluorocarbon nanoparticles possessed the ability to safely deliver significant payloads of melittin intravenously and to target and kill tumor cells14.
In respect, medical and commercial application targeting anti-inflammatory effects of BV has been prevalently manufactured in different fields of Korea. Common products manufactured with BV include anti-acne sprays, anti-blemishes, moisturizers, and nutrient-providing gels. Additionally, antibacterial use of BV based mist (BBM) has been inadvertently used and engendered attention for alleviating signs of fungal infections. Antifungal effects of BV have generally been less underlined compared to other inflammatory oriented diseases. Articles regarding antifungal use of BV include species of Candidal origin and Trichophyton species15. The antifungal activities of BV and sweet BV against 10 clinical isolates of Candida albicans that were cultured from blood and the vagina showed antifungal activity determined by using the disk diffusion assay, the broth micro-dilution assay and the killing-curve assay15. Moreover, antifungal activity of BV against T. rubrum and T. mentagrophytes showed stronger effect than that of fluconazole1. However, the underlying mechanism and the principal component from BV that elicits the antifungal effect needs to be determined.
In this investigation, the components of BV including melittin and apamin, previously known to generate the anti-inflammatory effect, along with the BV as in whole raw form and mist based product were separately applied to the colonies of T. rubrum to evaluate the possible component responsible for the antifungal effect.
The Colonies of natural honeybees (Apis mellifera L.) were maintained at the National Academy of Agricultural Science, Korea and the BV was collected by the collecting device (Chung Jin Biotech Co., Ltd., Ansan, Korea). An electric current was generated to the hive to cause the bees to sting at the glass plate. The venom which was later dried and was scraped off. The collected venom was diluted in cold sterile water and was centrifuged at 10,000g for 5 minutes at 4℃ to eliminate residues from the supernatant. BV was lyophilized by freeze dryer and refrigerated at 4℃ for later use. The BV used in this experiment was confirmed with size exclusion gel chromatography (AKTA Explorer; GE Healthcare, Pittsburgh, PA, USA) by dissolving in 0.02 M phosphate buffer with 0.25 M NaCl adjusted to pH 7.2 using a Superdex Peptide column (Amersham Biosciences; GE Healthcare)14. Other components including melittin and apamin were manufactured products at Sigma (St. Louis, MO, USA) and BBM (A.C. Care Bee's water essence) of Dongsung Pharmaceuticals (Seoul, Korea).
The medium used to culture the fungus was composed of potato dextrose cornmeal agar (PDACC; Catholic Skin Clinic, Daegu, Korea) with peptone, Tween 80 and antibiotics (chloramphenicol 500 mg L−1 and cycloheximide 500 mg L−1). Standard sized inoculums of T. rubrum derived from a spore suspension were applied to PDACC plates. The spore suspension was prepared by applying 5 ml of distilled water (DW) to a 3-week-old T. rubrum culture that was later gently withdrawn with a sterile pipette. Each PDACC plate was divided in half by scrapping off the midline in a sterile manner to retain identical conditions for both the experimental and the control groups. Using a sterile spreader, the spore suspension was applied on both sides of the PDACC plate. Forty eight plates were divided into four groups according to the specimen applied which were RBV, melittin, apamin and BBM. Every twelve plates were allocated and were further divided into three groups and labeled accordingly to the differently concentrated components with DW. The components were applied to the margins of the colonies and the amount of each component was variable as the areas of the colonies were different among the groups (Group 1: RBV 0.1 mg/DW 100 µl, Group 2: RBV 10 mg/DW 100 µl, Group 3: RBV 40 mg/DW 100 µl, Group 4: melittin 0.5 mg/100 DW µl, Group 5: melittin 1.0 mg/DW 100 µl, Group 6: melittin 1.5 mg/DW 100 µl, Group 7: apamin 0.5 mg/DW 100 µl, Group 8: apamin 1.0 mg/DW 100 µl, Group 9: apamin 1.5 mg/DW 100 µl, Group 10: BBM 100 µl/DW 100 µl, Group 11: BBM 200 µl/DW 100 µl, Group 12: BBM 300 µl/DW 100 µl).
The antifungal activity was measured by evaluating the area changes of each group for 14 days in an interval of two. Digital photography was taken every day using Canon EOS 750D (Canon Inc., Japan) and was followed until the 14th day. The lighting, position, and the background of the shooting were kept consistent throughout the experiment. The area was then converted into numerical values using Image Processing and analysis in Java (Image J version 1.50i; National Institutes of Health, Bethesda, MD, USA) which was then recalculated into ratios in order to calculate the kinetic interval changes of the areas observed in the colonies. The ratio at each interval indicates the area ratio to the previously calculated area in order to evaluate interval changes and the interaction among the intervals.
All data was gathered and transformed in numerical values. Primary efficacy endpoint was the area difference of the colonies from the baseline to day 13. This was calculated with a repetitive measurement linear-effects model, which included the study groups, baseline value, scheduled follow-up intervals, and the interaction of study group with scheduled intervals. The descriptive data are expressed in mean±standard deviation. All analysis was performed using IBM SPSS Statistics ver. 19.0 (IBM Co., Armonk, NY, USA) with p-value of less than 0.05 as statistically significant value. The analysis evaluated the interaction of the experimental group and the control over time intervals.
