Abstract
Objective
To investigate the incidence of early severe hypophosphatemia, and examine the associated clinical factors and outcomes, in extremely low birth weight infants (ELBWI) who received early amino acid administration.
Methods
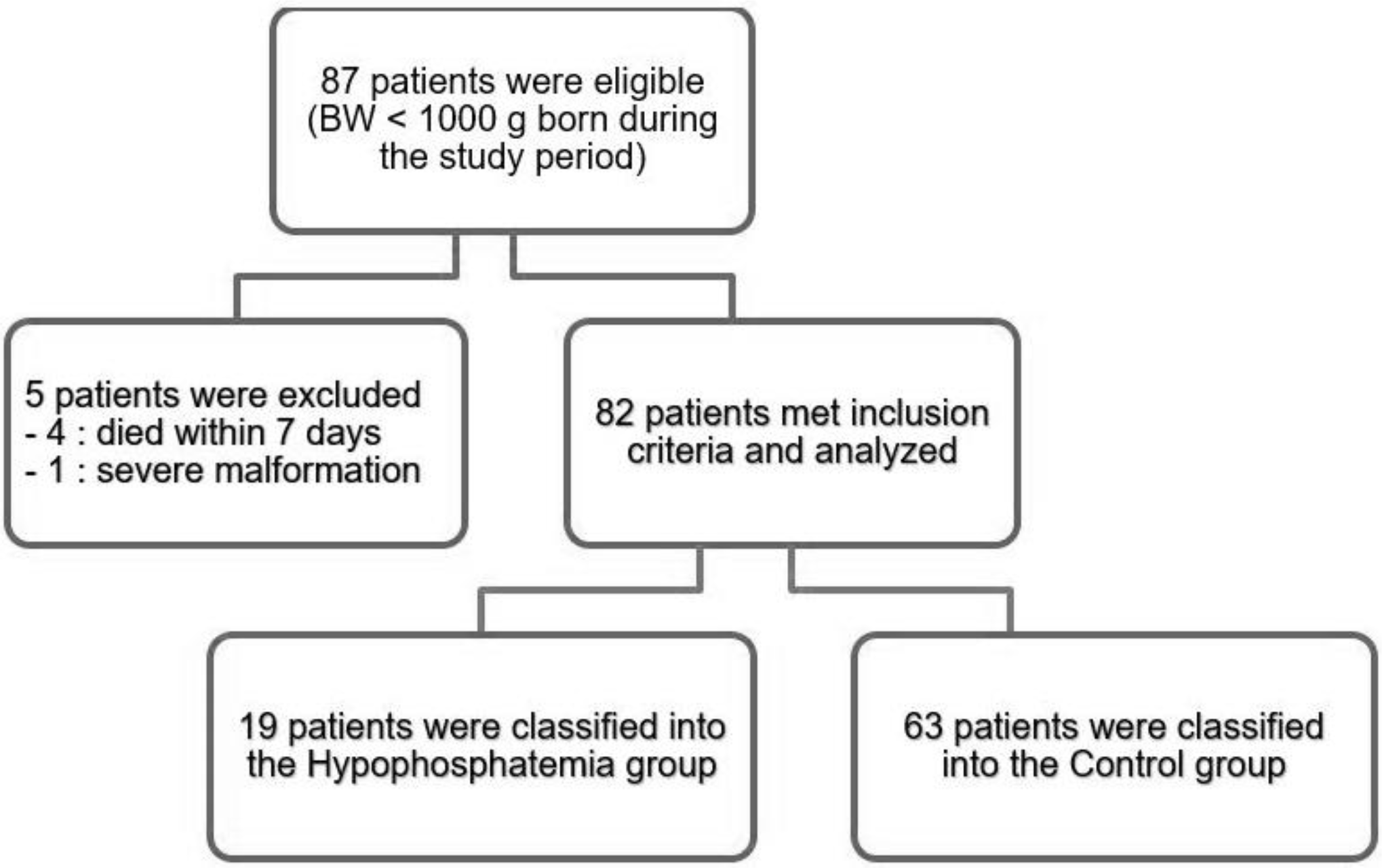
Medical records of 82 ELBWI were retrospectively reviewed. Severe hypophosphatemia was defined as a serum phosphate level <2 mg/dL during the first week after birth.
Results
Nineteen ELBWI (23.2%) experienced severe hypophosphatemia. The supplementation of phosphorus was started significantly later in the hypophosphatemia group compared to that in the control group (P=0.036). Small for gestational age infants (SGAI) (P=0.006) and bronchopulmonary dysplasia (P=0.016) were more prevalent in the hypophosphatemia group compared to that in the control group.
Go to : 
REFERENCES
1). Valentine CJ., Fernandez S., Rogers LK., Gulati P., Hayes J., Lore P, et al. Early amino-acid administration improves preterm infant weight. J Perinatol. 2009. 29:428–32.

