Abstract
We investigated the inhibitory effect of escitalopram, a selective serotonin reuptake inhibitor (SSRI), on voltage-dependent K+ (Kv) channels in freshly separated from rabbit coronary arterial smooth muscle cells. The application of escitalopram rapidly inhibited vascular Kv channels. Kv currents were progressively inhibited by an increase in the concentrations of escitalopram, suggesting that escitalopram inhibited vascular Kv currents in a concentration-dependent manner. The IC50 value and Hill coefficient for escitalopram-induced inhibition of Kv channels were 9.54±1.33 µM and 0.75±0.10, respectively. Addition of escitalopram did not alter the steady-state activation and inactivation curves, suggesting that the voltage sensors of the channels were not affected. Pretreatment with inhibitors of Kv1.5 and/or Kv2.1 did not affect the inhibitory action of escitalopram on vascular Kv channels. From these results, we concluded that escitalopram decreased the vascular Kv current in a concentration-dependent manner, independent of serotonin reuptake inhibition.
Selective serotonin reuptake inhibitors (SSRIs) are the most commonly used antidepressants and widely prescribed in the treatment of major depression and anxiety disorders. To date, numerous SSRIs such as citalopram, fluvoxamine, fluoxetine, paroxetine, sertraline, and dapoxetine have been developed. Escitalopram, a representative SSRI, is obtained from, citalopram [1]. The antidepressant effect of escitalopram is markedly larger than citalopram and other SSRIs [2]. Although escitalopram has been known to be relatively safe and effective in the treatment of depressive and anxiety disorders, some side effects of escitalopram are inevitable, such as decreased libido, delayed ejaculation, and anorgasmia [3]. The effects of escitalopram on ion channels have been investigated; escitalopram has been reported to inhibit ether-a-go-go-related gene (hERG) channels [4]. However, the study for the effects of escitalopram on vascular ion channels, specifically voltage-dependent K+ (Kv) channels, has been neglected.
K+ channels are ubiquitously expressed in most cell types and are involved in the control of many physiological functions such as cardiac and nerve excitability, neurotransmitter release, and muscle contraction [56]. Among the various types of K+ channels, Kv channel are regarded as one of the most crucial types of K+ channel, particularly, in vascular smooth muscle cells. Kv channels in arterial smooth muscle cells are mainly involved in determining resting membrane potential and consequently, vascular tone [78910]. In fact, the inhibition of Kv channels induced strong membrane depolarization in several arteries [1112]. In addition, alterations in Kv channel subtypes have been found in several circulatory and metabolic diseases. For example, mutations in the gene encoding Kv1.3 were identified in patients with acute coronary insufficiency [13]. The Kv2.1 channel also has an important role in mitigating type 2 diabetes [14]. Therefore, understanding the functions of Kv channels is essential to reverse arterial diseases.
Considering the efficacy of escitalopram in treating patients with depression and the pathophysiological importance of vascular Kv channels, the inhibitory effect of escitalopram on vascular Kv channels should be clearly identified.
In our present study, we demonstrated the effect of escitalopram on vascular Kv channels using freshly separated from arterial smooth muscle cells. Our results clearly demonstrated that escitalopram decreased vascular Kv channels in a dose-dependent manner without the involvement of serotonin reuptake inhibition.
Male New Zealand White rabbits were anesthetized by simultaneous injections of sodium pentobarbital (50 mg/kg) and heparin (100 U/kg) into the ear vein. All animal experiments procedures were approved by the Committee for Animal Experiments of Kangwon National University. The second branches of coronary arteries were carefully isolated from the heart. The connective tissues attached to the arteries were removed in normal Tyrode's solution under a stereomicroscope. The endothelium was also removed by passing an air bubble through the lumen of arteries. The arteries were washed in Ca2+-free normal Tyrode's solution, and transferred into an enzyme mixture solution containing papain (1.0 mg/ml), bovine serum albumin (BSA, 1.0 mg/ml), and dithiothreitol (DTT, 1.0 mg/ml), After incubation at 37℃ for 25 min, the arteries were transferred into another enzyme mixture solution containing collagenase (2.8 mg/ml), BSA (1.0 mg/ml), and DTT (1.0 mg/ml), and then incubated at 37℃ for 18~19 min. After the enzymatic procedures, single cells were obtained by gentle mechanical agitation in Kraft-Brühe (KB) solution with a fire-polished Pasteur glass pipette. Cells were stored at 4℃ and used within 6 h.
