Abstract
Purpose
The pedicled, descending-branch muscle-sparing latissimus dorsi (MSLD) flap has been widely used for breast reconstruction following total mastectomy. However, the superiority of the MSLD flap compared to the conventional latissimus dorsi (CLD) flap in preventing seroma formation has not been demonstrated. This study compares the morbidities related to seroma formation following pedicled MSLD flap and CLD flap breast reconstruction.
Methods
A total of 15 women who underwent partial mastectomy and immediate partial breast reconstruction with MSLD flaps were compared with 15 women under identical conditions with CLD flap breast reconstruction. The medical records were reviewed for both complications and demographic data. The authors compared morbidity, including donor-site seroma, total volume of drain discharge, indwelling period of drainage, and length of hospital stay following both MSLD flap and CLD flap breast reconstruction.
Results
The demographic data of the 2 groups were not significantly different. Donor-site seroma occurred in 2 MSLD patients (13.3%) and in 6 CLD patients (40.0%). The total volume of the drain discharge and the indwelling period of drainage at donor site were significantly lower in the MSLD group. The length of hospital stay was significantly shorter (by approximately a day and a half) for the MSLD group.
The use of the conventional latissimus dorsi (CLD) flap is a reliable and aesthetic method for primary reconstruction and a valid tool for correcting postoperative contour deformities of the breast. However, this technique is still associated with a high incidence of donor site seroma [123]. Although several preventive surgical techniques and/or medical interventions have been tried, it is still not possible to significantly reduce postoperative complications in a standard surgical procedure.
Recently, the muscle-sparing latissimus dorsi (MSLD) technique, based on either the descending or transverse branch of the thoracodorsal artery for breast reconstruction, has been introduced [4567]. Some advantages of the MSLD flap, such as latissimus dorsi (LD) muscle preservation, less axillary bulk, less contour deformity of the back, and placement of the donor site scar within the horizontal bra line have been described. Additionally, a significant reduction in the incidence of seroma was indicated as an additional advantage. Kim et al. [8] have reported the superiority of the MSLD flap in producing reduced donor-site morbidity compared to that of the extended latissimus dorsi flap method that evolved to include parascapular and lumbar fat. However, this study had a fundamental limitation in that the difference of flap size in each method was not considered. The larger the flap size is, the wider an area of dissection or shear will be. The increase in flap volume is correlated with greater donor-site morbidity, such as wound dehiscence, skin necrosis, and seroma formation. These morbidities with the larger flap size are due to damaged lymph vessels and inflammatory exudates caused by more extensive subcutaneous and muscle dissection. This important bias should be adequately controlled to guarantee a robust method and to achieve more reliable evidence. The purpose of this study was to compare the incidence of donor-site seroma in MSLD flap and in CLD flap in patients with a comparable flap volume.
In our hospital, the indication for MSLD over CLD included patients who were undergoing either lateral partial mastectomy or had a relatively small breast. After obtaining informed consent and Institutional Review Board approval, a retrospective medical record review was performed for a consecutive series of patients undergoing partial (less than half) mastectomy and immediate partial breast reconstruction using either pedicled MSLD flaps or pedicled CLD flaps between Jan 2011 and May 2015 in our hospital, which is associated with Kyoto Prefectural University of Medicine. In our hospital, CLD flap was used until June 2013, but that procedure was replaced by MSLD flap from July 2013 until the present.
Obese patients (body mass index [BMI] > 23 kg/m2), who required either a central or lateral lymphadenectomy, were excluded from the study because they were suspected of having a higher risk for seroma formation. The patients' charts were reviewed, and information regarding age, BMI, surgical location, presence or absence of neoadjuvant chemotherapy, history of smoking, flap size, single versus double mastectomy, drain discharge from the anterior chest and donor sites, indwelling period of drainage at the anterior chest and donor sites, incidence of seroma, and length of hospital stay were collected for each patient. Total flap sizes (weights) were measured intraoperatively by hanging the flap from a spring weight-scale. Finally, during the in-office final follow-up outpatient visit, patients were asked to score their satisfaction with breast reconstruction in both groups with a visual analogue scale scale, described by Tremp et al. [9] (ranging from 0 if not satisfied to 10 if completely satisfied).