2). Koletzko B., Goulet O., Hunt J., Krohn K., Shamir R. Guidelines on paediatric parenteral nutrition of the european society of paediatric gastroenterology, hepatology and nutrition (ESPGHAN) and the European Society for Clinical Nutrition and Metabolism (ESPEN), supported by the European Society of Paediatric Research (ESPR). J Pediatr Gastroenterol Nutr. 2005. 41(Suppl 2):S1–87.
3). Thureen PJ., Melara D., Fennessey PV., Hay WW Jr. Effect of low versus high intravenous amino acid intake on very low birth weight infants in the early neonatal period. Pediatr Res. 2003. 53:24–32.

4). Brener Dik PH., Galletti MF., Fernández Jonusas SA., Alonso G., Mariani GL., Fustiñana CA. Early hypophosphatemia in preterm infants receiving aggressive parenteral nutrition. J Perinatol. 2015. 35:712–5.

5). Skipper A. Refeeding syndrome or refeeding hypophosphatemia: a systematic review of cases. Nutr Clin Pract. 2012. 27:34–40.
6). Ross JR., Finch C., Ebeling M., Taylor SN. Refeeding syndrome in very-low-birth-weight intrauterine growth-restricted neonates. J Perinatol. 2013. 33:717–20.

7). Ichikawa G., Watabe Y., Suzumura H., Sairenchi T., Muto T., Arisaka O. Hypophosphatemia in small for gestational age extremely low birth weight infants receiving parenteral nutrition in the first week after birth. J Pediatr Endocrinol Metab. 2012. 25:317–21.

8). Boubred F., Herlenius E., Bartocci M., Jonsson B., Vanpee M. Extremely preterm infants who are small for gestational age have a high risk of early hypophosphatemia and hypokalemia. Acta Paediatr. 2015. 104:1077–83.

9). Moe K., Beck-Nielsen SS., Lando A., Greisen G., Zachariassen G. Administering different levels of parenteral phosphate and amino acids did not influence growth in extremely preterm infants. Acta Paediatr. 2015. 104:894–9.
10). Jobe AH., Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001. 163:1723–9.

11). Neu J. Necrotizing enterocolitis: the search for a unifying pathogenic theory leading to prevention. Pediatr Clin North Am. 1996. 43:409–32.
12). Papile LA., Burstein J., Burstein R., Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 1978. 92:529–34.

13). International Committee for the Classification of Retinopathy of Prematurity. The international classification of retinopathy of prematurity revisited. Arch Ophthalmol. 2005. 123:991–9.
14). Fenton TR. A new growth chart for preterm babies: Babson and Benda's chart updated with recent data and a new format. BMC Pediatr. 2003. 3:13.

15). Team RC. R: a language and environment for the statistical computing. [accessed on 16 Oct 2017]. Available at. https://cran.r-project.org/mirrors.html.
16). Bonsante F., Iacobelli S., Latorre G., Rigo J., De Felice C., Robillard PY, et al. Initial amino acid intake influences phosphorus and calcium homeostasis in preterm infants—it is time to change the composition of the early parenteral nutrition. PLoS One. 2013. 8:e72880.
17). Mizumoto H., Mikami M., Oda H., Hata D. Refeeding syndrome in a small-for-dates micro-preemie receiving early parenteral nutrition. Pediatr Int. 2012. 54:715–7.

18). Marik PE., Bedigian MK. Refeeding hypophosphatemia in critically ill patients in an intensive care unit. A prospective study. Arch Surg. 1996. 131:1043–7.
19). Dreyfus L., Fischer Fumeaux CJ., Remontet L., Essomo Megnier Mbo Owono MC., Laborie S., Maucort-Boulch D, et al. Low phosphatemia in extremely low birth weight neonates: a risk factor for hyperglycemia? Clin Nutr. 2016. 35:1059–65.

20). Zhao Y., Li Z., Shi Y., Cao G., Meng F., Zhu W, et al. Effect of hypophosphatemia on the withdrawal of mechanical ventilation in patients with acute exacerbations of chronic obstructive pulmonary disease. Biomed Rep. 2016. 4:413–6.