The composition (in mM) of normal Tyrode's solution was: NaCl, 135; KCl, 5.4; CaCl2, 1.8; MgCl2, 1; NaH2PO4, 0.33; HEPES, 5; glucose, 15; adjusted to pH 7.4 with NaOH. The composition (in mM) of KB solution was: KOH, 70; KCl, 55; L-glutamate, 50; KH2PO4, 20; taurine, 20; MgCl2, 3; glucose, 20; HEPES, 10; EGTA, 0.5; adjusted to pH 7.3 with KOH. The composition (in mM) of the pipette solution for the recording of Kv channels was: K-aspartate, 110; KCl, 25; NaCl, 5; MgCl2, 1; Mg-ATP, 4; EGTA, 10; HEPES, 10; adjusted to pH 7.2 with KOH. Escitalopram, DPO-1, and guangxitoxin were purchased from Tocris Cookson (Ellisville, MO, USA) and dissolved in dimethyl sulfoxide (DMSO). The final concentration of DMSO was less than 0.1%, a level at which DMSO had no significant effect on the recorded Kv current.
Kv currents recorded in this study were induced using an EPC-8 amplifier (Medical System Corp., Darmstadt, Germany), NI-DAQ-7 (National Instruments, CA, USA), and PatchPro software. The glass pipettes for the internal solution were made by a PP-830 vertical puller (Narishige Scientific Instrument Laboratory, Tokyo, Japan). When filled with the internal solution, the resistance of the patch pipette was 3~4 MΩ. The average cell capacitance was 12.67±0.72 pF (n=21).
Data analysis was conducted with Origin 7.5 software (Microcal Software, Inc., Northampton, MA, USA). A first-order blocking scheme was applied for the analysis of drug-channel interaction kinetics [1516]. The values for the half-maximal inhibitory (IC50) and the Hill coefficient (n) were obtained from concentration-dependent data and fitted to the following Hill equation:
where ƒ is the fractional inhibition of current (ƒ=1−Idrug/Icontrol) at the test potential, and [D] is the drug concentration.
A steady-state activation curve was obtained from the tail currents. Tail currents were induced by returning to a potential of −40 mV after depolarization of the cell from −80 to +60 mV in 10 mV increments. All recorded tail currents were normalized to the maximal tail current. The activation curve was fitted with the Boltzmann equation given below:
where V1/2 is the half-point of activation, V is the test potential, and k is the slope factor.
The steady-state inactivation curves were acquired using a two-pulse voltage protocol: preconditioning pulses of 7 s were applied at potentials ranging from −80 to +30 mV in the absence and presence of drugs, and then returned to +40 mV. Steady-state inactivation curves were fitted with another Boltzmann equation as follows:
where V1/2 is the potential of the half-maximal of inactivation point, V is the preconditioning potential, and k is the slope value.
The results are presented as mean±standard error of the mean (S.E.M.), and a Student's t-test was used to estimate statistical significance. P values greater than 0.05 were considered statistically significant.
