Abstract
Purpose
To identify the correlation between nasal eosinophilia and aeroallergen sensitization in children and adolescents.
Methods
This is a retrospective study of patients below 18 years of age who had a history of rhinitis that lasted more than 2 weeks or had been repeated more than once a year, received nasal eosinophil examinations, and had serum specific IgE to aeroallergens measured at an Allergy Clinic in a single tertiary teaching hospital in Seoul, Korea. The percentage of nasal eosinophils was calculated by the number of eosinophils per total leukocytes in a high-power field of 1,000× . Data was analyzed to determine the association between nasal eosinophilia and 18 aeroallergens.
Results
Of the 245 patients included, 156 (63.7%) were male and the mean age (±standard deviation) was 7.9 years (±3.8). In total, 175 patients (71.4%) were sensitized to at least 1 of the 18 aeroallergens tested, and sensitization to house dust mite was most common. In addition, 118 (48.2%) and 69 patients (28.2%) had nasal eosinophilia of at least 1% and 5%, respectively. There were no significant correlations between serum total IgE or age and the percentage of nasal eosinophils. However, the percentage of nasal eosinophils in the group sensitized to any aeroallergens was significantly increased compared to the nonsensitized group (P=0.002). The percentage of nasal eosinophils was significantly higher in patients who were sensitized to Birch-Alder Mix, oak white, Bermuda grass, orchard grass, timothy grass, sweet vernal grass, rye, mugwort, short ragweed, Alternaria alternata, cats, dogs or Dermatophagoides farinae compared to those nonsensitized.
REFERENCES
1. Settipane RA, Lieberman P. Update on nonallergic rhinitis. Ann Allergy Asthma Immunol. 2001; 86:494–507.

2. Kay AB. Allergy and allergic diseases. First of two parts. N Engl J Med. 2001; 344:30–7.
3. Ahmadiafshar A, Taghiloo D, Esmailzadeh A, Falakaflaki B. Nasal eosinophilia as a marker for allergic rhinitis: a controlled study of 50 patients. Ear Nose Throat J. 2012; 91:122–4.

4. Lans DM, Alfano N, Rocklin R. Nasal eosinophilia in allergic and nonallergic rhinitis: usefulness of the nasal smear in the diagnosis of allergic rhinitis. Allergy Proc. 1989; 10:275–80.

5. Miller RE, Paradise JL, Friday GA, Fireman P, Voith D. The nasal smear for eosinophils. Its value in children with seasonal allergic rhinitis. Am J Dis Child. 1982; 136:1009–11.
6. Miri S, Farid R, Akbari H, Amin R. Prevalence of allergic rhinitis and nasal smear eosinophilia in 11- to 15 yr-old children in Shiraz. Pediatr Allergy Immunol. 2006; 17:519–23.
7. Canakcioglu S, Tahamiler R, Saritzali G, Alimoglu Y, Isildak H, Guvenc MG, et al. Evaluation of nasal cytology in subjects with chronic rhinitis: a 7-year study. Am J Otolaryngol. 2009; 30:312–7.

8. Mandhane SN, Shah JH, Thennati R. Allergic rhinitis: an update on disease, present treatments and future prospects. Int Immunopharmacol. 2011; 11:1646–62.

9. Mullarkey MF, Hill JS, Webb DR. Allergic and nonallergic rhinitis: their characterization with attention to the meaning of nasal eosinophilia. J Allergy Clin Immunol. 1980; 65:122–6.

10. Lee HS, Majima Y, Sakakura Y, Shinogi J, Kawaguchi S, Kim BW. Quantitative cytology of nasal secretions under various conditions. Laryngoscope. 1993; 103:533–7.

11. Jirapongsananuruk O, Vichyanond P. Nasal cytology in the diagnosis of allergic rhinitis in children. Ann Allergy Asthma Immunol. 1998; 80:165–70.

12. Bentley AM, Jacobson MR, Cumberworth V, Barkans JR, Moqbel R, Schwartz LB, et al. Immunohistology of the nasal mucosa in seasonal allergic rhinitis: increases in activated eosinophils and epithelial mast cells. J Allergy Clin Immunol. 1992; 89:877–83.

13. Juliusson S, Pipkorn U, Karlsson G, Enerbäck L. Mast cells and eosinophils in the allergic mucosal response to allergen challenge: changes in distribution and signs of activation in relation to symptoms. J Allergy Clin Immunol. 1992; 90(6 Pt 1):898–909.

14. Takwoingi Y, Akang E, Nwaorgu G, Nwawolo C. Comparing nasal secretion eosinophil count with skin sensitivity test in allergic rhinitis in Ibadan, Nigeria. Acta Otolaryngol. 2003; 123:1070–4.