For CLD-flap harvest, almost all of the LD muscle was included in the flap, and the dissection extended to the main thoracodorsal artery (Fig. 1A). The MSLD flaps were elevated as described by Saint-Cyr et al. [4]. Briefly, the LD muscle was cut longitudinally along the muscle fiber medial to the descending branch of the thoracodorsal artery. The size of the harvested muscle strip did not exceed 5 cm in width (Fig. 1B). The transverse branch of the thoracodorsal artery and the main thoracodorsal nerve were not included in the harvested MSLD flap. The flap was then transferred to the breast pocket subcutaneously. The pivot point of the CLD flap was at the main pedicle level, while the pivot point of the MSLD flap was at the bifurcation level of the main pedicle into the descending and transverse branches.
In all of the patients, a 15F vacuum drainage tube was placed below the transplanted flap (anterior chest) and at the donor site (back) upon completion of the surgical procedure. The drains were removed when drainage was less than 30 mL/24 hours; otherwise, they were removed at 2 weeks, postoperatively. After removal of the drains, puncture drainage with a needle was performed every third day when there was liquid accumulation at the donor site; and we measured the quantity of fluid accumulation with a scale attachment syringe.
Statistical analysis was performed using InStat ver. 3.0 (GraphPad, New York, NY, USA). The independent t-test (age, BMI, and indwelling period), Mann-Whitney U-test (flap size, seroma volume, frequency of aspiration, drainage volume, and length of hospital stay), and Pearson chi-square test (seroma incidence) were used to analyze the effects of the interventions. The authors compared the identified parameters and set the level of significance at the 0.05 alpha level. All the results are shown either as mean ± standard deviation or median (interquartile range).
The average age of all the patients was 45.2 ± 8.9 years, the average BMI was 21.6 ± 2.8 kg/m2, and the average flap size was 157.1 ± 44.5 g. There were no statistically significant differences between the 2 groups with regard to age, BMI, and flap size (Table 1). The mean flap weight in the MSLD group (153.4 ± 42 mg) was comparable with that in the CLD group (160.9 ± 47 mg) (P = 0.691) (Table 1). Flap loss, haematoma, and delayed wound healing did not occur in any patient in either group. A flap with fat necrosis was found in one patient in the MSLD group, whereas no fat necrosis occurred in the CLD group. This difference, however, was not statistically significant (P = 0.31).
Two patients (13.3%) in MSLD group had a seroma, whereas six patients (40.0%) in the CLD group had a seroma (odds ratio, 0.23; relative risk, 0.69; P = 0.22) (Table 2). Patients who had breast reconstruction with the MSLD flap had about a 3.0-fold decreased risk of seroma compared to that of patients who had breast reconstruction with the CLD flap, although this difference did not reach significance.
The total volumes of the drain discharge in both the anterior chest and the back were significantly decreased in the MSLD group from a median of 180.5 mL (30–440 mL) to 168.7 mL (38–325 mL) (P = 0.74) and from a median of 697.3 mL (410–1,520 mL) to 431.1 mL (140–883 mL) (P = 0.013), respectively. The indwelling period of drainage in the back was shorter with a median of 4.27 days (5–14 days) in the MSLD group than that of 6.91 days (4–9 days) (P = 0.014) in the CLD group, whereas the indwelling period in the anterior chest was comparable between the two groups (P = 0.78) (Table 2). The length of the hospital stay was significantly shorter for the MSLD group by approximately 1.5 days (P = 0.024) (Table 2). Patient self-assessment of their satisfaction with the breast reconstruction revealed that the patients in MSLD group were slightly more satisfied (P = 0.065, chi-square test) with the result than patients after breast reconstruction using CLD flap (Fig. 2). Examples of breast reconstruction using the MSLD flap in our series are shown in Figs. 3, 4.
Since the MSLD flap was first introduced by Tobin et al. [10] in 1981, additional techniques and applications have been developed for breast reconstruction. The MSLD flap has several advantages over the thoracodorsal artery perforator (TAP) flap [1112]. First, the MSLD flap, including TAP within a strip of muscle, reduces the incidence of partial flap loss, compared with that of the TAP flap. Additional advantages are that the MSLD flap is easy to design irrespective of perforator location, it lacks dependence on adequately sized perforators, and it is a simple technical dissection. The MSLD flap is superior because it is less invasive than the CLD flap with more than half of the latissimus dorsi muscle preserved. Saint-Cyr et al. [4] reported that the MSLD flap with a transversely-oriented skin paddle results in minimal functional deficit of the donor site, absence of seroma, and a cosmetically acceptable scar. These findings seem to be also consistent with our result of higher patient satisfaction after breast reconstruction using MSLD flap. Consequently, the MSLD flap has recently been widely used for breast reconstruction following total mastectomy [456]. At our hospital, the indications to perform MSLD flap over CLD flap were for patients undergoing either a lateral partial mastectomy or a total mastectomy but who had a relatively small breast (less than 200 mL), which is commonly found in Asian patients.