The inhibitory effects of escitalopram on Kv currents were detected using freshly separated from coronary vascular smooth muscle cells. Kv currents were induced by applying incremental depolarizing pulses from −80 to +60 mV at a holding voltage of −80 mV. The activation of other K+ channels, such as ATP-dependent K+ (KATP) and large-conductance Ca2+-activated K+ (BKCa) channels, were inhibited by the inclusion of 4 mM ATP and 10 mM EGTA to inhibit the KATP and BKCa channels, respectively. In addition, involvement with inwardly rectifying K+ (Kir) channels was excluded by the use of coronary smooth muscle cells separated from the second branch of the arteries, as the Kir channel has only been expressed in small-diameter coronary arteries [17]. Fig. 1A shows the application of voltage-step induced Kv currents, which rapidly peaked and showed slow and partial inactivation. The Kv current was inhibited by 10 µM escitalopram within 2 min (Fig. 1B). This inhibition was partially washed out (approximately 50%, data not shown). The current-voltage (I−V) relationship is shown in Fig. 1C. At +40 mV, application of 10 µM escitalopram reduced Kv current by 47.54%.
Various concentrations (0, 0.1, 0.3, 1, 3, 10, 30, and 100 µM) of escitalopram were applied to evaluate whether the inhibition of Kv channels by escitalopram was concentration-dependent. Kv currents were induced by one-step depolarizing pulses of +60 mV from a holding voltage of −80 mV. As shown in Fig. 2A, the Kv currents were progressively decreased by an increase in escitalopram concentration. These results suggested that the escitalopram-induced inhibition of Kv channels was concentration-dependent. From analysis of the steady-state inhibition, we obtained an IC50 value of 9.53±1.33 µM and a slope factor (Hill coefficient) of 0.75±0.10 by fitting the dose-dependent results using the Hill equation (Fig. 2B).
To confirm whether the escitalopram-induced inhibition of Kv current was caused by shifting of activation and/or inactivation curves, we tested the effect of escitalopram on the steady-state activation and inactivation curves. The steady-state activation curves were acquired from tail currents and fitted with a Boltzmann function (see Materials and Methods). As shown in Fig. 3A, steady-state activation curve for Kv channels was not affected by the addition of 10 µM escitalopram. The activation potential for the half-maximum (V1/2) and slope value (k) were −3.47±0.58 mV and 14.09±0.47, respectively, in the absence of escitalopram and −3.52±1.05 mV and 15.18±0.79, respectively, in the presence of escitalopram.
The steady-state inactivation curve was acquired using a two-pulse voltage protocol and was fitted to another Boltzmann function (see Materials and Methods). Similar to the activation curve, addition of 10 µM escitalopram did not affect the steady-state inactivation curve of Kv channels (Fig. 3B). The half-maximal inactivation potential (V1/2) and slope value (k) were −38.68±1.05 mV and 7.46±0.54, respectively, in the absence of escitalopram and −37.36±1.54 mV and 8.64±1.44, respectively, in the presence of escitalopram.
To evaluate the involvement of Kv1.5 and/or Kv2.1 subtypes in escitalopram-induced inhibition, we determined the inhibitory effect of escitalopram on Kv currents in the presence of a Kv1.5 inhibitor (1 µM DPO-1) and/or a Kv2.1 inhibitor (30 nM guangxitoxin). The currents were induced by applying a one-step depolarizing pulse to +60 mV from a holding voltage of −80 mV. As shown in Figs. 4A and B, the application of DPO-1 decreased Kv current, and addition of escitalopram induced further inhibition of Kv currents. However, the inhibitory effect of escitalopram was unaffected by DPO-1 pretreatment. Similar to the results of DPO-1, addition of guangxitoxin also induced further inhibition of Kv currents and additional application of the escitalopram induced further inhibition of Kv currents (Figs. 4C and D). In addition, pretreatment with DPO-1 and guangxitoxin reduced Kv currents; however, escitalopram additionally induced the inhibition of Kv currents (Figs. 4E and F). These results suggested that the main targets of escitalopram were not the subtypes Kv1.5 and Kv2.1.