15. Romero JN, Scadding G. Eosinophilia in nasal secretions compared to skin prick test and nasal challenge test in the diagnosis of nasal allergy. Rhinology. 1992; 30:169–75.
16. Kim GR, Chung TI, Kim KM. Clinical and statistical study of allergens in the allergic rhinitis. Yonsei Med J. 1975; 16:54–64.

17. Jung BS, Cho YS, Kim E, Yoon JS, Kim HH, Lee JS. Association between nasal eosinophil count and other clinical indices in children with chronic nasal symptoms. Korean J Asthma Allergy Clin Immunol. 2008; 28:214–9.
18. Powe DG, Bonnin AJ, Jones NS. 'Entopy': local allergy paradigm. Clin Exp Allergy. 2010; 40:987–97.

19. Cheng KJ, Zhou ML, Xu YY, Zhou SH. The role of local allergy in the nasal inflammation. Eur Arch Otorhinolaryngol. 2017; 274:3275–81.

20. Rondón C, Romero JJ, López S, Antúnez C, Martín-Casañez E, Torres MJ, et al. Local IgE production and positive nasal provocation test in patients with persistent nonallergic rhinitis. J Allergy Clin Immunol. 2007; 119:899–905.

21. Rondón C, Campo P, Herrera R, Blanca-Lopez N, Melendez L, Canto G, et al. Nasal allergen provocation test with multiple aeroallergens detects polysensitization in local allergic rhinitis. J Allergy Clin Immunol. 2011; 128:1192–7.

22. Pelikan Z. The changes in the nasal secretions of eosinophils during the immediate nasal response to allergen challenge. J Allergy Clin Immunol. 1983; 72:657–62.

