1. Rottenstreich A, Kleinstern G, Spectre G, Da'as N, Ziv E, Kalish Y. Thromboembolic events following splenectomy: risk factors, prevention, management and outcomes. World J Surg. 2018; 42:675–681. PMID:
28808782.

2. Ruiz-Tovar J, Priego P. Portal vein thrombosis after splenic and pancreatic surgery. Adv Exp Med Biol. 2017; 906:241–251. PMID:
27638624.

3. Kimmig LM, Palevsky HI. Review of the association between splenectomy and chronic thromboembolic pulmonary hypertension. Ann Am Thorac Soc. 2016; 13:945–954. PMID:
27058013.

4. Holdsworth RJ, Irving AD, Cuschieri A. Postsplenectomy sepsis and its mortality rate: actual versus perceived risks. Br J Surg. 1991; 78:1031–1038. PMID:
1933181.

5. Bisharat N, Omari H, Lavi I, Raz R. Risk of infection and death among post-splenectomy patients. J Infect. 2001; 43:182–186. PMID:
11798256.

6. de'Angelis N, Abdalla S, Lizzi V, Esposito F, Genova P, Roy L, et al. Incidence and predictors of portal and splenic vein thrombosis after pure laparoscopic splenectomy. Surgery. 2017; 162:1219–1230. PMID:
28919051.
7. Di Sabatino A, Carsetti R, Corazza GR. Post-splenectomy and hyposplenic states. Lancet. 2011; 378:86–97. PMID:
21474172.

8. Uranues S, Grossman D, Ludwig L, Bergamaschi R. Laparoscopic partial splenectomy. Surg Endosc. 2007; 21:57–60. PMID:
17031738.

9. Van Wyck DB, Witte MH, Witte CL, Thies AC Jr. Critical splenic mass for survival from experimental pneumococcemia. J Surg Res. 1980; 28:14–17. PMID:
7359904.

10. Liu DL, Xia S, Xu W, Ye Q, Gao Y, Qian J. Anatomy of vasculature of 850 spleen specimens and its application in partial splenectomy. Surgery. 1996; 119:27–33. PMID:
8560382.

11. Christo MC. Partial regulated splenectomies. Preliminary note on the first 3 cases operated on. Hospital (Rio J). 1959; 56:645–650. PMID:
13810141.
12. Morgenstern L, Shapiro SJ. Partial splenectomy for nonparasitic splenic cysts. Am J Surg. 1980; 139:278–281. PMID:
7356115.

13. Poulin EC, Thibault C, DesCôteaux JG, Côté G. Partial laparoscopic splenectomy for trauma: technique and case report. Surg Laparosc Endosc. 1995; 5:306–310. PMID:
7551284.
14. Resende V, Petroianu A. Functions of the splenic remnant after subtotal splenectomy for treatment of severe splenic injuries. Am J Surg. 2003; 185:311–315. PMID:
12657380.

15. Wang L, Xu J, Li F, Zhan H, Liu H, Chen W, et al. Partial splenectomy is superior to total splenectomy for selected patients with hemangiomas or cysts. World J Surg. 2017; 41:1281–1286. PMID:
28058472.

16. Petroianu A. Subtotal splenectomy for the treatment of chronic lymphocytic leukemia. Ann Hematol. 2003; 82:708–709. PMID:
12904901.

17. Vasilescu C, Stanciulea O, Tudor S, Stanescu D, Colita A, Stoia R, et al. Laparoscopic subtotal splenectomy in hereditary spherocytosis: to preserve the upper or the lower pole of the spleen? Surg Endosc. 2006; 20:748–752. PMID:
16544076.
18. Vasilescu C, Tudor S, Popa M, Tiron A, Lupescu I. Robotic partial splenectomy for hydatid cyst of the spleen. Langenbecks Arch Surg. 2010; 395:1169–1174. PMID:
20393743.

19. Palmieri I, Natale E, Crafa F, Cavallaro A, Mingazzini PL. Epithelial splenic cysts. Anticancer Res. 2005; 25:515–521. PMID:
15816621.
20. Uranues S, Alimoglu O. Laparoscopic surgery of the spleen. Surg Clin North Am. 2005; 85:75–90. PMID:
15619530.

21. Hong TH, Lee SK, You YK, Kim JG. Single-port laparoscopic partial splenectomy: a case report. Surg Laparosc Endosc Percutan Tech. 2010; 20:e164–e166. PMID:
20975492.
22. Giulianotti PC, Buchs NC, Addeo P, Ayloo S, Bianco FM. Robot-assisted partial and total splenectomy. Int J Med Robot. 2011; 7:482–488. PMID:
21954176.

