This article has been corrected. See "ERRATUM: Correction of the 1st and 2nd authors' affiliation: Transfusion free radical antegrade modular pancreaticosplenectomy of metastatic neuroendocrine tumor of the pancreas in Jehovah's Witness patient" in Volume 88 on page 178.
Abstract
In a popular sense, Jehovah's Witnesses (JW) have their creeds, one of which is refusal of blood transfusion. Such refusal may impinge on their proper management, especially in critical situations. We present a case of successful bloodless multimodality therapy, which was performed for a JW. The patient was a 49-year-old woman and JW who had general weakness 7 days before admission. She was diagnosed with a pancreatic neuroendocrine tumor (PNET) with hepatic metastases. Transcatheter arterial chemoembolization and Sandostatin LAR injection were performed, and then she was given a transfusion-free Radical antegrade modular pancreatosplenectomy sequentially. We gave recombinant human erythropoietin and iron hydroxide sucrose complex daily for five days after surgery. She was discharged at postoperative day 12 without any surgical complications. Multimodality therapy is very important for optimal treatment of PNET. Along with intimate interdepartmental cooperation, careful patient selection and appropriate perioperative management could possibly enhance the surgical outcome.
Pancreatic neuroendocrine tumors (PNETs) form a rare subgroup of tumors found in the pancreas, where liver is the most common site of metastasis [1]. Sometimes the liver metastases are unresectable; however, a combination treatment with perioperative intervention for the liver and resection of the primary pancreatic tumor may provide patients with survival benefits [2]. For the surgery of the pancreas to be successful, intensive concentration and expertise are required. Although transfusion rates associated with pancreas resection have decreased over time, the resection procedure still could cause bleeding, especially challenging in Jehovah's Witness (JW) patients. Experience of pancreas resection in JW patients has been limited, with most of the published literature in the form of case reports [3]. To the best of our knowledge, this is the first case report of radical antegrade modular pancreatosplenectomy (RAMPS) for a PNET with multiple liver metastases in a Jehovah's Witness patient. In this report, we review the experience of pancreatic surgery in a JW patient, focusing on safety and effectiveness of RAMPS procedure, along with the perioperative techniques used for synchronous unresectable liver metastasis.
A 49-year-old female patient came to the outpatient clinic with complaint of general weakness, mild discomfort and heat sensation in the abdomen. Diagnosed with PNET with multiple liver metastases at another hospital, she was transferred to our clinic for bloodless surgery owing to the patient's religious faith. The patient's height was 152 cm and weight was 53.6 kg. She didn't have any other underlying medical diseases such as hypertension, diabetes, hepatitis, tuberculosis and so on. Physical exams revealed mildly distended abdomen and a non-tender palpable intraabdominal mass. The serum tumor markers including CEA, CA 19-9, and α-FP were within normal limits, whereas liver function test levels including total bilirubin (1.3 mg/dL), aspartate transaminase (58 U/L), and alanine transaminase (49 U/L) were slightly elevated. Radiological tools including abdominal-pelvic CT scan, biliary and pancreas dynamic MRI with contrast, PET/CT scan, and bone scan contributed to diagnosis and therapeutic plan. According to the results of radiological imaging studies, a huge well capsulated tumor (about 8-cm diameter on the longest distance) was located in the pancreatic body and tail, which was highly enhanced by the contrast material. Multiple hepatic metastases were found in both lobes (Fig. 1A, B).
Sono-guided biopsies on liver and pancreas showed proliferation of uniform round cells in ribbon, consistent with metastatic well-differentiated PNET from the pancreas. Results revealed positive staining of chromogranin A and cytokeratin (CK). Ki67 labeling index was about 4%-5% that corresponded to grade 2. For treatment of PNET with multiple liver metastases, serial therapies were scheduled. First of all, transarterial chemoembolization (TACE) was performed prior to surgery. The interventional radiologist performed TACE with mitomycin and lipiolol, which were injected to the patient's metastatic hepatic tumors. After a total of 3 sessions of successful TACE for multiple liver metastases over 4 months (Fig. 1C, D), the size of multiple liver metastases decreased dramatically in the following abdomen CT (Fig. 1E, F). Meanwhile, the patient suffered from liver failure with total bilirubin increased up to 23 mg/dL. Careful conservative management without transfusion was carried out for treatment of liver impairment. For the systemic treatment, octreotide acetate injection was given before surgery. Initial dose was 0.1 mg via subcutaneous injection every 8 hours over 3 days for response test, and then 4 injections at 20 mg were given daily via intramuscular route per 28 days.
