TO THE EDITOR: Primary non-Hodgkin lymphoma (NHL) of bone is a rare disorder [1]. Primary bone lymphoma involving multiple sites is even rarer and in the majority of cases, the diagnosis is diffuse large B-cell lymphoma (DLBCL). Here we report the case of a young patient with unexplained diffuse bone pain that was diagnosed as primary bone lymphoma (B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and Burkitt lymphoma) with multifocal osteolytic lesions.
CASE
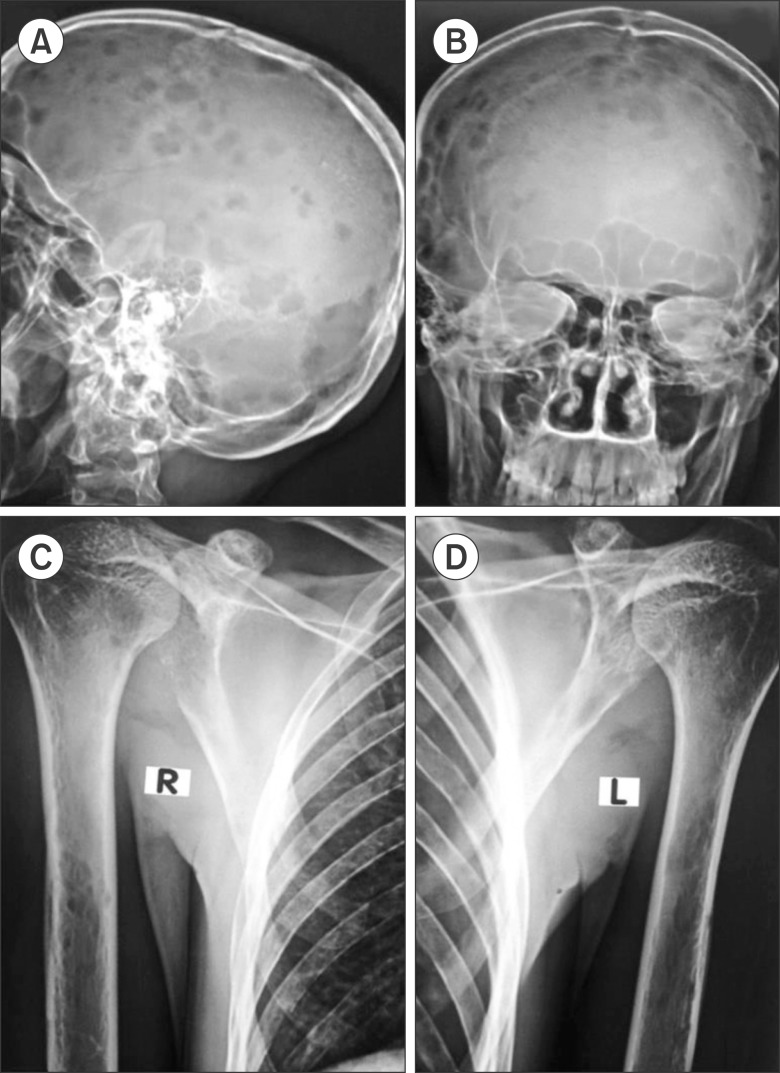
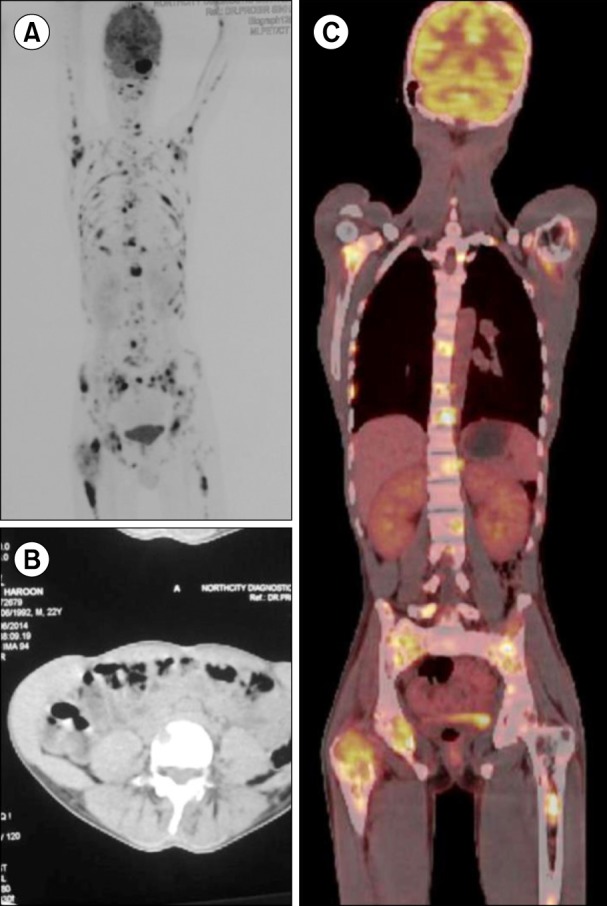
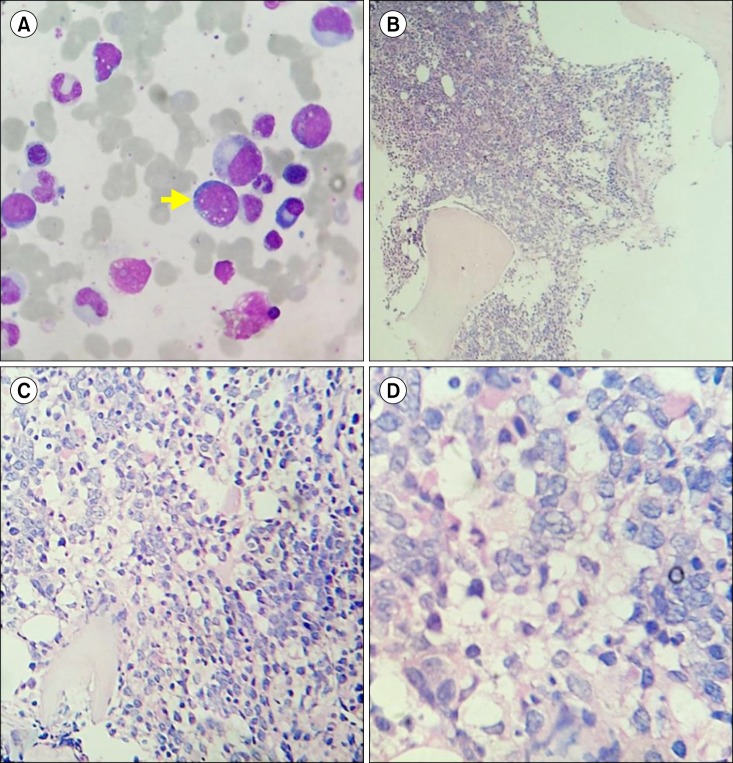
A 22-year-old male was admitted to the orthopedic ward complaining of pain in the right side of his groin. He had experienced difficulty in walking for 3 months prior followed by diffuse bone pain in his whole body and weight loss for 2 months. He had 6 brothers and 1 sister; one of his brothers had been treated for spinal tuberculosis 9 years earlier. He was managed with analgesics and proton pump inhibitors. A skeletal survey (Fig. 1) revealed osteolytic lesions in multiple long and flat bones. Bone scintigraphy with technetium-99 showed high accumulation in the skull, vertebrae, ribs, pelvis, both humeri, and the bilateral femurs (Fig. 2A). A whole-body positron emission tomography-computed tomography (PET-CT) scan (Fig. 2B, C) revealed multiple metabolically active lytic lesions all over the skeletal system. No other metabolically active lesions were observed. Serum carcinoembryonic antigen, alpha-fetoprotein, and prostate-specific antigen levels were normal; the patient's thyroid profile was also normal. The patient was then referred to the hematology department. There was no history of pallor, bleeding, arthralgia or arthritis, nor any history of blood transfusion. On examination, there was mild pallor, but no icterus, pedal edema, or palpable lymph nodes. The liver and spleen were not palpable, but bony tenderness was present. The patient was afebrile and his vital signs were stable. The results of hematologic tests were as follows: hemoglobin 