In this study, we demonstrated the inhibitory effect of escitalopram on vascular Kv currents in coronary vascular smooth muscle cells from rabbits. Our results suggested that escitalopram inhibited Kv channels in a dose-dependent manner regardless of serotonin reuptake inhibition. In addition, we discovered that escitalopram did not affect the activation and inactivation curves of Kv channels, which suggested that escitalopram did not affect the gating properties of Kv channels.
According to our results, escitalopram inhibited Kv channels regardless of serotonin reuptake inhibition. First, the IC50 value for the escitalopram-induced inhibition of Kv channel was 9.53 µM, which was 4500 times larger than the IC50 value for escitalopram-induced serotonin reuptake inhibition (2.1 nM) [18]. The magnitude of this discrepancy also suggested that escitalopram-induced inhibition of Kv channels was independent of serotonin reuptake inhibition. Second, serotonin has been reported to inhibit vascular Kv channels, consequently inducing membrane depolarization and vasoconstriction [1219]. Therefore, increases in serotonin level induced by SSRIs could lead to an increase of blood pressure. However, our single cell experimental system was able to exclude the SSRI-induced increase of serotonin level in the bloodstream. Therefore, this body of evidence indicated that the escitalopram-induced inhibition of Kv channel might be through direct inhibition and not due to the inhibition of serotonin reuptake.
Selective serotonin reuptake inhibitors (SSRIs) are a member of anti-depressants that are recently used as first-line drugs in depressive patients. The therapeutic mechanism of SSRIs is to cause the inhibition of serotonin reuptake in the brain [20]. The SSRI escitalopram is the S-enantiomer of citalopram, which contains the same amounts of the S- and R-forms in a racemic mixture [1]. Since escitalopram exhibits more potent serotonin reuptake inhibition than that of citalopram, escitalopram shows better efficacy in the treatment of depression [2]. Although escitalopram contains only the S-enantiomer of citalopram, it shares many similar properties with citalopram. Recently, several studies suggested the effects of citalopram on ion channels. For example, citalopram inhibited the Kv1.3 and Kv1.5 subtypes in cell lines co-expressing Nav1.5, Kir2.1, Kv1.3, and Kv1.5 [21]. In addition, citalopram inhibited the Nav1.7 and Nav1.8 channels that are expressed in human embryonic kidney (HEK) 293 cells [22]. Citalopram also inhibited the delayed rectifier K+ current in mouse cortical neurons, mainly by inhibition of the Kv2.2 subtype rather than the Kv2.1 subtype [23]. These results were similar to ours, as blocking the Kv2.1 subtype did not alter the inhibitory effect of escitalopram on Kv channels (Fig. 4). Investigation into the Kv1.5 subtype expressed in Chinese hamster ovary (CHO) cells revealed that it was inhibited by citalopram in a state-, time-, and use-dependent manner [24]. Our results, however, showed that the inhibitory effect of escitalopram on Kv channels was not involved in activation of the Kv1.5 subtype. We could not specify the exact reasons; these differences might be due to the difference in cell systems expressing specific subtype vs native cells, or to different sensitivities of the specific Kv subtype to the two drugs. To date, limited studies are available regarding the effects of escitalopram on ion channels. Therefore, more studies on the effects of escitalopram on several ion channels should be performed extensively to confirm the safety and efficacy of the clinical usage of escitalopram.
Several types of K+ channels, including KATP, BKCa, Kv, and Kir channels, are known to be expressed in vascular smooth muscle cells [8]. Among the different types, Kv channels are recognized as one of the most important channels in deciding resting membrane potential and consequently vascular tone [78910]. Therefore, a clear understanding of the effects of drugs on vascular Kv channels is necessary to avoid the misinterpretation of clinical and experimental data. To date, many drugs have been shown to inhibit arterial Kv channels regardless of their own intended functions. Regarding the effects of SSRIs on Kv channels, we recently reported that sertraline inhibited arterial Kv channels independently of serotonin reuptake inhibition [25]. This study suggested that sertraline changed the steady-state inactivation curves in a negative potential. Therefore, we concluded that sertraline directly bound to channels in the inactivated states. Fluvoxamine, another SSRI, has also been shown to inhibit arterial Kv channels irrespective of serotonin reuptake inhibition. Similar to the results with sertraline, fluvoxamine also inhibited vascular Kv channels by changing the steady-state inactivation curves in a negative potential [26]. In our present study, we exhibited the inhibitory effect of escitalopram on arterial Kv channels. Our results should be considered in conjunction with previous studies when using escitalopram for experimental or clinical purposes.