23. Balaphas A, Buchs NC, Meyer J, Hagen ME, Morel P. Partial splenectomy in the era of minimally invasive surgery: the current laparoscopic and robotic experiences. Surg Endosc. 2015; 29:3618–3627. PMID:
25740639.

24. Bouchet A. New glimpses on the structure and vascularization of the greater omentum. Arch Anat Histol Embryol. 1962; 45:1–31. PMID:
14014337.
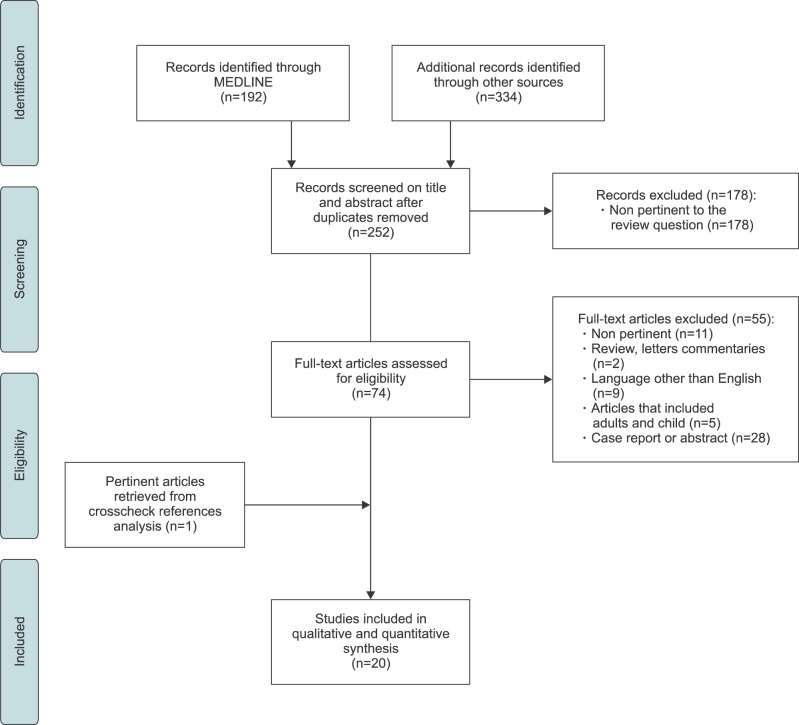
25. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Ann Intern Med. 2009; 151:W65–W94. PMID:
19622512.

26. Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008; 336:924–926. PMID:
18436948.

27. Li H, Wei Y, Peng B, Li B, Liu F. Feasibility and safety of emergency laparoscopic partial splenectomy: a retrospective analysis. Medicine (Baltimore). 2017; 96:e6450. PMID:
28422834.
28. Cai H, An Y, Wu D, Chen X, Zhang Y, Zhu F, et al. Laparoscopic partial splenectomy: a preferred method for select patients. J Laparoendosc Adv Surg Tech A. 2016; 26:1010–1014. PMID:
27467747.

29. Lee SH, Lee JS, Yoon YC, Hong TH. Role of laparoscopic partial splenectomy for tumorous lesions of the spleen. J Gastrointest Surg. 2015; 19:1052–1058. PMID:
25835566.

30. Costi R, Castro Ruiz C, Zarzavadjian le, Scerrati D, Santi C, Violi V. Spleen hydatidosis treated by hemi-splenectomy: a low-morbidity, cost-effective management by a recently improved surgical technique. Int J Surg. 2015; 20:41–45. PMID:
26074292.

31. Han XL, Zhao YP, Chen G, Wu WM, Dai MH. Laparoscopic partial splenectomy for splenic hemangioma: experience of a single center in six cases. Chin Med J (Engl). 2015; 128:694–697. PMID:
25698206.
32. de la Villeon B, Zarzavadjian Le, Vuarnesson H, Munoz Bongrand N, Halimi B, Sarfati E, et al. Laparoscopic partial splenectomy: a technical tip. Surg Endosc. 2015; 29:94–99. PMID:
24962862.

33. Wang X, Wang M, Zhang H, Peng B. Laparoscopic partial splenectomy is safe and effective in patients with focal benign splenic lesion. Surg Endosc. 2014; 28:3273–3278. PMID:
24939157.

34. Dudi-Venkata NN, Houli N, Weinberg L, Nikfarjam M. Laparoscopic partial splenectomy performed by monopolar saline-cooled radiofrequency coagulation. J Laparoendosc Adv Surg Tech A. 2014; 24:502–505. PMID:
24919036.

35. Paudel GR, Agarwal R, Pathania OP, Agrawal CS. Partial splenectomy for epithelial (epidermoid) splenic cysts: report of two case. JNMA J Nepal Med Assoc. 2013; 52:391–394. PMID:
24362667.