12.1 g/dL, red blood cell (RBC) count 4.28×1012/L, white blood cell count 11.3×109/L, and platelet count 468×109/L. In addition, a peripheral smear showed normocytic, normochromic RBCs, neutrophils 64%, lymphocytes 29%, monocytes 5%, eosinophils 1%, and basophils 1%. Blood biochemistry tests revealed normal serum bilirubin, aspartate transaminase, alanine transaminase, and fasting plasma glucose levels. The following results were also obtained: serum total protein 4.9 g/dL, albumin 2.2 g/dL, globulin 2.7 g/dL, urea 86 mg/dL, creatinine 2.9 mg/dL, uric acid 9.9 mg/dL, sodium 128 mEq/L, potassium 2.7 mEq/L and corrected calcium 14.20 mg/dL. The glomerular filtration rate was 23.17 mL/min, and the antinuclear antibody test result was negative. Serum and urine protein electrophoresis with immunofixation did not reveal any monoclonal paraproteins or light chains. The serum free light chain ratio was 2.54 (renal failure range, 0.37-3.17) and the β2-microglobulin level was 4,900 µg/L. Bone marrow aspiration and a trephine biopsy (Fig. 3) revealed a hypercellular marrow with diffuse infiltration by medium-tolarge cells with scanty cytoplasm, vesicular nuclei with irregular nuclear membranes, and occasional cells with prominent nucleoli. A diagnosis of high-grade non-Hodgkin lymphoma (NHL) was made. The conventional cytogenetic study test results were normal, with a 46, XY karyotype. Immunohistochemistry showed that the tumor cells expressed CD20, CD10, and c-MYC, and were negative for CD3, TdT, BCL-2, and CD34. The MIB-1 labeling index was 90%. Thus, a diagnosis of B-cell lymphoma, unclassifiable, with features intermediate between diffuse large B-cell lymphoma (DLBCL) and Burkitt lymphoma (BL) was made. After counseling, the patient was treated with the R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristin and prednisolone) chemotherapy regimen. A PET-CT after 4 cycles of chemotherapy revealed the presence of residual disease in the trochanteric region of the right femur. The patient was advised to undergo another 4 cycles of R-CHOP. At the end of a total of 8 cycles of R-CHOP, a repeat PET-CT was carried and the patient was determined to be in complete remission.
