Abstract
We present a rare case of abdominal compartment syndrome due to a bulimic attack in a 19-year-old female patient with bulimia nervosa. She was admitted to our emergency room with complaints of progressive abdominal pain following bulimia. Computed tomography showed dilated stomach with food and air pressed other visceral organs and major abdominal vessels. Decompression using nasogastric tube or gastric lavage tube failed. At laparotomy, we performed gastrotomy and decompression was performed. After decompression, she fell into hypovolemic shock due to bleeding in the intra-gastric and peritoneal cavity. Twelve hours after the operation, the patient died due to refractory hypovolemic shock from uncontrolled bleeding following decompression of abdominal compartment. It should keep in mind that binge-eating habits in patients with bulimic nervosa could cause abdominal compartment syndrome due to gastric distension and this may be a potentially fatal condition.
Abdominal compartment syndrome due to psychogenic polyphagia is extremely rare. Increased intra-abdominal pressure impairs venous return and arterial inflow. Tissue ischemia occurs and inflammatory mediators are activated. Hepatosplanchnic flow, renal blood flow, venous return to the heart is decreased. Eventually, multiple organ failure and ultimately death could occur. Abdominal decompression is essential in its management, but ischemia-reperfusion injury may occur as a complication of decompression [1]. We present a rare case of abdominal compartment syndrome caused by a bulimic attack in an anorexia-bulimia patient.
A 19-year-old female patient visited our emergency room with the chief complaint of abdominal fullness and pain for 4 hours. The pain was described as continuous and significant in intensity. She had persistent nausea, but was unable to vomit. Four hours before visiting our hospital, she ate large amounts of food. During the previous 2 years, she had a tendency to ingest large amounts of food and vomit (binge and purge). She had never received any medical treatment for these symptoms. At admission, she was unable to vomit. Her height and weight were 168 cm and 47.0 kg, respectively.
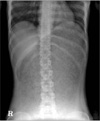
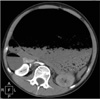
The patient's vital signs were normal but she appeared to be in discomfort and both legs were pale. On physical examination, her abdomen was markedly distended with mild tenderness and tension. Bowel sound was absent, both pulses of dorsalis pedis were absent, and both femoral pulses were weak. The complete blood count was normal, whereas the base excess negatively increased to -8.4 mEq/L. Other laboratory values were within the normal range. A plain abdominal film showed a massively dilated stomach with food material and air. No bowel gases were identified (Fig. 1). An abdominal computed tomography (CT) was immediately obtained, which revealed a severe distended stomach with large amounts of food materials occupying the abdominal cavity entirely from the diaphragm to the pelvis. The esophagogastric junction was shifted towards the right, and the aorta and major branch vessels had collapsed due to the dilated stomach. CT showed infarctions of the left kidney, spleen, body and tail of pancreas, left hepatic lobe, small bowel, transverse colon and descending colon (Fig. 2).
A nasogastric tube for drainage was inserted for decompression but failed, possibly due to the tube having twisted in the esophagogastric junction. Therefore, we attempted to insert a gastric lavage tube, but the lavage tube failed to advance over 40 cm. The lavage tube was also unable to pass the esophagogastric junction.
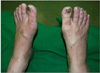
Immediately after the failure in decompression with the nasogastric tube and lavage tube, the patient was taken to the operation room, where cyanosis was noted in the feet (Fig. 3). When inserting foley catheter, the amount of urine collected was only 30 mL. During operation, there was no urination. Under general anesthesia, we inserted an 18-gauge needle into the mid-abdomen through the skin, after which a large amount of airs were released. We performed gastrotomy through a 5 cm skin incision for decompression (Fig. 4), and evacuated over 5,000 mL of food materials and fluids. After that, the abdomen became soft and flat, and both dosalis pedis pulsations recovered. At that time, her blood pressure dropped continuously to 70/40 mmHg and her pulse rate increased to 160 beats/min. The anesthetist increased crystalloid infusion but blood pressure was not corrected. Vasoactive agents were infused, and there was oozing bleeding from the gastric cavity. We could not control the bleeding due to diffuse oozing of the stomach wall. In addition, further bleeding occurred from the peritoneal cavity. The patient was thought to have disseminated intravascular coagulation (DIC). We inserted a JP (Jackson-Pratt) drain in the peritoneal cavity and performed tube gastrostomy using a foley catheter. The patient was sent to the intensive care unit in an unstable hemodynamic state. Twelve hours after the operation, the patient died due to refractory hypovolemic shock from uncontrolled gastric mucosal and intra-peritoneal bleeding following decompression of abdominal compartment syndrome.