21). Kilic O., Demirkol D., Ucsel R., Citak A., Karabocuoglu M. Hypophosphatemia and its clinical implications in critically ill children: a retrospective study. J Crit Care. 2012. 27:474–9.

22). Gustavsson CG., Eriksson L. Acute respiratory failure in anorexia nervosa with hypophosphataemia. J Intern Med. 1989. 225:63–4.

23). Hasselstrøm L., Wimberley PD., Nielsen VG. Hypophosphatemia and acute respiratory failure in a diabetic patient. Intensive Care Med. 1986. 12:429–31.

24). Liu PY., Jeng CY. Severe hypophosphatemia in a patient with diabetic ketoacidosis and acute respiratory failure. J Chin Med Assoc. 2004. 67:355–9.
Go to : 
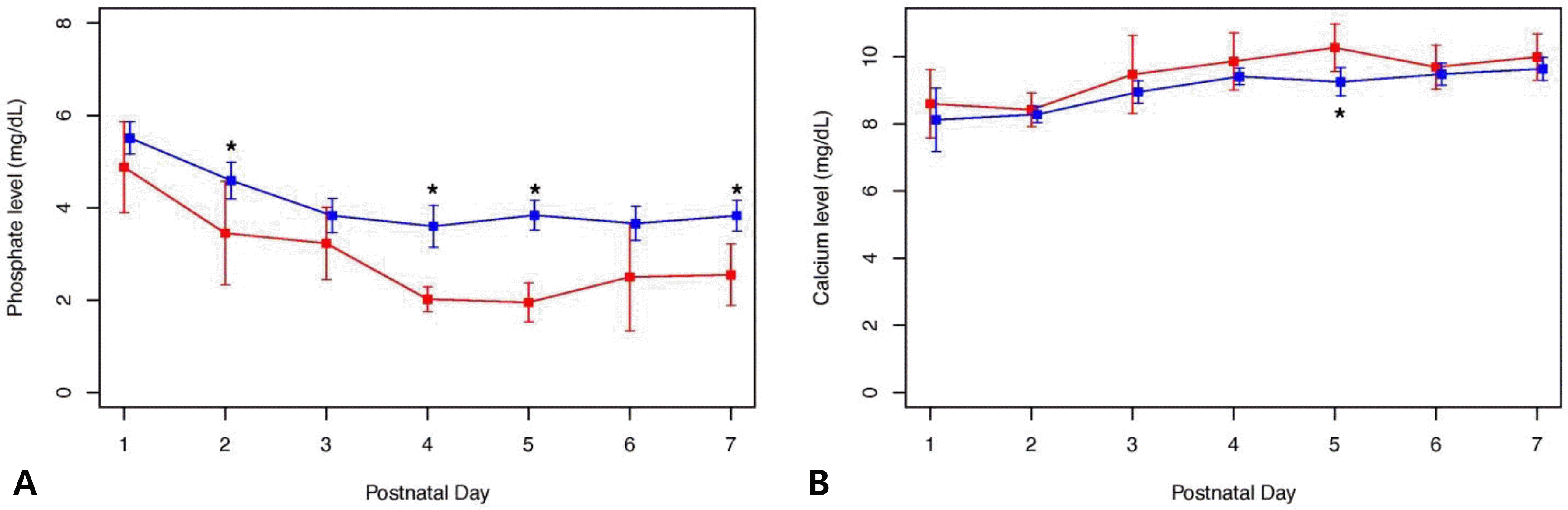 | Fig. 2The serum phosphate and calcium levels during the first week after birth are shown for the hypophosphate mia and control groups. (A) Mean phosphate levels during the first week after birth: there was no group difference in phosphate level on day 1, but there were significant group differences on day 2 (3.5±1.9 vs. 4.6±1.34 mg/dL, P=0.015), day 4 (1.9 [1.8; 2.2] vs. 3.5 [2.9; 4.3] mg/dL, P<0.001), day 5 (2.0±10.6 vs. 3.8±1.0 mg/dL, P<0.001), and day 7 (2.5±1.2 