To date, many Kv subtypes have been detected in arterial smooth muscle, including, Kv1.1, Kv1.2, Kv1.4, Kv1.5, Kv2.1, and Kv 9.3 [2728]. These studies were mainly performed using mouse or rat samples, rather than rabbits. Therefore, the identification of the exact Kv subtypes in rabbit arterial smooth muscle is uncertain. However, most studies of vascular Kv subtypes have agreed that Kv1.5 and Kv2.1 subtypes are commonly expressed in vascular smooth muscle. For this reason, we tested the involvement of Kv1.5 and/or Kv2.1 subtype in escitalopram-induced inhibition of Kv channels. Our results showed that pretreatment with a Kv1.5 and/or Kv2.1 inhibitor did not change the inhibitory effect of escitalopram on Kv channels, suggesting that the Kv1.5 and Kv2.1 subtypes were not related in the inhibitory effect of escitalopram. We could not identify the exact Kv subtypes involved in the escitalopram effect; therefore, additional studies are necessary regarding the specific Kv subtypes involved in the inhibitory effect of escitalopram on Kv channels using a Kv subtype expression system.
In summary, we identified the inhibitory effect of escitalopram on Kv channels. This inhibition was not involved in serotonin reuptake inhibition. Therefore, great caution should be considered when prescribing escitalopram as an anti-depressant drug in patients with cardiovascular diseases.
Figures and Tables
Fig. 1
Inhibition of voltage-dependent K+ (Kv) current by escitalopram.
Kv currents were recorded by 600-ms depolarizing step pulses from −80 to +60 mV in steps of 10 mV at a holding potential of −80 mV. Representative Kv current in control conditions (A) and in the presence of 10 µM escitalopram (B). (C) Current-voltage (I−V ) relationship at a steady-state Kv current in control conditions (○) and the presence of 10 µM escitalopram (●). n=7. *p<0.05.

Fig. 2
Concentration-dependent inhibition of Kv channels by escitalopram.
(A) Superimposed current traces obtained by a 600-ms one-step depolarizing pulse of +60 mV from a holding potential of −80 mV in the presence of 0, 0.1, 0.3, 1, 3, 10, 30, and 100 µM escitalopram. (B) Summary of the average concentration-dependent inhibition of Kv currents at steady-state. The percentage inhibition of Kv current is plotted against the various concentrations of escitalopram. All n=6.
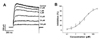
Fig. 3
Effects of escitalopram on steady-state activation and inactivation curves.
(A) Activation curves in the control conditions (○) and in the presence of 10 µM escitalopram (●). Activation curves were obtained by applying short depolarizing (20~50 ms) step pulses between −80 and +60 mV in 10-mV increments at a holding potential of −80 mV, then returned to −40 mV to elicit a tail current. n=7. (B) Inactivation curves in the control conditions (○) and in the presence of 10 µM escitalopram (●). The currents were recorded at a returning pulse of +40 mV after a 7-s preconditioning pulse between −80 and +30 mV. The induced currents were normalized to the peak amplitude of the test potential. n=6.
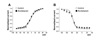
Fig. 4
Effect of Kv 1.5 and/or Kv 2.1 inhibitors on escitalopram-induced inhibition of Kv channels.