The etiology of seroma is considered to be multifactorial, including the disruption of lymphatic vessels, consequence of inflammatory exudates due to subcutaneous and muscle dissection, and amount of dead space [131415]. Accordingly, we believe that the correlation between increased flap size with the incidence of seroma might be strong. However, a previous study compared the different sized flaps in donor-site morbidities related with seroma. Taking the size factor into account, the current study, comparing similarly sized MSLD and CLD flaps, overcomes this variable size limitation of the previous study.
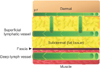
Some mechanisms have been suggested to explain why the MSLD flap is superior to the CLD flap with similar flap sizes. The MSLD flap and the CLD flap differ mostly in anatomic locations dissected. Deep lymph vessels exist below the fascia and drain lipoproteins and chylomicrons in blood vessels within muscle [1617] (Fig. 5). In the MSLD flap, the proportion of muscle where comparatively deep lymph vessels are located is smaller than that in the CLD flap. Moreover, deep lymph vessels seem to be potentially affected by reflection from muscle movement and accordingly are more difficult to seal. Santanelli et al. [12] reported that seroma was not observed at the donor site of seven patients who underwent total breast reconstruction with a pedicled TAP flap without muscle. Another possible explanation is that the MSLD flap might be less invasive than the CLD flap because the dissected site for elevating the MSLD flap is along muscle fibers. Further study is needed to examine which MSLD flap factors reduce the frequency of donor-site morbidities related to seroma.
There are some limitations to this study with the foremost being the small sample size. The subcategories of race, smoking status, and hypertension are extremely small in number, having an imperceptible effect on the statistical analysis. Based on our method, there was some selection bias in both groups, considering the limitations of this nonrandomized retrospective study. Additional prospective randomized controlled trials will be required to obtain powerful and reliable evidence for the superiority of the MSLD flap over that of the CLD flap in donor-site morbidities related to seroma.
In summary, although the data are limited, the MSLD flap for breast reconstruction may be effective and a useful option for reducing the indwelling period of drainage and thus the length of the hospital stay.
In conclusion, postoperative seroma is the most common complication of latissimus dorsi flap surgery for breast reconstruction. We found that the MSLD flap is superior to the CLD flap in terms of reducing seroma formation at the donor site, length of the drainage period, surgical time, and length of the hospital stay. Thus, the MSLD flap may be a useful option for small breast reconstruction if earlier discharge from a hospital is demanded.
Figures and Tables
Fig. 1
The area of flap elevation. Flap is elevated along dotted line. Red-shaded area shows muscle part of flap, while yellow-shaded area shows fat tissue of flap. (A) Conventional latissimus dorsi (CLD) flap elevation. (B) Muscle-sparing latissimus dorsi (MSLD) flap elevation. Flap elevation and separation of descending branch of thoracodorsal artery is shown. Latissimus dorsi muscle is split vertically along its natural muscle fiber orientation. Pedicle width of harvested latissimus dorsi muscle is shown.
Fig. 2
Patient satisfaction. Satisfaction was higher in patients after nipple reconstruction in muscle-sparing latissimus dorsi (MSLD) flap group than in conventional latissimus dorsi (CLD) flap group.
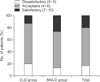
Fig. 3
(A) Preoperative view of 42-year-old patient with left breast cancer (ductal carcinoma in situ). (B) Intraoperative view after harvest of 7 × 14-cm muscle-sparing latissimus dorsi (MSLD) flap based on descending branch of thoracodorsal artery with 4 cm wide latissimus dorsi muscle cuff. Pivot point of MSLD flap was at bifurcation level of main pedicle into descending and transverse branches. Flap weight is 245 g. (C) Intraoperative view of donor site. Dotted line indicates preserved latissimus dorsi muscle in left back. (D) Postoperative appearance 9 months after breast reconstruction using MSLD flap. (E) 9-month postoperative view of donor site.
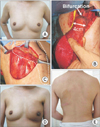
Fig. 4
(A) Preoperative photographs of 39-year-old patient with left breast cancer. (B) Intraoperative view after harvest of 6.5 × 13-cm muscle-sparing latissimus dorsi flap. Latissimus dorsi muscle was cut longitudinally along muscle fiber medial to descending branch of thoracodorsal artery. Size of harvested muscle strip is 5 cm in width. Flap weight is 146 g. (C) Image of flap in-setting to lateral partial defect in left breast. (D) Postoperative photographs at 14 months. (E) Donor scar within horizontal bra strap region.