vs. 3.8±1.0 mg/dL, P<0.001). (B) Mean calcium levels during the first week after birth: on day 5 (10.0 [9.7; 10.8] vs. 9.2 [8.6; 9.9] mg/dL, P=0.012), there was a significant group difference in calcium levels. |
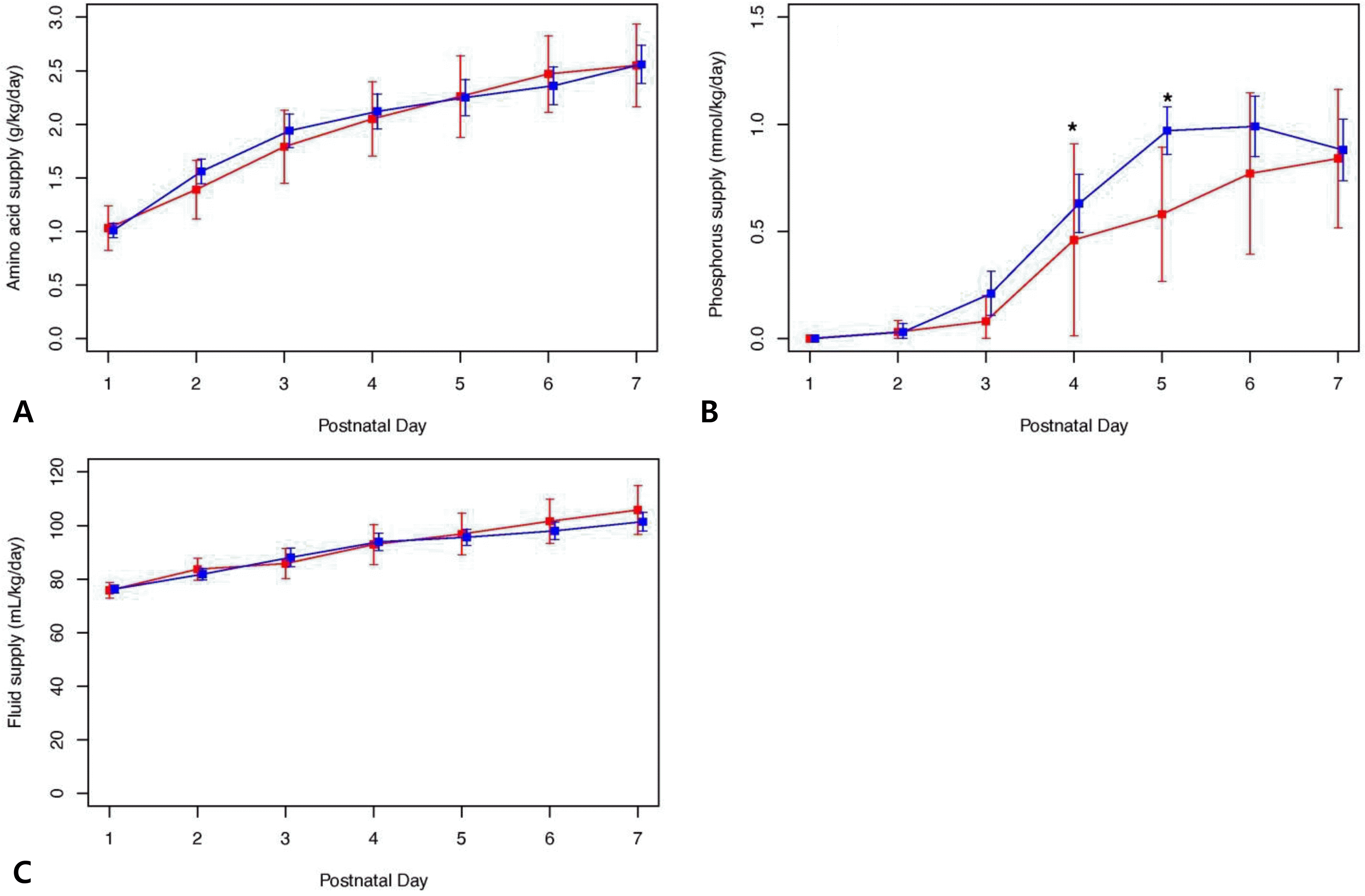 | Fig. 3The supplementation of amino acid, phosphorus, and fluid during the first week after birth is shown for the hypophosphatemia and control groups. (A) Amino acids supplementation during the first week after birth: there was no group difference in the amount of amino acid administration during the first week after birth. (B) Phosphorus supplementation during the first week after birth: there were significant group differences in the amount of phosphorus administered on day 4 (0.0 [0.0; 0.5] vs. 0.6 [0.0; 1.0] mmol/kg/day, P=0.37) and day 5 (0.5 [0.0; 1.0] vs. 1.0 [0.8; 1.0] mmol/kg/day, P=0.017). (C) Fluid supplementation during the first week after birth: there was no group difference in the amount of fluid administration. |
Table 1.
Patient Characteristics
Table 2.
Clinical Outcomes in the Neonatal Intensive Care Unit




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download