23. Choi SS, Shin KS, Hahn YS. Correlation of appearance of nasal eosinophils with levels of total eosinophil counts, total IgE, and house dust mite specific IgE in children with symptoms of rhinitis. Pediatr Allergy Respir Dis. 2005; 15:35–43.
Fig. 1.
Comparison of nasal eosinophils according to sensitization to inhalant allergens. Mann-Whitney test was used for statistical analysis. ∗ P<0.05.
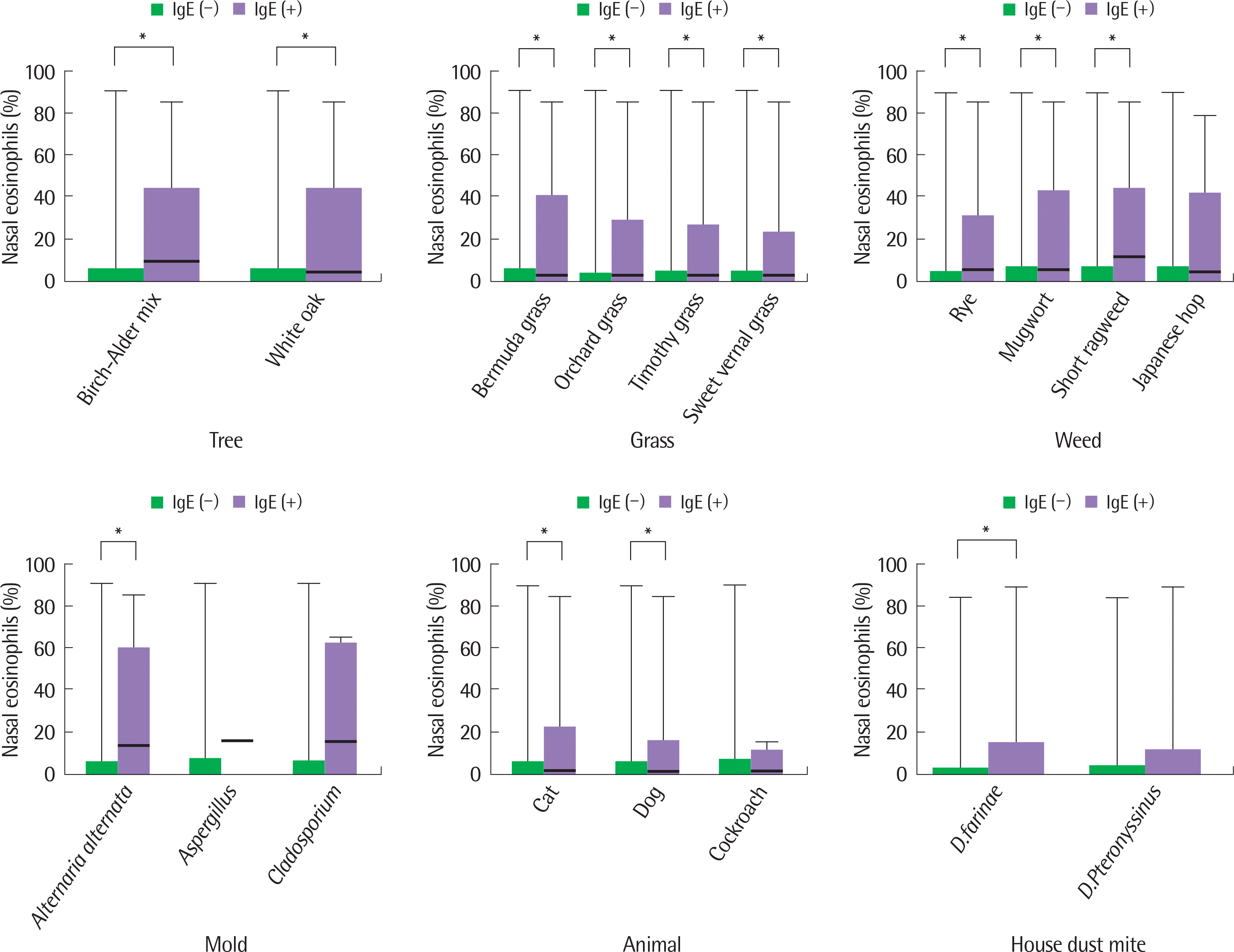
Fig. 2.
Correlation between nasal eosinophils and serum total IgE in 106 patients. Nasal eosinophils tended to increase with increasing total IgE level but not statistically significant.
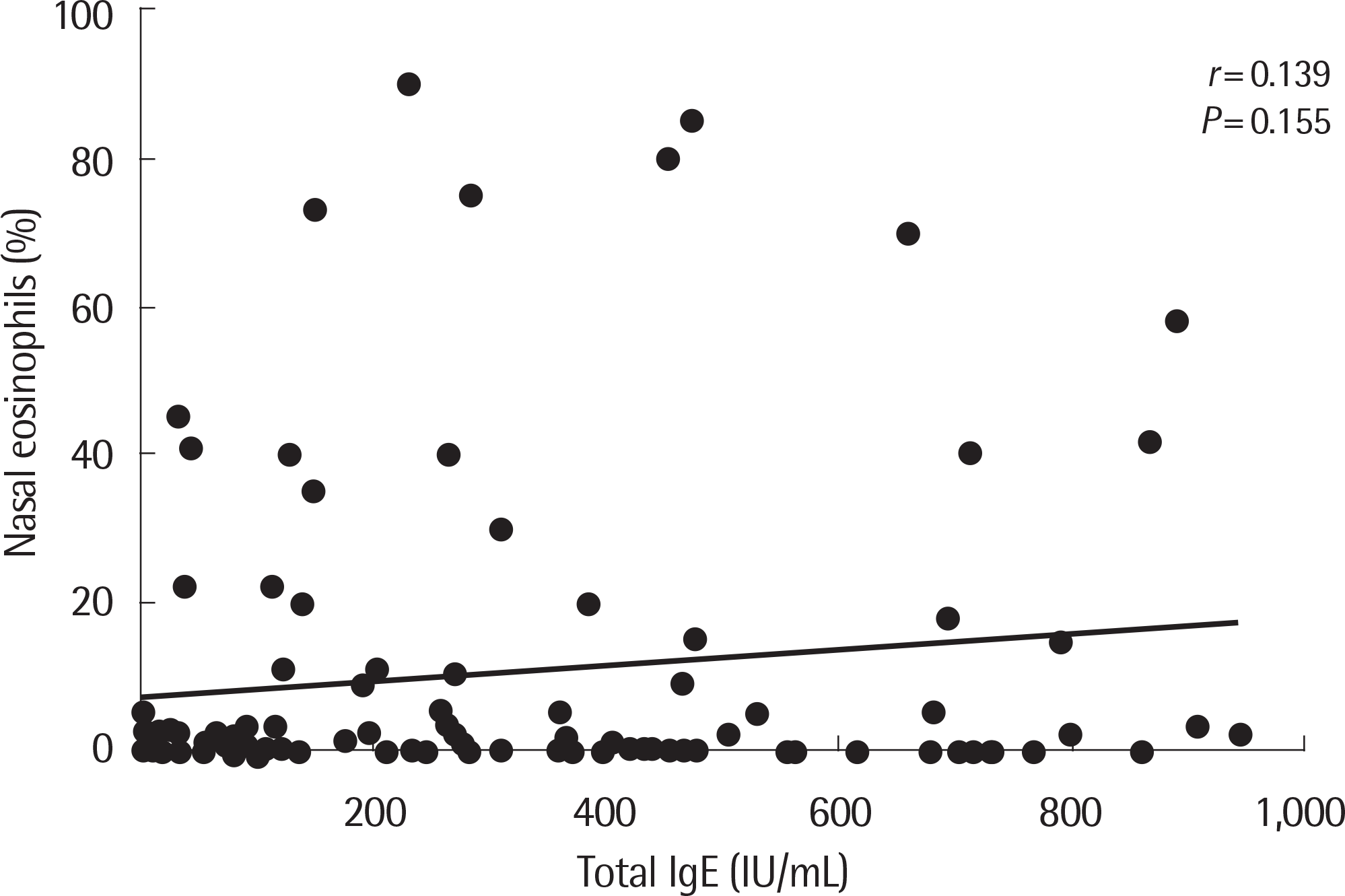
Fig. 3.
Correlation of nasal eosinophils to age. There was no correlation between nasal eosinophils and age.
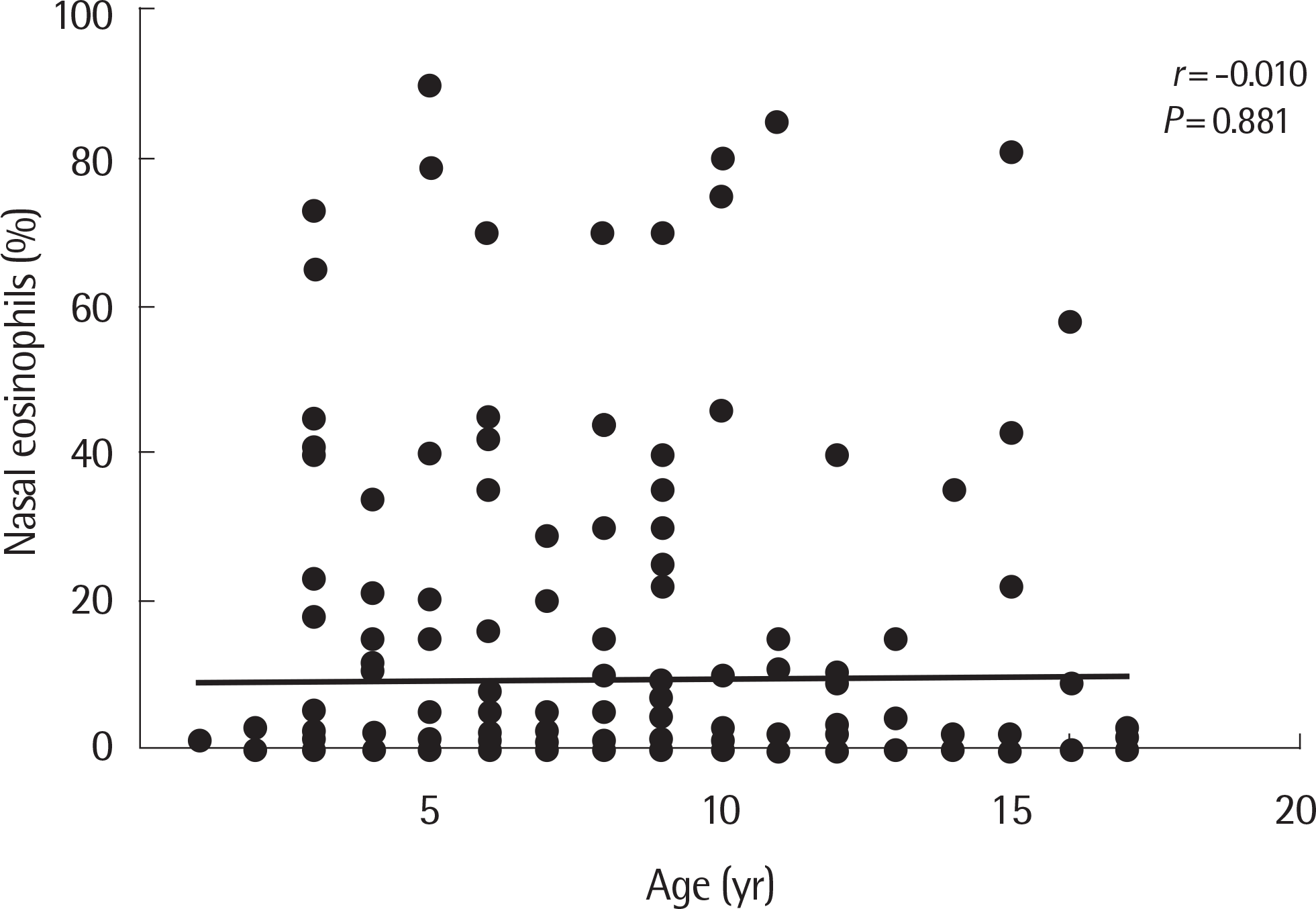
Table 1.
Characteristics of study participants (n=245)
| Characteristic | Value |
|---|---|
| Age (yr) | |
| Mean±SD | 7.9±3.8 |
| Median (IQR) | 7.0 (1–17) |
| Male sex | 156 (63.7) |
| Eosinophils in nasal smear (% of total leukocytes), median (IQR) | 0 (0–7) |
| Clinical diagnosis∗ | |
| AR | 59 (24.1) |