The antifungal efficacies of various components were treated against T. rubrum as shown in (Fig. 1). The PDACC plates were divided and spore suspension was applied on both sides of the plates. The left indicates the control group while the right side indicates experimental group. The experimental groups that were treated with raw BV shows a slower growth difference compared to than that of the control group at 10 mg/100 µl and 40 mg/100 µl concentrations (Fig. 1B, C). Noticeably growth rate difference in BBM treated groups between the experimental groups at 200 µl and 300 µl concentrations were also observed on gross inspection (Fig. 1K, L).
For every component, three differently graded concentrations were treated on each colony and the areas were measured every other day for 14 days. The numerical values were then transformed into ratios in order to evaluate the actual level of area difference and interaction of the growth nature throughout the study.
The main effect between the groups while ignoring the interval variable was only statistically significant in the group with 40 mg/100 µl concentration (p=0.000), which was noticeable compared to no statistically significant values observed in groups with 0.1 mg/100 µl concentration (p=0.054) and 10 mg/100 µl (p=0.098). The main effect among the intervals, while ignoring the group variable showed statistically significant value within all three concentration groups. The interaction effect between the groups over time interval was not statistically significant in the group treated with 0.1 mg/100 µl concentrated of RBV. However the values were different as the concentrations of RBV were increased to 10 mg/100 µl and 40 mg/100 µl with respective p-values of 0.026 and 0.000 (Table 1). The mean difference of ratio in groups treated with RBV of 10 mg/100 µl was evident in day 3 and day 5 (Fig. 2B). The mean ratio was 1.152 in the experimental group compared to 1.425 in the control group at day 3 and 1.122 in the experimental group compared to 1.403 in the control group at day 5 (Table 1). Similar result was shown in the group treated RBV with 40 mg/100 µl. The evident difference was seen in day 3 and day 5 (Fig. 2C). At day 3, 1.075 ratio of area increment in the experimental group was observed compared to 1.514 in the control group (Table 1). At day 5, 1.055 ratio of area increment in the experimental group was observed compared to 1.322 in the control group (Table 1).
All of the groups that were treated with melittin or apamin, regardless of concentration gradient, the significant main effect of ratio differences were not observed in any of the measures within the intervals, the groups or within the groups over specific time intervals (Table 1).
In BBM, similar pattern to RBV was observed. While, the group treated with BBM of 100 µl exhibited no significant difference of ratio difference between the groups over time intervals (p=0.052), the higher concentration groups showed significant levels of ratio differences within the groups over time intervals (p=0.000 in 200 µl, 0.030 in 300 µl), (Table 1). The mean difference of ratio in the group treated with 200 µl of BBM was noticeable at day 3 and at day 7 (Fig. 2K). At day 3, the mean ratio difference was 1.072 in the experimental group and 1.387 in the control group (Table 1). Additional the mean ratio difference was 1.090 in the experimental group and 1.218 in the control group at day 7 (Table 1). Moreover, the mean difference of ratio in the group treated with 300µl of BBM was noticeable at day 3 (Fig. 2L). The mean ratio difference was 1.130 in the experimental group and 1.284 in the control group (Table 1).
BV or its series of components have shown a broad array of effects from anti-inflammatory agents, anti-nociceptive, antibacterial, to anti-tumorous effects12345678. Extensive in vitro studies have confirmed the versatile properties that BV retains. In respect to the wide-ranging studies performed with BV, information regarding the antifungal effect of BV is still scarce. However, continual scope of research and aim conducting BV in relation to antifungal properties has stated BV as a possible agent for its medicinal application.
Moreover, off-label uses of BBM products have been targeted to control acne eruptions. Based on the reports of robust antibacterial activities of BV against both Gram negative and Gram positive bacteria, in addition to the role of BV in inflamed lesions, a number of BV based acne care products have been manufactured. However theses lines of beauty products are inadvertently used and alleviation of lesions with tinea pedis have been suggested by these products.
Therefore, the objective of this study was to identify the possible underlying component in BV that hinders the growth and spreading of the most common fungal pathogen, T. rubrum. In the study comparing the antifungal effects between the experimental groups and the control groups at certain time interval, only BV in its whole form regardless of raw nature or in a mist based product showed statistically significant values. In elaboration, the time point regarding the antifungal effect exhibited by RBV was dose dependent as 10 mg/100 µl (p=0.026) and 40 mg/100 µl (p=0.000) concentrated specimen showed statistically significant area difference compared to that of 0.1 mg/100 µl (p=0.084). Based on this finding, some level of fungicidal or fungistatic properties produced by raw bee venom can be inferred. Furthermore, as the antifungal effect of bee venom generally withered after day 5, a time span of 5 days for its effectiveness of RBV was also postulated. Prominent difference according to time interval is exhibited through graphical values (Fig. 2). The antifungal effect was not observed in colonies that were treated either in melittin or apamin. The analysis between the experimental groups and the control groups did not show any statistically significant difference regarding the fungal growth on any concentration level. Similar results were exhibited in the colonies treated with apamin. Melittin is known to inhibit platelet-derived growth factor and vascular smooth muscle cell proliferation by suppressing NF-κB, Akt activation, and the mitogen-activated protein kinase pathway1415161718. Downward reaction inhibits the transcription of inflammatory cytokine which exerts various protective events induced from melittin. No parameter regarding cell growth or antifungal effect in that manner was observed.
