Abstract
Extramammary Paget disease (EMPD) is a rare cutaneous neoplasm. Perianal Paget disease (PPD) is a subset of EMPD manifesting perianal lesions. Two cases of PPD in Severance Hospital are described in this article. A 65-year-old female and 78-year-old male patients visited our institution because of an unhealed perianal skin lesion despite treatment for a long period with topical agents. PPD was diagnosed by skin biopsies in both cases, and the patients underwent surgical treatment. Clinical manifestations, preoperative work-ups, and surgical treatments including different reconstruction methods are described in detail. As only sporadic PPD cases have been reported and no standard treatment has been established, we hope that our experience could contribute to improving the diagnosis and treatment of PPD patients.
Extramammary Paget disease (EMPD) is a rare neoplastic condition occurring in the skin except the breast nipples. The most prevalent areas of EMPD are the vulva and the penoscrotal and perianal skin. Perianal Paget disease (PPD) is a subset of EMPD occurring in the perianal skin, and it is generally classified into 2 categories: primary PPD originating from the intraepidermal stem cells and secondary PPD associated with underlying malignancy in remote sites. Secondary PPD is more common and has a poorer prognosis. Because of its rarity, sporadic case reports have been published, and treatments have not been standardized yet [1]. In this article, we present two cases of primary PPD and describe the clinical manifestations, diagnostic workups, and surgical treatments.
A 65-year-old female patient was referred to Severance Hospital on March 15, 2016 after being diagnosed with PPD in another hospital. The perianal skin lesion of the patient was coincidentally found during her perineal skin preparation before she underwent an operation for urinary incontinence in December 2015. The patient complained of itching and stinging sensation in the perianal and perineal area. On physical examination, a 7 × 8-cm erythematous perianal skin lesion with slightly elevated patches, erosion, and brownish pigmentation was noted. The patient had applied imiquimod cream on the lesion for 2 months before visiting our institution, but no certain improvement was reported. Two pathology slides from the referring hospital were reviewed again. Paget cells in the epidermis were confirmed, and positive immunohistochemical(IHC) stain for cytokeratin 7 (CK7) and negative IHC stain for human melanoma black-45 (HMB-45) were also confirmed (Fig. 1). A chest posterioranterior radiograph and abdominoperineal computed tomography revealed no distant metastasis or pathologic lymphadenopathy. A colonoscopy revealed 3 polyps without any coexisting rectal malignancy. After evaluation, a multidisciplinary team including a colorectal surgeon, a dermatologist, a radiation oncologist, an oncologist, and a radiologist decided to perform wide excision and flap surgery for the skin defect.
On April 28, wide excision and flap surgery were performed. On the day before the operation, the dermatologist conducted mapping biopsies with local anesthesia to obtain adequate safety margins in advance (Fig. 2A). However, despite the repeated mapping biopsies, the excision line for the area around the vulva and the vaginal orifice could not be determined. Therefore, the excision line for that area was determined by intraoperative frozen section analysis.
On the day of the operation, the patient was placed in lithotomy position under general anesthesia. First, wide excision and frozen biopsies for resection margins were performed. Lateral skin incision was made along the previous mapping, and the anterior margin which was not determined by the mapping biopsies was excised four times until the resection margins were free from tumor in the frozen biopsies. As a result, the left labia minora and posterior vaginal wall were partially resected. The anoderm was excised up to the anorectal junction without anal sphincter injury, and the frozen biopsies for the proximal anoderm revealed tumor negative resection margins (Fig. 2B, C). After wide excision, laparoscopic-assisted loop ileostomy was performed for fecal diversion.
Then, the plastic surgery team took over the surgery. An internal pudendal artery perforating flap (IPAP) surgery was performed for vaginal reconstruction (Fig. 2D, E), and bilateral V-Y advancement flap surgery was performed for the wide perianal soft tissue defect and reconstruction of the neoanus (Fig. 2F). The bilateral V-Y advancement flaps were healed without any event, but the distal portion of the IPAP flap showed congestion and developed skin necrosis. Wound culture from the lesion revealed Enterococcus gallinarum infection, so ciprofloxacin was administrated as a therapeutic antibiotic agent. For the wound complication, the necrotic skin was excised, and the flap was primarily repaired on postoperative day 15.
The patient was discharged on postoperative day 21 from the initial operation. Pathologic reports revealed wide disease extent of 10.2 cm × 6.5 cm and tumor free margins. There has been no evidence of recurrence for 6 months.