There are 12 case reports on gastric necrosis or perforation caused by a bulimic attack using MEDLINE search between 1968 and 1998. Through emergency operation, 11 patients were recovered and 1 patient died. Another fatality caused by heavy meal was reported in 2003 [2]. These two fatal cases were from sepsis due to necrosis and perforation of stomach. However, it is thought that the cause of death in our case is refractory hypovolemic shock caused by ischemia-reperfusion injury.
Previous cases associated with bulimic patients showed problems related to the stomach, but our patient showed general visceral problems, as well as stomach problems, indicating abdominal compartment syndrome. Generally, when over 3 L of intraluminal fluid abruptly distends the stomach, increased pressure of the stomach wall can lead to ischemia of the stomach wall due to venous insufficiency. Rupture can occur with intragastric pressures of more than 120 mmHg (corresponding to approximately 4 L of fluid). Chronic distension of the stomach in eating disorders, such as polyphagia and bulimia, increases the capacity of the stomach to the point where distension of over 15 L may be tolerable [3]. We believe that in our case, distension of the stomach over 4 L occurred without necrosis, so abdominal compartment syndrome occurred before gastric rupture. Although measurement of intra-abdominal pressure was not taken, physical examination and CT implied abdominal compartment syndrome.
CT showed the portal vein, mesenteric artery and renal perfusion were insufficient and anuria occurred. Bailey and Shapiro [4] reported that when intra-abdominal pressure is above 30 mmHg, anuria progresses. Severe progressive reduction in mesenteric blood flow has been shown with graded elevation in intra-abdominal pressure from approximately 70% baseline at 20 mmHg, to 30% at 40 mmHg. According to their report, we assumed that the intra-abdominal pressure of our case was above 40 mmHg, which was grade IV according to the abdominal compartment grading system.
According to Parsak and Sakman [5], mortality in grade IV abdominal compartment syndrome is between 71 to 85%. In decompressive laparotomies, sudden expansion in abdominal and pelvic veins occurs, and reperfusion syndrome such as hypotension, asystoly attack and supraventricular arrhythmia happen. This appearance relates to high mortality [5]. According to the report by Mohapatra [6], when gut ischemia occurs, inflammatory mediators, expression of cell adhesion molecules on endothelial and immune cell surface are generated and the pro-coagulatory properties of the vascular endothelial surface is increased. During reperfusion, gut injury may be amplified by increased production of free oxygen radicals and exhaustion of the endogenous antioxidant defense mechanism. This continuous damage leads to multiple organ dysfunction and death in critically ill patients. The author recommended a prophylactic solution with mannitol and sodium bicarbonate to prevent the systemic reperfusion injury. According to a report by McCord [7]., when ischemia occurs in cats, the mucosal lesions are characterized by extensive lifting of the epithelium from the villi, epithelial necrosis, disintegration of the lamina propria, hemorrhage, and ulceration due to free radicals. In our case, during early decompression, there was no bleeding. But when the reperfusion was made after decompression through gastrotomy, bleeding occurred in the gastric and peritoneal cavity. However, we did not find the source of bleeding specifically. We assume this bleeding was due to intestinal damage of the ischemia-reperfusion injury. We also believe that DIC occurred due to continuous bleeding, which was not possible to control. Her death was associated with irreversible hypovolemic shock and metabolic acidosis.