Interestingly, the BBM has shown similar effectiveness to RBV in terms of growth retardation. The concentration levels effective for in hindering the colony growth were prominent in the ones treated with 10 mg/100 µl (p=0.000) and in 40 mg/100 µl (p=0.030). The underlying factor may be due to a variety of component contained in mist. Extracts of bee venom, extracts of royal jelly, propolis, grapefruit, Beta-glucan, phytoncide, and portulaca oleracea extracts were in the material. In addition to bee venom, propolis may have also played a role in the antifungal effect. Farghaly17 reported the use of propolis on tested microscopic fungi, Aspergillus fumigatus (19.2±0.63 mm) showed the most sensitive results to the component1617. However, the sensitivity decreased in other microscopic fungi including Geotricum candidum, C. albicans (16.9±0.58, 16.4±1.2), respectively, which yet remain to be determined17. However since many of the bee venom based products are already available through highly refined beauty products such as the one used in this study, it may be relevantly useful to directly apply such products to the affected area upon more safety and clinical evaluation.
Numerous studies have been investigated to evaluate the antifungal properties of bee venom. Lee reported that anti-candidal activities of bee venom and sweet bee venom were observed by using the disk diffusion method and the broth microdilution method, confirming that the compounds have a potentialfor use as anti-candidal agents1315. Phytochemicals exhibiting antifungal effects against T. rubrum and T. mentagrophytes were evaluated in different studies. Turmeric oil from Curcuma longa had minimum inhibitory concentrations (MICs) in a range of 229.8~919.2 ppm (parts per million)18. Four phenolic amides, dihydro-N-caffeoyltyramine, trans-N-feruloyloctopamine, trans N caffeoyltyramine, and cis-N-caffeoyltyramine isolated from Lycium chinense were reported to have anti-fungal activity in a range of 5~10 ppm19. Other molecules such as 6α-O-(β-D-xylopyranosyl-(1→3)-β-D-quinovopyranosyl)-(25,S)-5α-spirostan-3β-ol had IC50 values of 25 µg/ml had also shown antifungal effect agains against T. mentagrophytes and T. rubrum20. Limonene was also shown to exert a potent antifungal effect against T. rubrum with MIC value of 0.5%21.
In respect to these findings, this experiment strived to assess the antifungal effect from each component underlying cause of effect and interesting result is that only with the bee venom in its whole form delivered a significant level of inhibition and we concluded that the venom in separated forms are not effective.
One theory is that while mellitin induces reactive oxygen species to generate the antifungal effect, other components such as phospholipase A2, hyaluronidase which respectively raise the permeation of the cell membrane and the capillary membranes are necessary to give the whole antifungal effect113142223. Additionally, studies state that the lipids and carbohydrates in the venom may act as an opsonin for further inflammatory responses to repeatedly generate the antifungal effect2223.
Although there are a number of studies evaluating the antifungal effect of bee venom, component-specific evaluation within this mixture has not been evaluated before this study. In addition, the use and effect of bee venom based products was also conducted to evaluate the antifungal effect exerted from these lines of products. The investigation was conclusive with the specified followings. First of all, the antifungal effect was observed in bee venom regardless of raw or manufactured form. The colonies that were treated with only the whole form of bee venom have proven the antifungal effect throughout our study. Separated forms of bee venom components showed no value in hindering the growth of the colonies. Secondly, the effects were dose dependent as statistically significant values were observed within the differently concentrated groups at different time period. Thirdly, the effect showed a lasting period as the colony sizes were mostly regressed up till the 5th day. It was conclusive that the 5th day was the critical time point as the effect started to wear off after day 5 and further re-application of the material seems necessary in order to generate the continual antifungal effect. Moreover, in terms of bee venom based product, successive research must be considered before recommending its use against various fungal infections. However, the already prevalent use of these manufactured products along with approved safety concerns may ease the commercialization of these manufactured products for targeting fungal infections.
Even though, this study demonstrated the antifungal effects by the components of bee venom along with bee venom based beauty product, further experiments with larger sample sizes, more fungal species and more components of the bee venom should be carried out to evaluate the molecular interaction and possible underlying mechanisms among the components. Further in vivo efficacy studies are warranted for clinical application.
ACKNOWLEDGMENT
This work was carried out with the support of “Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ01132501)” Rural Development Administration, Republic of Korea.
References
1. Yu AR, Kim JJ, Park GS, Oh SM, Han CS, Lee MY. The antifungal activity of bee venom against dermatophytes. J Appl Biol Chem. 2012; 55:7–11.