DISCUSSION
Coley et al. [2] described the criteria for the diagnosis of primary bone lymphoma (subsequently known as Coley's criteria) as early as 1950. The criteria includes: (i) a primary focus in a single bone, (ii) unequivocal histologic proof from the bone lesion, and (iii) no evidence of distant soft tissue or distant lymph node involvement.
Primary lymphoma of bone is a rare entity and is generally a type of NHL; it represents <1% of all NHLs and 5% of all extranodal NHLs [1]. NHL primarily arising in bone is unusual; its peak incidence is in the fifth decade of life. About half of primary NHLs of bone occur in the long bones [3]. It commonly presents with localized bone pain and occasionally a palpable mass [3]. Economopoulos et al. [4] from Greece studied 37 cases of primary lymphoma involving multiple extranodal sites at presentation and found only 1 case of primary bone involvement of DLBCL at multiple sites. In another study from British Columbia of 131 patients with primary bone lymphoma, the most common diagnosis was DLBCL (N=103), followed by follicular lymphoma (N=7), anaplastic large-cell lymphoma of T-cell phenotype (N=4), marginal zone lymphoma (N=4), peripheral T-cell lymphoma (N=2) and lymphoblastic lymphoma (N=2). Among others included (N=9) were 2 cases of Burkitt or Burkitt-like lymphoma and 1 unclassifiable case [5]. In a recent study from Bangalore, India, Singh et al. [6] reported 2 cases of primary bone lymphoma, both of which were diagnosed as DLBCL. Sato et al. [7] reported a case of primary bone lymphoma in a 6-year-old girl presenting with multifocal osteolytic lesions without any systemic symptoms or identifiable non-osseous primary tumors. In a recent study, it was concluded that assessment of bone marrow involvement with fluorine-18-2-deoxy-2-fluoro-D-glucose PET/CT provided a better diagnostic performance in newly diagnosed DLBCL than a bone marrow biopsy [8].