| AR + paranasal sinusitis | 40 (16.3) |
| AR + asthma or wheezing | 33 (13.5) |
| AR + common cold | 11 (4.5) |
| Paranasal sinusitis | 46 (18.8) |
| Asthma or wheezing | 5 (2.0) |
| Common cold | 17 (6.9) |
| NARES | 4 (1.6) |
| Other rhinitis | 30 (12.2) |
| Sensitization to aeroallergen | |
| Birch-Alder mix | 25 (10.2) |
| White oak | 15 (6.1) |
| Bermuda grass | 30 (12.2) |
| Orchard grass | 42 (17.1) |
| Timothy grass | 37 (15.1) |
| Sweet vernal grass | 40 (16.3) |
| Rye | 39 (15.9) |
| Mugwort | 23 (9.4) |
| Short ragweed | 22 (9.0) |
| Japanese hop | 22 (9.0) |
| Alternaria alternata | 18 (7.3) |
| Aspergillus | 1 (0.4) |
| Cladosporium | 5 (2.0) |
| Cat | 44 (18.0) |
| Dog | 36 (14.7) |
| Cockroach | 6 (2.4) |
| D. farinae | 136 (55.5) |
| D. pteronyssinus | 138 (56.3) |
Table 2.
Comparison of nasal eosinophils according to sensitization to inhalant allergen
| Aeroallergen | Nasal eosinophils (%) | P-value† | |||
|---|---|---|---|---|---|
| Antigen-specific IgE postive | Antigen-specific IgE negative | ||||
| No. | Median (IQR) | No. | Median (IQR) | ||
| Tree | |||||
| Birch-Alder mix | 25 | 9 (0–44) | 220 | 0 (0–5) | 0.006∗ |