2. Kwon YB, Lee JD, Lee HJ, Han HJ, Mar WC, Kang SK, et al. Bee venom injection into an acupuncture point reduces arthritis associated edema and nociceptive responses. Pain. 2001; 90:271–280. PMID: 11207399.

3. Kim KS, Choi US, Lee SD, Kim KH, Chung KH, Chang YC, et al. Effect of bee venom on aromatase expression and activity in leukaemic FLG 29.1 and primary osteoblastic cells. J Ethnopharmacol. 2005; 99:245–252. PMID: 15894134.

4. Peng XL, Gao XL, Chen J, Huang X, Chen HS. Effects of intravenous Injections Paederiae and Stauntonia on spontaneous pain, hyperalgesia and inflammation induced by cutaneous chemical tissue injury in the rat. Sheng Li Xue Bao. 2003; 55:516–524. PMID: 14566397.
5. Han SM, Lee KG, Yeo JH, Kweon HY, Kim BS, Kim JM, et al. Antibacterial activity of the honey bee venom against bacterial mastitis pathogens infecting dairy cows. Int J Indust Entomol. 2007; 14:137–142.
6. Kwon YB, Lee HJ, Han HJ, Mar WC, Kang SK, Yoon OB, et al. The water-soluble fraction of bee venom produces antinociceptive and anti-inflammatory effects on rheumatoid arthritis in rats. Life Sci. 2002; 71:191–204. PMID: 12031688.