36. Liese J, Kohler S, Moench C, Bechstein WO, Ulrich F. Partial spleen resection with a radiofrequency needle device--a pilot study. Langenbecks Arch Surg. 2013; 398:449–454. PMID:
23385735.

37. Karadayi K, Turan M, Sen M. A new technique for partial splenectomy with radiofrequency technology. Surg Laparosc Endosc Percutan Tech. 2011; 21:358–361. PMID:
22002274.
38. Patrzyk M, Glitsch A, Hoene A, von Bernstorff W, Heidecke CD. Laparoscopic partial splenectomy using a detachable clamp with and without partial splenic embolisation. Langenbecks Arch Surg. 2011; 396:397–402. PMID:
20683622.

39. Szczepanik AB, Meissner AJ. Partial splenectomy in the management of nonparasitic splenic cysts. World J Surg. 2009; 33:852–856. PMID:
19172349.

40. Petroianu A, Cabezas-Andrade MA, Berindoague Neto R. Laparoscopic subtotal splenectomy. Surg Laparosc Endosc Percutan Tech. 2008; 18:94–97. PMID:
18287996.

41. Wu SC, Wang CC, Yong CC. Partial splenectomy for benign splenic cysts with the aid of a Lin clamp: technical note. World J Surg. 2007; 31:2144–2147. PMID:
17896130.

42. Jiao LR, Tierris I, Ayav A, Milicevic M, Pellicci R, Navarra G, et al. A new technique for spleen preservation with radiofrequency. Surgery. 2006; 140:464–466. PMID:
16934610.

43. Balshem H, Helfand M, Schünemann HJ, Oxman AD, Kunz R, Brozek J, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011; 64:401–406. PMID:
21208779.

44. Hong TH, You YK, Lee KH. Transumbilical single-port laparoscopic cholecystectomy: scarless cholecystectomy. Surg Endosc. 2009; 23:1393–1397. PMID:
19118436.
45. Lee YS, Kim JH, Moon EJ, Kim JJ, Lee KH, Oh SJ, et al. Comparative study on surgical outcomes and operative costs of transumbilical single-port laparoscopic appendectomy versus conventional laparoscopic appendectomy in adult patients. Surg Laparosc Endosc Percutan Tech. 2009; 19:493–496. PMID:
20027094.

46. Vasilescu C, Stanciulea O, Tudor S. Laparoscopic versus robotic subtotal splenectomy in hereditary spherocytosis. Potential advantages and limits of an expensive approach. Surg Endosc. 2012; 26:2802–2809. PMID:
22476842.

47. Berelavichus SV, Smirnov AV, Ionkin DA, Kriger AG, Dugarova RS. Robot-assisted and laparoscopic partial splenectomy for nonparasitic cysts. Khirurgiia (Mosk). 2015; (7):41–48.

48. Habib NA, Spalding D, Navarra G, Nicholls J. How we do a bloodless partial splenectomy. Am J Surg. 2003; 186:164–166. PMID:
12885611.

49. Itamoto T, Fukuda S, Tashiro H, Ohdan H, Asahara T. Radiofrequency-assisted partial splenectomy with a new and simple device. Am J Surg. 2006; 192:252–254. PMID:
16860640.

50. Gumbs AA, Bouhanna P, Bar-Zakai B, Briennon X, Gayet B. Laparoscopic partial splenectomy using radiofrequency ablation. J Laparoendosc Adv Surg Tech A. 2008; 18:611–613. PMID:
18721016.

51. Stacey MJ, Rampaul RS, Rengaragan A, Duffy JP, Macmillan RD. Use of FloSeal matrix hemostatic agent in partial splenectomy after penetrating trauma. J Trauma. 2008; 64:507–508. PMID:
17413522.

52. Mignon F, Brouzes S, Breitel DL, Bastie JN, Poirier H, Legendre C, et al. Preoperative selective embolization allowing a partial splenectomy for splenic hamartoma. Ann Chir. 2003; 128:112–116. PMID:
12657551.
53. Guan YS, Hu Y. Clinical application of partial splenic embolization. ScientificWorldJournal. 2014; 2014:961345. PMID:
25538966.

54. Kimura F, Ito H, Shimizu H, Togawa A, Otsuka M, Yoshidome H, et al. Partial splenic embolization for the treatment of hereditary spherocytosis. AJR Am J Roentgenol. 2003; 181:1021–1024. PMID:
14500222.

55. Coccolini F, Montori G, Catena F, Kluger Y, Biffl W, Moore EE, et al. Splenic trauma: WSES classification and guidelines for adult and pediatric patients. World J Emerg Surg. 2017; 12:40. PMID:
28828034.
56. Liu S, Lei J, Zeng Z, Zhang Y. Management of traumatic splenic rupture in adults: a single center's experience in mainland China. Hepatogastroenterology. 2014; 61:966–971. PMID:
26158150.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download