A 78-year-old male patient visited our outpatient dermatology clinic complaining of unhealed perianal skin erosion on January 22, 2016. The patient was treated with topical agents in a local clinic at first, but the itching sensation did not subside. The perianal skin had gradually eroded and an exudative discharge had developed for 4 years. On physical examination, welldemarcated eroded macules with some whitish patches in the perianal area were noted. As intertrigo, Bowen disease, or EMPD were suspected, skin biopsy was performed, and EMPD was diagnosed. CEA and CK7 were positive in atypical cells in IHC stains (Fig. 3). The patient was referred to a gastroenterologist to check the extent of anal mucosal lesion and existence of any pathology in the colorectum. Edema and patchy exudate on the anoderm were noted, but the rectal mucosa above the dentate line seemed normal on colonoscopy.
The patient was referred to the division of colorectal surgery for curative resection, and the operation was performed on April 28, 2016. On the day before the operation, mapping biopsies were performed in advance to acquire adequate resection margins (Fig. 4A). Wide excision was performed by the colorectal surgery team; the perianal skin and grossly affected anoderm were resected and sent for frozen biopsy. The first frozen biopsy revealed tumor positive resection margin of the anoderm; therefore, two more resections were performed, and the rectal mucosa above the dentate line was finally removed (Fig. 4B). The final resection margins were free from tumor in the following frozen biopsies, and laparoscopic-assisted loop ileostomy was formed for temporary fecal diversion.
The operation was then taken over by the plastic surgery team. Split thickness skin graft (STSG) and full thickness skin graft (FTSG) were performed with the skin graft of the right thigh and the left inguinal skin, respectively. The STSG was 15 cm × 7 cm. A Pneumatic Zimmer was used, and the thickness of the graft was 10/1,000 inches. The FTSG was 4 cm × 16 cm. MatriDerm (Dr. Otto Suwelack Skin & Health Care AG, Billerbeck, Germany) was laid on the defect site. Then, the STSG and the FTSG were grafted on the defect. The subcutaneous tissue was approximated with vicryl 3-0, and the skin grafts and rectal mucosa were approximated with nylon 4-0 to reconstruct the neoanus (Fig. 4C, D). Vacuum dressing was applied over the wound.
Despite prophylactic antibiotic use of second-generation cephalosporin, wound infection developed around 10 days after the operation, and E. faecalis and Pseudomonas aeruginosa were cultured, so ciprofloxacin was administrated intravenously as a therapeutic antibiotic agent. On postoperative day 28, a second operation for the graft failure was performed. Debridement was performed, and new STSGs retrieved from the left anteromedial thigh were grafted on the defect. The grafts were 15 cm × 7 cm and 11 cm × 6 cm, and their thickness was 11/1,000 inches. The graft was fixed with black silk 4-0, and tie-over dressing was performed.
On postoperative day 42 from the initial operation, the patient was discharged without any event after the second graft surgery (Fig. 2E). The pathology report revealed a wide affected area of 14.3 cm × 9.6 cm and clear resection margins. There has been no evidence of recurrence for 6 months.
PPD is an uncommon cutaneous neoplasm arising from the apocrine glands or underlying carcinoma in the perianal area. Minicozzi et al. [2] estimated that PPD comprises about 20% of EMPD cases, less than 1% of anal diseases, and 6.5% of all Paget diseases. Most of the articles about PPD are sporadic case reports. The largest number of PPD patients (65 patients) reported to date was by Perez et al. [1] in 2014.
Because of its rarity, the exact incidence or survival rate of PPD is not known, and treatment strategies have not been standardized. Among all EMPD cases, PPD tends to have the worst survival rate (10-year overall survival: about 20% in patients with PPD vs. about 60% in patients with EMPD in the trunk), and patients with intraepithelial disease show much better prognosis than patients with invasive PPD or with synchronous malignancy [3]. The incidence and recurrence rates of PPD with invasiveness or synchronous malignancy have been reported to be around twice as those of intraepithelial disease (about 37% vs. 63% for incidence; about 29% vs. 65% for recurrence) [1]. Herrel et al. [3] reported that the 10-year overall survival rate of PPD patients was about 20%.
The treatment of PPD has not been standardized yet because of its rarity. Möller et al. [4] recently revised staging system with treatment options according to its stages based on updated data in 2014 (Table 1).
These staging systems represent 2 major theories of the pathogenesis of PPD. One (primary PPD) is epidermal stem cell-originated intraepithelial neoplasia. If the neoplasm penetrates the basement membrane and invades the stroma, then lymph node metastasis and distant metastasis may occur. The other (secondary PPD) is extracutaneous synchronous malignancy-originated neoplasia, and spreading of the remote carcinoma to the cutaneous lesion may be contiguous or not [5]. Therefore, it is essential to evaluate thoroughly whether patients have synchronous malignancy in remote sites or not.