| White oak | 15 | 4 (0–46) | 230 | 0 (0–5) | 0.028∗ |
| Grass | |||||
| Bermuda grass | 30 | 3 (0–40.5) | 215 | 0 (0–5) | 0.024∗ |
| Orchard grass | 42 | 3 (0–29.3) | 203 | 0 (0–4) | 0.007∗ |
| Timothy grass | 37 | 3 (0–27) | 208 | 0 (0–4.8) | 0.007∗ |
| Sweet vernal grass | 40 | 3 (0–23.3) | 205 | 0 (0–4.5) | 0.013∗ |
| Weed | |||||
| Rye | 39 | 4 (0–30) | 206 | 0 (0–3.3) | 0.002∗ |
| Mugwort | 23 | 4 (0–42) | 222 | 0 (0–5) | 0.029∗ |
| Short ragweed | 22 | 10 (0–43) | 223 | 0 (0–5) | 0.009∗ |
| Japanese hop | 22 | 3 (0–40.5) | 223 | 80 (0–5) | 0.065 |
| Mold | |||||
| Alternaria alternata | 18 | 12.5 (0.8–59.8) | 227 | 0 (0–5) | 0.001∗ |
| Aspergillus | 1 | 15 | 244 | 0 (0–6.5) | 0.192 |
| Cladosporium | 5 | 15 (0–61.5) | 240 | 0 (0–5) | 0.221 |
| Animal | |||||
| Cat | 44 | 1.5 (0–22) | 201 | 0 (0–5) | 0.030∗ |
| Dog | 36 | 1.5 (0–15) | 209 | 0 (0–5) | 0.047∗ |
| Cockroache | 6 | 1.5 (0–10.5) | 239 | 0 (0–7) | 0.920 |
| HDM | |||||
| D. farinae | 136 | 1 (0–14.3) | 109 | 0 (0–2.5) | 0.027∗ |
| D. pteronyssinus | 138 | 0 (0–10.3) | 107 | 0 (0–3) | 0.523 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download