Tanaka et al. [9] reported the case of a 12-year-old boy complaining of fever and migratory arthralgia. A magnetic resonance imaging scan of his whole body showed multiple abnormal signals in his bones. Peripheral blood and bone marrow examinations did not show any abnormalities. A bone and bone marrow biopsy confirmed the diagnosis of BL on further examination. Ahn et al. [10] reported the case of a 2-year-old male Korean child admitted for left hip pain. The patient was diagnosed with intermediate DLBCL/BL based on morphological features intermediate between BL and DLBCL, expression of CD10, BCL6, BCL2, the Ki67 labeling index, and a complex karyotype with 8q34/MYC.
B-cell lymphomas with features intermediate between DLBCL and BL are aggressive lymphomas that have the morphological and genetic features of both DLBCL and BL, but biological and clinical reasons are not included in these categories [11]. Morphologic features are useful in the differential diagnosis of intermediate DLBCL/BL. There are various cellular forms; those resembling BL cells are smaller than typical DLBCL cells and those resembling DLBCL cells are larger than typical BL cells. Immunophenotypically, the cells are akin to BL with positivity for CD19, CD20, CD22, CD79a, CD10 and BCL6. BCL2 expression may be absent, weak, or strong. The Ki67 labeling index shows varying positivity [12]. Perry et al. [13] studied 39 cases of B-cell lymphoma, unclassifiable (B-UCL), presented at a median age of 69 years. The majority (62%) of patients presented with advanced-stage disease and 54% of patients had high (3-5) International Prognostic Index scores. Genetic heterogeneity was observed; 11 patients had 'double-hit' lymphomas with rearrangements of both MYC and BCL2 or BCL6. None of the immunohistochemical or genetic features were predictive of survival and the cases were very aggressive and resistant to chemotherapy. The 2008 World Health Organization classification includes provisional borderline categories for cases that are not clearly DLBCL or BL. This new category is called B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and BL (intermediate DLBCL/BL) [11]. The justification for this new category is the recognition that these tumors are frequently refractory to chemotherapy and the patients have a poor survival rate [14]. Sirelkhatim et al. [15] studied cases with unexplained bone pain and concluded that lymphoma/leukemia should be kept in mind when investigating a case of unexplained bone pain or an unexplained bone lesion, although it is a rare entity.
Our case presented with unexplained diffuse bone pain and, on evaluation, the patient was diagnosed with primary bone lymphoma (B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and BL). The features included: a young age at presentation, serum hypercalcemia, multifocal osteolytic lesions present all over the body, and a rare type of histologic diagnosis. The patient achieved complete remission with R-CHOP chemotherapy. Ramadan et al. [5] treated the cases with CHOP (and R-CHOP in the rituximab era), radiotherapy or a combination of both.
NHL presenting as a primary bone tumor with multifocal disease is extremely rare. Primary bone lymphoma should be considered in the differential diagnosis of bony lesions in young patients. A very high index of suspicion, judicious use of the investigative armamentarium, and awareness of provisional borderline categories like B-cell lymphoma, unclassifiable, with features intermediate between DLBCL and BL will help clinicians to achieve timely diagnosis and management.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download