7. Kim HW, Kwon YB, Ham TW, Roh DH, Yoon SY, Lee HJ, et al. Acupoint stimulation using bee venom attenuates formalin-induced pain behavior and spinal cord fos expression in rats. J Vet Med Sci. 2003; 65:349–355. PMID: 12679565.

8. Lee SM, Lim J, Lee JD, Choi DY, Lee S. Bee venom treatment for refractory postherpetic neuralgia: a case report. J Altern Complement Med. 2014; 20:212–214. PMID: 24093469.

9. Argiolas A, Pisano JJ. Facilitation of phospholipase A2 activity by mastoparans, a new class of mast cell degranulating peptides from wasp venom. J Biol Chem. 1983; 258:13697–13702. PMID: 6643447.

10. Akdis CA, Akdis M, Blesken T, Wymann D, Alkan SS, Müller U, et al. Epitope-specific T cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J Clin Invest. 1996; 98:1676–1683. PMID: 8833918.

11. Han S, Lee K, Yeo J, Baek H. Determination of major constituents of honeybee venom from Korea. Korean J Apic. 2009; 24:175–178.
12. Lee G, Bae H. Anti-Inflammatory applications of melittin, a major component of bee venom: detailed mechanism of action and adverse effects. Molecules. 2016; 21:E616. PMID: 27187328.

13. Park C, Lee DG. Melittin induces apoptotic features in Candida albicans. Biochem Biophys Res Commun. 2010; 394:170–172. PMID: 20188067.

14. An HJ, Kim KH, Lee WR, Kim JY, Lee SJ, Pak SC, et al. Anti-fibrotic effect of natural toxin bee venom on animal model of unilateral ureteral obstruction. Toxins (Basel). 2015; 7:1917–1928. PMID: 26035488.
15. Lee SB. Antifungal activity of bee venom and sweet bee venom against clinically isolated Candida albicans. J Pharmacopuncture. 2016; 19:45–50. PMID: 27280049.

16. Han SM, Lee KG, Yeol JH, Baek HJ, Park KK. Antibacterial and anti-inflammatory effects of honeybee (Apis mellifera) venom against acne-inducing bacteria. J Med Plants Res. 2010; 4:459–464.
17. Farghaly DS. Effect of some honey bee and wasp products on some pathogenic bacteria and fungi: in vitro study. Middle East J Appl Sci. 2016; 6:468–473.
18. Apisariyakul A, Vanittanakom N, Buddhasukh D. Antifungal activity of turmeric oil extracted from Curcuma longa (Zingiberaceae). J Ethnopharmacol. 1995; 49:163–169. PMID: 8824742.

19. Lee MH, Lee KB, Oh SM, Lee BH, Chee HY. Antifungal activities of dieckol isolated from the marine brown alga Ecklonia cava against Trichophyton rubrum. J Korean Soc Appl Biol Chem. 2010; 53:504–507.

20. Arif T, Mandal TK, Dabur R. Natural products: anti-fungal agents derived from plants. In : Tiwari VK, Mishra BB, editors. Opportunity, challenge and scope of natural products in medicinal chemistry. Kerala: Research Signpost;2011. p. 283–311.
21. Chee HY, Kim H, Lee MH. In vitro antifungal activity of limonene against Trichophyton rubrum. Mycobiology. 2009; 37:243–246. PMID: 23983542.
22. Son DJ, Lee JW, Lee YH, Song HS, Lee CK, Hong JT. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharmacol Ther. 2007; 115:246–270. PMID: 17555825.

23. Eiseman JL, von Bredow J, Alvares AP. Effect of honeybee (Apis mellifera) venom on the course of adjuvant-induced arthritis and depression of drug metabolism in the rat. Biochem Pharmacol. 1982; 31:1139–1146. PMID: 6177321.