The attending surgeon and anesthesiologist discussed the risks of bloodless major operations. The patient signed a consent form for the operation without the use of blood products. She allowed albumin and acute normovolemic hemodilution (ANH). Considering that it was a cancer surgery, Cell Saver was not allowed due to risk of shed cancer cells. The preoperative albumin level was 3.7 g/dL, hemoglobin was 12.6 mg/dL, and hematocrit was 36.8%. The dissection of RAMPS procedure started from the left side, which is the neck of the pancreas, to the right side with splenic vessels and celiac node dissection. Afterwards, the plane of dissection proceeded posteriorly in a sagittal plane along the celiac artery and the superior mesenteric artery down to the level of the aorta, in sequence, going on laterally to the left adrenal gland (Fig. 2). There was severe adhesion of omentum to the hepatic tumors and pancreatic tumor, however, the celiac trunk and the superior mesenteric artery were relatively well isolated from the tumor. The pancreatic tumor was well vascularized, capsulated, and abutted to the renal arteries and veins, but not invading (Fig. 3A-C). Histological diagnosis was consistent with well-differentiated pancreatic neuroendocrine carcinoma with negative resection margin but extending to the peripancreatic soft tissue and splenic venous wall. It showed angioinvasion and neural invasion containing ischemic necrosis. Immunohistochemical examination revealed that chromogranin A, CD56, CD10, and CK were positive (Fig. 3D-G). Pathologic staging was T3N0M1 stage IV according to American Joint Committee on Cancer/Union for International Cancer Control TNM staging for pancreatic tumors.
Total operative time was 4 hours 30 minutes. Estimated blood loss was 800 mL and while the patient received 3 pints of ANH and intravenous dextran during the operation. The postoperative albumin was 2.8 g/dL, hemoglobin 11.5 mg/dL, hematocrit 33.8%, and fibrinogen 248 mg/dL. As an assistant therapy for a transfusion free patient, recombinant human erythropoietin 4,000 IU and 1 ample of iron hydroxide sucrose complex were given daily for five days after surgery. Postoperative abdominal CT scan didn't show any evidence of pancreatic fistula or fluid collections around the pancreatic resection margin.
The patient took a sip of water on day 2 and began a soft diet on day 4, postoperatively. She was discharged 12 days after the operation. We had future plans to give octreotide acetate injection for the systemic treatment and additional TACE for liver metastasis
The fact is well known that JW patients adamantly refuse blood products despite the risk of death. Recently, numerous centers have reported successful transfusion-free procedures in JW patients, including coronary artery bypass grafting, liver transplantation, right trisectionectomy, and pancreaticoduodenectomy. Experience with pancreas surgery in JW patients is limited, and most of the existing literatures are in the form of case reports [3]. To the best of our knowledge, this is the first report on multimodality therapy on a JW patient with metastatic PNETs.
Neuroendocrine tumors are a group of various neoplasms, which arise from many organs of the body such as gastrointestinal tract, respiratory tract, and also pancreas. PNETs are a rare subgroup, consisting 1%-2% of all pancreatic tumors. PNETs more often arise in the pancreatic tail where they can be treated with distal pancreatosplectomy (DPS) [4]. Conventional DPS was typically performed in a left-to right manner, with the spleen mobilized first and then the posterior aspect of the pancreas dissected from tail to body. Radical antegrade pancreatosplenectomy (RAMPS) is a modification of the conventional DPS developed to provide a complete node dissection based on the established anatomy of lymph node drainage [5]. Park et al. [6] compared 54 DPS and 38 RAMPS. Median estimated blood loss was lower in RAMPS without statistical significance (325 mL in RAMPS and 400 mL in DPS, P = 0.088). Median tumor size was 3.1 cm in RAMPS and 3.8 cm in DPS. There were no comments about blood transfusion amount. Retrieved lymph node counts were significantly more in RAMPS (median, 14; range, 5-52 vs. median, 9; range, 1-36; P = 0.002). R0 resection rate showed no difference (89.4% vs. 85.1%, P = 0.775) [6]. In terms of anatomical dissection, RAMPS not only removed more lymph nodes but also tended to have less blood loss than DPS in pancreatic cancer operation. Therefore, for the purpose of transfusion-free pancreas cancer surgery, RAMPS seems to be more suitable than DPS for JW patients. In our patient, the amount of blood loss was 800 mL due to the large size of the tumor. However, there was no significant change in perioperative hemoglobin level. In addition, we could achieve pathologic negative resection margin.
Liver is the most common site of metastasis and the single most important prognostic factor [1]. According to Hill et al., surgical therapy significantly improves the survival rate in patients not only with localized but also with metastatic nonfunctional PNETs [7]. Consequently, complete resection or cytoreductive surgery is recommended even when the disease is advanced. Liver transplantation may be helpful for patients with hepatic metastases without extrahepatic disease. Furthermore, interventional procedures including TACE, radiofrequency ablation, neoadjuvant radiostope yttrium-90 therapy, and medical treatment such as somatostatin analogs, cytotoxic chemotherapy, and target therapy have been developed for metastatic, progressive disease, or relief of symptoms [8]. We selected appropriate treatments from the various therapeutic methods mentioned above that are relevant to the specific situation. In this patient, chemoembolization of the hepatic artery was performed by infusion of mitomycin and lipiodol prior to RAMPS, which achieved significant size reduction of liver metastasis. Although the efficacy of aggressive surgery on a primary tumor continues to be controversial in PNETs with unresectable metastasis, some case series of efficacy of multidisciplinary treatment consisting of resection of primary pancreatic tumor following intervention with unresectable synchronous liver metastases have been reported [9].