In addition, histologic findings are significant to differentiate primary PPD not only from other diseases mimicking PPD such as Bowen disease, cutaneous superficial spreading melanoma (SSM), and dermatitis, but also from secondary PPD. The key component of the differential diagnosis is Paget cells, which are large tumor cells with pale clear cytoplasm and large hyperchromatic nuclei. Periodic acid-Schiff (PAS) and mucicarmine stains show positive results in PPD, in contrast to Bowen disease which shows negative staining results. On immunohistochemistry, Paget cells display strong cytoplasmic CEA positivity, and may show positive expression of Her-2/neu (C-erb B2) [6]. For discriminating PPD from SSM, HMB-45, a melanoma-specific monoclonal antibody, is commonly used, and SSM displays positive HMB-45 in contrast to PPD that shows negative HMB-45 [6]. To differentiate primary PPD from secondary PPD, primary PPD usually displays positive expressions of CK7 and gross cystic disease fluid protein-15 (GCDFP-15), which a sensitive maker for apocrine differentiation along with CD-15 (Leu-M1) and lysozyme, with a lack of CK20 expression, presenting sweat gland differentiation. On the contrary, secondary PPD usually displays positive CK7 and CK20 with negative GCDFP-15, presenting endodermal differentiation [5]. However, pankeratin, S100, CD15, lysozyme, PAS, and Alcian blue have no different staining patterns between primary and secondary PPD [5].
Despite the lack of standardized treatment, treatment is focused on local control because of the multifocality and high local recurrence rate of the disease. Determination of the resection margin is important. The most widely used approaches are Mohs micrographic surgery (MMS), intraoperative frozen biopsy, and scouting biopsy (mapping biopsy). MMS has the advantage of maximal preservation of anatomically important structures through accurate intraoperative microscopic evaluation. Intraoperative frozen biopsy is also a good tool for determination of the resection margin, but false-negative rates up to 40% were reported [14]. Scouting biopsy has been reported as a useful preoperative method to evaluate the disease extent not only in the perianal skin but also within the anal canal. This later method is useful for deciding on the surgical extent [4].
No consensus on the extent of the resection margin exists. Recent studies on the surgical treatment of PPD showed recurrence rates of 33% to 60% with a lateral resection margin of 1–3 cm and a deep margin of 0.5 cm [2]. Sasaki et al. [7] and Mizuno et al. [8] reported wide excision of the rectal mucosa up to 2 cm above the dentate line for lesions without involvement of the dentate line, and up to 4.5 cm above the dentate line for lesions with involvement of the dentate line for non-invasive disease. For patients with invasive or secondary PPD, synchronous malignancy should be removed, so resection of the rectum or abdominoperineal resection with combined chemotherapy may be needed [1].
Some other nonsurgical therapeutic modalities have been suggested. Radiotherapy may provide benefits to patients with inoperable lesion because of its large size or to elderly patients with comorbidities [9]. Chemoradiation, systemic chemotherapy, topical agents such as imiquimod and 5-fluoruracil, and photodynamic therapy have been also reported. However, these therapeutic modalities need to be validated [12349].
After wide excision, a large soft tissue defect is inevitable in most cases. Several ways to manage soft tissue defects have been introduced. Waiting for secondary healing is one of them, and STSG and FTSG with or without a vacuumassisted dressing device can be also applied [10]. Reconstruction by using bilateral myocutaneous flaps of the gluteus maximus or thigh muscles or by using V-Y advancement flap is also used [7]. For those with vaginal mucosal defect, an IPAP flap may be additionally applied for concurrent vaginal reconstruction as in case 1. Complications of reconstruction include wound dehiscence, infection, flap hematoma, and anal stricture. Wound infection and consecutive dehiscence may be prevented by fecal diversion.
The cases reported in this article had long standing and unhealed perianal eczematous lesions despite topical treatment and had anal canal involvement. The image work-ups and colonoscopy confirmed no distant metastasis and no synchronous malignancy. Moreover, hematoxylin and eosin and IHC staining revealed Paget cells confined to the epidermis and positive CK7, suggesting primary noninvasive PPD. Both patients were successfully treated with wide excision and flap surgeries and had temporary loop ileostomy for fecal diversion. These patients are on regular followup and have not shown local recurrence (Table 2).