Fig. 5
Schematic diagram demonstrates lymphatic system across different layers, including dermal, subdermal, fascia, and muscle.
References
1. Clough KB, Louis-Sylvestre C, Fitoussi A, Couturaud B, Nos C. Donor site sequelae after autologous breast reconstruction with an extended latissimus dorsi flap. Plast Reconstr Surg. 2002; 109:1904–1911.
2. Roy MK, Shrotia S, Holcombe C, Webster DJ, Hughes LE, Mansel RE. Complications of latissimus dorsi myocutaneous flap breast reconstruction. Eur J Surg Oncol. 1998; 24:162–165.
3. Schwabegger A, Ninkovic M, Brenner E, Anderl H. Seroma as a common donor site morbidity after harvesting the latissimus dorsi flap: observations on cause and prevention. Ann Plast Surg. 1997; 38:594–597.
4. Saint-Cyr M, Nagarkar P, Schaverien M, Dauwe P, Wong C, Rohrich RJ. The pedicled descending branch muscle-sparing latissimus dorsi flap for breast reconstruction. Plast Reconstr Surg. 2009; 123:13–24.
5. Brackley PT, Mishra A, Sigaroudina M, Iqbal A. Modified muscle sparing latissimus dorsi with implant for total breast reconstruction - extending the boundaries. J Plast Reconstr Aesthet Surg. 2010; 63:1495–1502.
6. Colohan S, Wong C, Lakhiani C, Cheng A, Maia M, Arbique G, et al. The free descending branch muscle-sparing latissimus dorsi flap: vascular anatomy and clinical applications. Plast Reconstr Surg. 2012; 130:776e–787e.
7. Zhu L, Mohan AT, Saint-Cyr M. A simple approach to harvest of the pedicled descending branch muscle-sparing latissimus dorsi flap. J Plast Reconstr Aesthet Surg. 2015; 68:e179–e181.
8. Kim H, Wiraatmadja ES, Lim SY, Pyon JK, Bang SI, Oh KS, et al. Comparison of morbidity of donor site following pedicled muscle-sparing latissimus dorsi flap versus extended latissimus dorsi flap breast reconstruction. J Plast Reconstr Aesthet Surg. 2013; 66:640–646.
9. Tremp M, di Summa PG, Schaakxs D, Oranges CM, Wettstein R, Kalbermatten DF. Nipple reconstruction after autologous or expander breast reconstruction: a multimodal and 3-dimensional analysis. Aesthet Surg J. 2017; 37:179–187.
10. Tobin GR, Moberg AW, DuBou RH, Weiner LJ, Bland KI. The split latissimus dorsi myocutaneous flap. Ann Plast Surg. 1981; 7:272–280.
11. Hamdi M, Van Landuyt K, Hijjawi JB, Roche N, Blondeel P, Monstrey S. Surgical technique in pedicled thoracodorsal artery perforator flaps: a clinical experience with 99 patients. Plast Reconstr Surg. 2008; 121:1632–1641.
12. Santanelli F, Longo B, Germano S, Rubino C, Laporta R, Hamdi M. Total breast reconstruction using the thoracodorsal artery perforator flap without implant. Plast Reconstr Surg. 2014; 133:251–254.
13. Woodworth PA, McBoyle MF, Helmer SD, Beamer RL. Seroma formation after breast cancer surgery: incidence and predicting factors. Am Surg. 2000; 66:444–450.
14. McCaul JA, Aslaam A, Spooner RJ, Louden I, Cavanagh T, Purushotham AD. Aetiology of seroma formation in patients undergoing surgery for breast cancer. Breast. 2000; 9:144–148.
15. Di Monta G, Caraco C, Crispo A, Marone U, Mozzillo N. Collagen sealant patch to reduce lymphatic drainage after lymph node dissection. World J Surg Oncol. 2012; 10:275.
16. Suami H, Taylor GI, Pan WR. The lymphatic territories of the upper limb: anatomical study and clinical implications. Plast Reconstr Surg. 2007; 119:1813–1822.
17. Campisi C, Bellini C, Campisi C, Accogli S, Bonioli E, Boccardo F. Microsurgery for lymphedema: clinical research and long-term results. Microsurgery. 2010; 30:256–260.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download