The development of blood-conservation strategies, including ANH, autologous blood donation, and intraoperative cell salvage (Cell Saver), has been important for preservation of the national blood supply, and some of these approaches can be used in surgical procedures in JW patients. Erythropoietin, often combined with administration of iron sulfate, can be used preoperatively to improve red cell mass, even in the absence of anemia [10]. The main intraoperative modalities used to conserve blood in this patient were the use of ANH and intravenous dextran. Preoperative and postoperative hematocrit levels were 36.8% and 33.8% without blood transfusion. The postoperative course was uneventful and the patient was discharged 12 days after the operation.
In conclusion, if there is no other distant metastasis, for PNETs with unresectable synchronous liver metastases, a multidisciplinary approach could be helpful with transarterial treatment and other systemic treatment. Surgical resection of large PNETs could be achieved in JW patients. For successful treatment of transfusion-free patient, restrictive patient selection with indication, minimization of iatrogenic blood loss (e.g., reliable surgical technique), alternative methods of blood supplement play important roles in patient blood management, and RAMPS is an appropriate procedure for pancreatic cancer surgery for bloodless surgery in JW patients. Multimodality therapy is very important for optimal treatment of PNET. Along with intimate interdepartment cooperation, careful patient selection and appropriate perioperative management could possibly improve the surgical outcome.
Figures and Tables
Fig. 1
Abdominal CT scan. (A, B) A huge well-capsulated tumor (indicated by the arrow) is located in pancreatic body and tail, highly enhanced by contrast material. (C-F) Multiple hepatic metastases were found in both lobes. Successful transarterial chemoembolization was done for multiple lesions in liver.
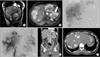
Fig. 2
Surgical dissection plan for radical antegrade modular pancreaticosplenectomy. A, left adrenal gland; C, colon; K, left kidney; P, pancreas; S, spleen.
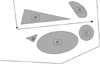
Fig. 3
(A) Surgical field after removal of specimen showed splenic artery stump, left adrenal gland, and superior mesenteric artery (SMA). Gross specimen consists of distal pancreas with tumor mass and spleen. (B, C) On cross cut section, shows roughly ovoid white gray mass (8 cm × 6.5 cm), 1.5 cm from resection margin. Mass is composed of white gray solid creamy tissue with focal cystic change and necrosis. Mass seems to infiltrate into peripancreatic fat tissue. Several enlarged lymph nodes are found in peripancreatic fat tissue. Small clear tumor cells are spreading with trabecular configuration, and destruction of surrounding exocrine tissue show fibrosis, infiltration of mononuclear cells and disarrangement of parenchymal structure were apparent at splenic side of tumor, indicating diffuse chronic pancreatitis (D: H&E, ×10). Extensive angioinvasion is found (E: Victoria blue H&E, ×20). Tumor cells were positively stained for chromogranin A (F, ×20) and CD53 (G, ×20).

References
1. Chamberlain RS, Canes D, Brown KT, Saltz L, Jarnagin W, Fong Y, et al. Hepatic neuroendocrine metastases: does intervention alter outcomes? J Am Coll Surg. 2000; 190:432–445.
2. Hung JS, Chang MC, Lee PH, Tien YW. Is surgery indicated for patients with symptomatic nonfunctioning pancreatic neuroendocrine tumor and unresectable hepatic metastases? World J Surg. 2007; 31:2392–2397.
3. Magner D, Ouellette JR, Lee JR, Colquhoun S, Lo S, Nissen NN. Pancreaticoduodenectomy after neoadjuvant therapy in a Jehovah's witness with locally advanced pancreatic cancer: case report and approach to avoid transfusion. Am Surg. 2006; 72:435–437.
4. Halperin DM, Kulke MH. Management of pancreatic neuroendocrine tumors. Gastroenterol Clin North Am. 2012; 41:119–131.
5. Strasberg SM, Drebin JA, Linehan D. Radical antegrade modular pancreatosplenectomy. Surgery. 2003; 133:521–527.
6. Park HJ, You DD, Choi DW, Heo JS, Choi SH. Role of radical antegrade modular pancreatosplenectomy for adenocarcinoma of the body and tail of the pancreas. World J Surg. 2014; 38:186–193.
7. Hill JS, McPhee JT, McDade TP, Zhou Z, Sullivan ME, Whalen GF, et al. Pancreatic neuroendocrine tumors: the impact of surgical resection on survival. Cancer. 2009; 115:741–751.
8. Muniraj T, Vignesh S, Shetty S, Thiruvengadam S, Aslanian HR. Pancreatic neuroendocrine tumors. Dis Mon. 2013; 59:5–19.
9. Kondo NI, Ikeda Y, Maehara S, Sugimoto R, Nishiyama K, Sakaguchi Y. Role of resection of the primary pancreatic neuroendocrine tumor in the multidisciplinary treatment of patients with unresectable synchronous liver metastases: a case series. JOP. 2013; 14:415–422.
10. Marsh JC, Bevan DH. Haematological care of the Jehovah's Witness patient. Br J Haematol. 2002; 119:25–37.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download