In conclusion, it is important to suspect PPD in the presence of unhealed perianal skin lesions. Wide excision with negative margins is the most important aspect in surgical treatment. Regular and close follow-up with colonoscopy, computed tomography scan, and random skin biopsy are highly recommended because PPD has high incidence rates of local recurrence and multifocality and a relatively poorer prognosis compared with other types of EMPD [13].
Figures and Tables
Fig. 1
H&E and IHC stain in case 1. (A) Clusters of large epithelioid cells with pleomorphic nuclei and pale cytoplasm are present in the epidermis, consistent with EMPD (H&E, ×200). (B) Tumor cells show strong immunoreactivity for CK7 with cytoplasmic staining pattern (×100). (C) Tumor cells are negative for HMB-45 IHC, ruling out the possibility of melanoma (×100). CK7, cytokeratin 7; EMPD, extramammary Paget's disease; H&E, hematoxylin and eosin; HMB-45, human melanoma black-45; IHC, immunohistochemistry.

Fig. 2
Case 1. (A) Erythematous perianal skin lesion with slightly elevated patches, erosion, and brownish pigmentation; scout line determined by mapping biopsies (purple). (B) Wide excision with sphincter preservation. (C) Surgical specimen. (D, E) Internal pudendal artery perforator (IPAP) flap. (F) After completion of the IPAP flap and V-Y advancement flap surgery.
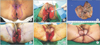
Fig. 3
H&E and IHC in case 2. (A) Clusters of large epithelioid cells with pleomorphic nuclei and pale cytoplasm in the epidermis (H&E, ×200). (B) Tumor cells showing strong immunoreactivity for CK7 (×100; red, in cytoplasm). (C) Tumor cells showing immunoreactivity for CEA (×100; brown, in cytoplasm). CEA, carcinoembryonic antigen; CK7, cytokeratin 7; H&E, hematoxylin and eosin; IHC, immunohistochemistry.

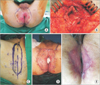
Fig. 4
Case 2. (A) Eroded macules with some whitish patches in the perianal area; scout line determined by mapping biopsies (purple). (B) Wide excision with sphincter preservation. (C, D) Skin graft surgery. (E) Healed wound.
References
1. Perez DR, Trakarnsanga A, Shia J, Nash GM, Temple LK, Paty PB, et al. Management and outcome of perianal Paget's disease: a 6-decade institutional experience. Dis Colon Rectum. 2014; 57:747–751.
2. Minicozzi A, Borzellino G, Momo R, Steccanella F, Pitoni F, de Manzoni G. Perianal Paget's disease: presentation of six cases and literature review. Int J Colorectal Dis. 2010; 25:1–7.
3. Herrel LA, Weiss AD, Goodman M, Johnson TV, Osunkoya AO, Delman KA, et al. Extramammary Paget's disease in males: survival outcomes in 495 patients. Ann Surg Oncol. 2015; 22:1625–1630.
4. Moller MG, Lugo-Baruqui JA, Milikowski C, Salgado CJ. Staged marginal contoured and central excision technique in the surgical management of perianal Paget's disease. Am J Surg. 2014; 207:485–492.
5. Nowak MA, Guerriere-Kovach P, Pathan A, Campbell TE, Deppisch LM. Perianal Paget's disease: distinguishing primary and secondary lesions using immunohistochemical studies including gross cystic disease fluid protein-15 and cytokeratin 20 expression. Arch Pathol Lab Med. 1998; 122:1077–1081.
6. Miyamoto A, Akasaka K, Oikawa H, Akasaka T, Masuda T, Maesawa C. Immunohistochemical study of HER2 and TUBB3 proteins in extramammary Paget disease. Am J Dermatopathol. 2010; 32:578–585.
7. Sasaki K, Nozaki M, Kikutchi Y, Yamaki T, Soejima K. Reconstruction of perianal skin defect using a V-Y advancement of bilateral gluteus maximus musculocutaneous flaps: reconstruction considering anal cleft and anal function. Br J Plast Surg. 1999; 52:471–475.
8. Mizuno H, Iwasaki Y, Noda H, Kawai M, Yamamoto S, Takesue Y. The surgical treatment of perianal paget's disease. Nishinihon J Dermatol. 2001; 63:309–313.
9. Brown RS, Lankester KJ, McCormack M, Power DA, Spittle MF. Radiotherapy for perianal Paget's disease. Clin Oncol (R Coll Radiol). 2002; 14:272–284.
10. Seckel BR, Schoetz DJ Jr, Coller JA. Skin grafts for circumferential coverage of perianal wounds. Surg Clin North Am. 1985; 65:365–371.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download