Fig. 1
Serial photographs of Trichophyton rubrum colony plates from day 1 to day 13. The colony plates varying each component group. Each plate field is divided in half to inoculate the colonies of experimental (right side of field) and control (left side of field) groups. (A) 0.1 mg/100 µl of raw bee venom (RBV) treated group, (B) 10 mg/100 µl of RBV treated group, (C) 40 mg/100 µl of RBV treated group, (D) 0.5 mg/100 µl of melittin treated group, (E) 1.0 mg/100 µl of melittin treated group, (F) 1.5 mg/100 µl of melittin treated group, (G) 0.5 mg/100 µl of apamin treated group, (H) 1.0 mg/100 µl of apamin treated group, (I) 1.5 mg/100 µl of apamin treated group, (J) 100 µl/100 µl of bee venom based mist (BBM) treated group, (K) 200 µl/100 µl of BBM treated group, (L) 300 µl/100 µl of BBM treated group.

Fig. 2
Graphical interpretation of ratio interaction over time in experimental and control groups. Graphical interpretation of ratio difference interaction over time (interval). (A) 0.1 mg/100 µl of raw bee venom (RBV) treated group, (B) 10 mg/100 µl of RBV treated group, (C) 40 mg/100 µl of RBV treated group, (D) 0.5 mg/100 µl of melittin treated group, (E) 1.0 mg/100 µl of melittin treated group, (F) 1.5 mg/100 µl of melittin treated group, (G) 0.5 mg/100 µl of apamin treated group, (H) 1.0 mg/100 µl of apamin treated group, (I) 1.5 mg/100 µl of apamin treated group, (J) 100 µl/100 µl of bee venom based mist (BBM) treated group, (K) 200 µl/100 µl of BBM treated group, (L) 300 µl/100 µl of BBM treated group.
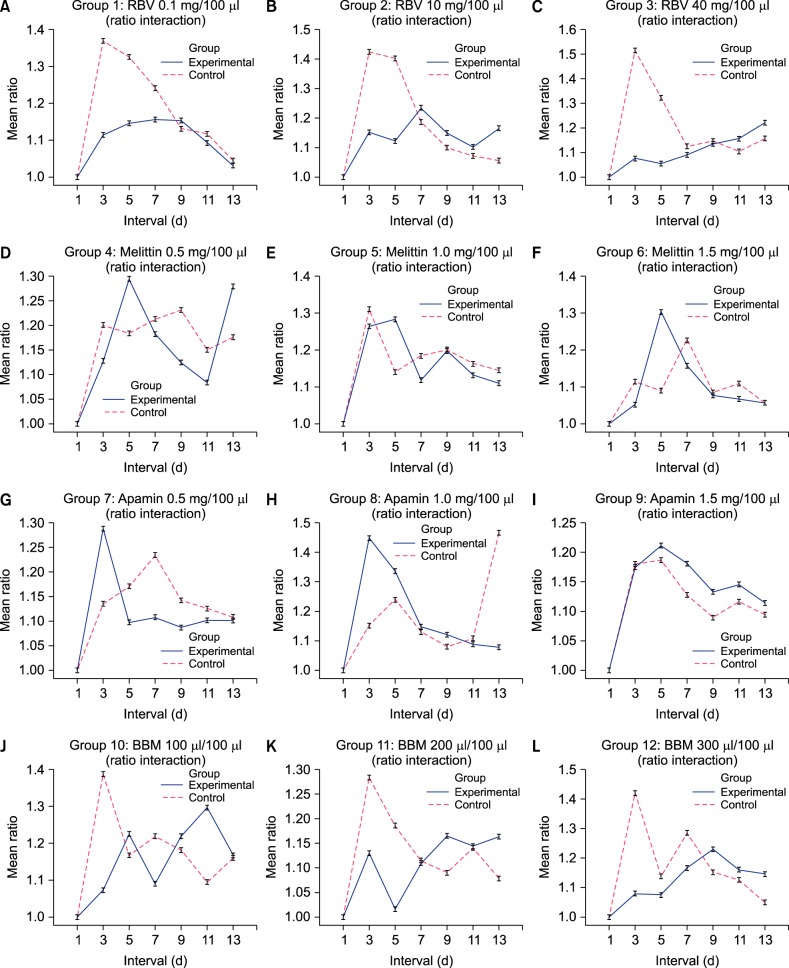
Table 1
Ratio interaction of groups treated with RBV, melittin, apamin, BBM




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download