Guan et al. [8] showed that using in vivo microscope imaging of mouse jejunal mucosa, after long ischemia (40 to 50 minutes) with subsequent reperfusion, jejunal tissue suffered irreversible damage. However, short ischemia (15 minutes) suffered reversible damage during reperfusion. Kato et al. [9] showed that when experimental rats underwent selective portal vein occlusion for 75 minutes, all of the rats died within 60 minutes from unclamping. The mortality rate from selective portal vein occlusion in rats increased with a longer duration of ischemia and death occurred earlier for a long period (≥30 minutes) of ischemia rather than for a short periods (≤25 minutes). Although these results came from an animal lab, we assumed that in humans, ischemic time of the intestine is very important for resuscitation. In our case, the ischemic time from CT to decompression was about 3 hours, which may be critical and irreversible.
Clinically, the most common symptom of acute gastric dilatation is emesis. However, the inability to vomit can appear. The reason behind the inability to vomit is not fully understood. It has been suggested that this may be due to the occlusion of the gastroesophageal junction by the distended fundus, which angulates the esophagus against the right crus of the diaphragm, producing a one-way valve [10]. In our case, the patient could not vomit food material, the nasogastric tube was not able to enter the gastroesophageal junction, the gastric lavage tube could not advance over 40 cm from the mouth. CT showed that esophagogastric junction deviated left from the usual position. We thought that the esophagogastric junction became angled and closed, thus decompression using the nasogastric tube or gastric lavage tube failed.
In conclusion, we present a case of abdominal compartment syndrome due to bulimic attack. Clinicians should be aware that binge-eating habits in patients with bulimic nervosa could cause abdominal compartment syndrome due to gastric distension and this may be a potentially fatal condition. At decompression, if ischemic time is long, clinicians should consider the possibility of critical ischemia-reperfusion injury. A prophylactic solution with mannitol and sodium bicarbonate may be able to help minimize reperfusion injury.
Figures and Tables
Fig. 1
Plain abdominal X-ray revealed severely distended stomach with large amount of food materials.
References
1. An G, West MA. Abdominal compartment syndrome: a concise clinical review. Crit Care Med. 2008. 36:1304–1310.
2. Turan M, Sen M, Canbay E, Karadayi K, Yildiz E. Gastric necrosis and perforation caused by acute gastric dilatation: report of a case. Surg Today. 2003. 33:302–304.
3. Vettoretto N, Viotti F, Taglietti L, Giovanetti M. Acute idiopathic gastric necrosis, perforation and shock. J Emerg Trauma Shock. 2010. 3:304.
4. Bailey J, Shapiro MJ. Abdominal compartment syndrome. Crit Care. 2000. 4:23–29.
5. Parsak CK, Sakman G. Acute gastric dilatation and abdominal compartment syndrome as complication of pyloric stenosis. Internet J Gastroenterol [Internet]. 2007. cited 2011 Jan 18. 5(2):Available from: http://www.ispub.com/journal/the_internet_journal_of_gastroenterology/volume_5_number_2_13/article/acute_gastric_dilatation_and_abdominal_compartment_syndrome_as_complication_of_pyloric_stenosis.html.
6. Mohapatra B. Abdominal compartment syndrome. Indian J Crit Care Med. 2004. 8:26–32.
7. McCord JM. Oxygen-derived free radicals in postischemic tissue injury. N Engl J Med. 1985. 312:159–163.
8. Guan Y, Worrell RT, Pritts TA, Montrose MH. Intestinal ischemia-reperfusion injury: reversible and irreversible damage imaged in vivo. Am J Physiol Gastrointest Liver Physiol. 2009. 297:G187–G196.
9. Kato LT, Poggetti RS, Fontes B, Massarollo PC, Younes RN, Heimbecker AM, et al. Evaluation of the mortality rate caused by different periods of selective portal vein occlusion in rats. Acta Cir Bras. 2007. 22:279–284.
10. Luncă S, Rikkers A, Stănescu A. Acute massive gastric dilatation: severe ischemia and gastric necrosis without perforation. Rom J Gastroenterol. 2005. 14:279–283.




 Citation
Citation Print
Print



 XML Download
XML Download