Current traces were acquired by one-step depolarizing pulses of +60 mV at a holding potential of −80 mV. (A) Representative current traces in the control conditions, in the presence of DPO-1, and in the presence of DPO-1+escitalopram. (B) A summary of these results in (A). n=6. *p<0.05. (C) Representative current traces in the control conditions, in the presence of guangxitoxin, and in the presence of guangxitoxin+escitalopram. (D) A summary of these results in (C). n=5. *p<0.05. (E) Representative current traces in the control conditions, in the presence of DPO-1+guangxitoxin, and in the presence of DPO-1+guangxitoxin+escitalopram. (F) A summary of these results in (E). n=4. *p<0.05.

ACKNOWLEDGEMENTS
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (Ministry of Education: 2016-R1D1A3B03930169) (Ministry of Science, ICT and Future Planning: 2012-M3A9C7050184, 2015-R1A4A1038666). This study was supported by 2016 Research Grant from Kangwon National University (No. 520160430).
Notes
Author contributions W.S.P. and H.S.K. designed the studies, H.S.K., H.L., H.W.K., S.E.S., and M.S.S. performed the experiments, J.R.A., I.W.C., G.C., and D.L. contributed materials and analysis tools, K.S.H., E.T.H., and S.H.H. contributed data analysis, H.S.K. and H.L. wrote the manuscript, W.S.P. wrote the final manuscript.
References
1. Garnock-Jones KP, McCormack PL. Escitalopram: a review of its use in the management of major depressive disorder in adults. CNS Drugs. 2010; 24:769–796.
2. Waugh J, Goa KL. Escitalopram: a review of its use in the management of major depressive and anxiety disorders. CNS Drugs. 2003; 17:343–362.
3. Bolton JM, Sareen J, Reiss JP. Genital anaesthesia persisting six years after sertraline discontinuation. J Sex Marital Ther. 2006; 32:327–330.
4. Chae YJ, Jeon JH, Lee HJ, Kim IB, Choi JS, Sung KW, Hahn SJ. Escitalopram block of hERG potassium channels. Naunyn Schmiedebergs Arch Pharmacol. 2014; 387:23–32.
5. Haddy FJ, Vanhoutte PM, Feletou M. Role of potassium in regulating blood flow and blood pressure. Am J Physiol Regul Integr Comp Physiol. 2006; 290:R546–R552.
6. Wulff H, Zhorov BS. K+ channel modulators for the treatment of neurological disorders and autoimmune diseases. Chem Rev. 2008; 108:1744–1773.
7. Ko EA, Han J, Jung ID, Park WS. Physiological roles of K+ channels in vascular smooth muscle cells. J Smooth Muscle Res. 2008; 44:65–81.
8. Nelson MT, Quayle JM. Physiological roles and properties of potassium channels in arterial smooth muscle. Am J Physiol. 1995; 268:C799–C822.
9. Lee HM, Hahn SJ, Choi BH. Blockade of Kv1.5 channels by the antidepressant drug sertraline. Korean J Physiol Pharmacol. 2016; 20:193–200.
10. Lee HM, Hahn SJ, Choi BH. Blockade of Kv1.5 by paroxetine, an antidepressant drug. Korean J Physiol Pharmacol. 2016; 20:75–82.
11. Shimoda LA, Sylvester JT, Sham JS. Inhibition of voltage-gated K+ current in rat intrapulmonary arterial myocytes by endothelin-1. Am J Physiol. 1998; 274:L842–L853.
12. Bae YM, Kim A, Kim J, Park SW, Kim TK, Lee YR, Kim B, Cho SI. Serotonin depolarizes the membrane potential in rat mesenteric artery myocytes by decreasing voltage-gated K+ currents. Biochem Biophys Res Commun. 2006; 347:468–476.
13. Xu R, Cao M, Wu X, Wang X, Ruan L, Quan X, Lü C, He W, Zhang C. Kv1.3 channels as a potential target for immunomodulation of CD4+ CD28null T cells in patients with acute coronary syndrome. Clin Immunol. 2012; 142:209–217.
14. Zhuang GQ, Wu W, Liu F, Ma JL, Luo YX, Xiao ZX, Liu Y, Wang W, He Y. SNAP-25(1-180) enhances insulin secretion by blocking Kv2.1 channels in rat pancreatic islet beta-cells. Biochem Biophys Res Commun. 2009; 379:812–816.
15. Snyders DJ, Yeola SW. Determinants of antiarrhythmic drug action. Electrostatic and hydrophobic components of block of the human cardiac hKv1.5 channel. Circ Res. 1995; 77:575–583.
16. Lee HA, Hyun SA, Park SG, Kim KS, Kim SJ. Comparison of electrophysiological effects of calcium channel blockers on cardiac repolarization. Korean J Physiol Pharmacol. 2016; 20:119–127.
17. Park WS, Ko JH, Kim N, Son YK, Kang SH, Warda M, Jung ID, Park YM, Han J. Increased inhibition of inward rectifier K+ channels by angiotensin II in small-diameter coronary artery of isoproterenol-induced hypertrophied model. Arterioscler Thromb Vasc Biol. 2007; 27:1768–1775.
18. Höschl C, Svestka J. Escitalopram for the treatment of major depression and anxiety disorders. Expert Rev Neurother. 2008; 8:537–552.
19. Sung DJ, Noh HJ, Kim JG, Park SW, Kim B, Cho H, Bae YM. Serotonin contracts the rat mesenteric artery by inhibiting 4-aminopyridine-sensitive Kv channels via the 5-HT2A receptor and Src tyrosine kinase. Exp Mol Med. 2013; 45:e67.
20. Neumeister A. Tryptophan depletion, serotonin, and depression: where do we stand? Psychopharmacol Bull. 2003; 37:99–115.
21. Fujii M, Hayashi K, Ohya S, Yamamura H, Imaizumi Y. New screening system for selective blockers of voltage-gated K+ channels using recombinant cell lines dying upon single action potential. J Pharmacol Sci. 2013; 123:147–158.
22. Thériault O, Poulin H, Beaulieu JM, Chahine M. Differential modulation of Nav1.7 and Nav1.8 channels by antidepressant drugs. Eur J Pharmacol. 2015; 764:395–403.
23. Zhan XQ, He YL, Yao JJ, Zhuang JL, Mei YA. The antidepressant citalopram inhibits delayed rectifier outward K+ current in mouse cortical neurons. J Neurosci Res. 2012; 90:324–336.
24. Lee HM, Hahn SJ, Choi BH. Open channel block of Kv1.5 currents by citalopram. Acta Pharmacol Sin. 2010; 31:429–435.
25. Kim HS, Li H, Kim HW, Shin SE, Choi IW, Firth AL, Bang H, Bae YM, Park WS. Selective serotonin reuptake inhibitor sertraline inhibits voltage-dependent K+ channels in rabbit coronary arterial smooth muscle cells. J Biosci. 2016; 41:659–666.
26. Hong DH, Li H, Kim HS, Kim HW, Shin SE, Jung WK, Na SH, Choi IW, Firth AL, Park WS, Kim DJ. The effects of the selective serotonin reuptake inhibitor fluvoxamine on voltage-dependent K+ channels in rabbit coronary arterial smooth muscle cells. Biol Pharm Bull. 2015; 38:1208–1213.
27. Xu C, Lu Y, Tang G, Wang R. Expression of voltage-dependent K+ channel genes in mesenteric artery smooth muscle cells. Am J Physiol. 1999; 277:G1055–G1063.
28. Yuan XJ, Wang J, Juhaszova M, Golovina VA, Rubin LJ. Molecular basis and function of voltage-gated K+ channels in pulmonary arterial smooth muscle cells. Am J Physiol. 1998; 274:L621–L635.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download