Abstract
Beta-2 microglobulin is synthesized in all nucleated cells and forms the light chain subunit of the major histocompatibility complex class I antigen. Despite its potential role as a convenient and non-invasive prognostic indicator in malignant lymphomas, the influence of serum β2 microglobulin is currently underestimated, and therapeutic decision making is rarely affected by this marker. Recent studies that included relatively large numbers of patients with specific histologic subtypes showed that serum β2 microglobulin is a potent prognostic marker in malignant lymphomas. In follicular lymphoma, this effort led to the incorporation of serum β2 microglobulin as an indicator in a new prognostic model. In this review, we summarize the current evidence supporting the role of serum β2 microglobulin as a prognostic factor in patients with malignant lymphoma and discuss perspectives for future investigations.
Go to : 
Beta-2 microglobulin (B2M) is synthesized in all nucleated cells and forms the light chain subunit of the major histocompatibility complex (MHC) class I antigen [1, 2]. B2M binds non-covalently to MHC class I molecules and is not directly attached to the cell membrane. Free soluble B2M is detected in body fluids, such as blood, urine, and cerebrospinal fluid, because of its release from the cell surface or cytoplasm [3]. B2M on the cell surface can be substituted by free soluble B2M. Since the identification of B2M in the early 1970s, the serum B2M level has been widely investigated for its prognostic value in multiple hematologic disorders [1, 2]. In multiple myeloma, measurement of serum B2M levels is considered essential for staging and clinical management [4]. Despite the potential role of the serum B2M level as a convenient, non-invasive, and well-established prognostic indicator, its influence is underestimated and rarely affects the therapeutic decision making process in patients with malignant lymphomas. This could be attributed to the fact that previous studies were mostly based on a small number of patients and only a few large-scale multi-center studies have assessed the prognostic value of serum B2M in patients with non-Hodgkin lymphoma (NHL) or Hodgkin lymphoma (HL). However, recent studies that included a relatively large number of patients with specific lymphoma subtypes showed that serum B2M is a potent prognostic marker. In follicular lymphoma, this effort led to the incorporation of serum B2M as an indicator in a new prognostic model [5]. The clinical implications of serum B2M levels in malignant lymphomas need to be re-evaluated to improve the risk stratification of patients in daily practice and in clinical trials. In the present article, we review the current evidence supporting the role of B2M as a prognostic factor in patients with malignant lymphomas and discuss perspectives for future investigations.
Go to : 
The mechanisms underlying the prognostic value of serum B2M in malignant lymphomas remain unclear. A widely accepted hypothesis is that the serum B2M level is directly related to tumor burden, because free soluble B2M, which is released from the cell membrane or cytoplasm, is correlated with cell turnover rate [3]. This was based on previous findings showing that elevated serum B2M was associated with advanced disease stage, elevated lactate dehydrogenase (LDH), and high International Prognostic Index (IPI) risk groups, indicating a greater tumor burden [6, 7]. However, several studies showed that the prognostic significance of serum B2M was independent from that of other validated prognostic factors including stage and serum LDH levels, which reflect tumor burden [5, 7, 8]. This suggests that the serum B2M level is related to specific biologic or tumor microenvironmental features of malignant lymphomas. Recent findings that B2M is involved in cell survival, proliferation, and metastasis in various types of cancer support this hypothesis [3, 9, 10].
Go to : 
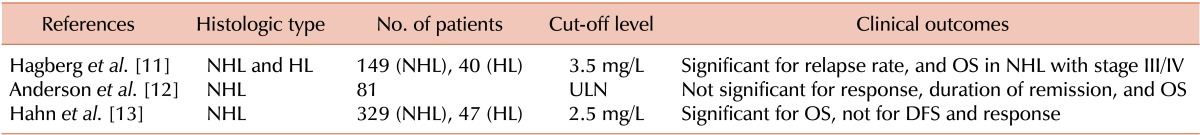
Early studies on serum B2M were performed in heterogeneous study populations that included unselected NHL subtypes (Table 1). One of the earliest studies on serum B2M showed that it is significantly associated with the response to treatment and overall survival (OS) in advanced NHL patients with Ann Arbor stage (III/IV) [11]. However, later studies reported conflicting results regarding the relationship between serum B2M levels and clinical outcomes [12, 13]. The discrepancy among studies could be attributed to the inclusion of various histologic subtypes, considering that clinical outcomes vary according to the histology in patients with NHLs. Moreover, because the magnitude of the prognostic impact of serum B2M may differ among histologic subtypes, the results of these studies cannot be considered informative in an era, in which optimal therapeutic strategies are tailored to each subtype.
Go to : 
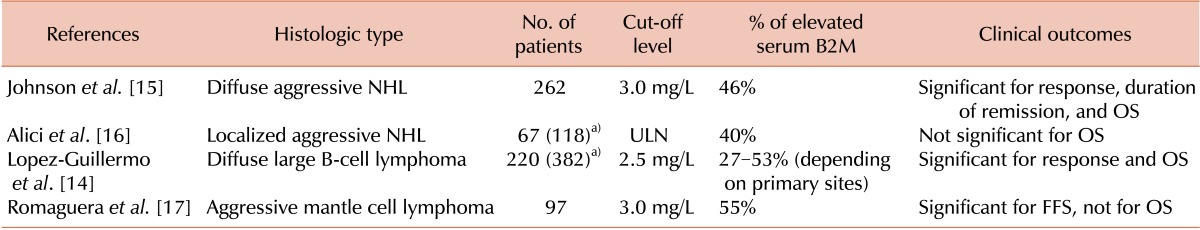
In patients with aggressive NHL, the serum B2M level was reported to be increased in 40-55% of patients (Table 2). Interestingly, a previous study reported that the proportion of patients with elevated serum B2M varied according to the primary site of the disease [14], with significantly lower levels (27%) in patients whose primary disease site was Waldeyer's ring than those in patients with other primary sites (39-53%). Among early studies that included patients with high grade NHL with various histologic subtypes [15, 16], one study suggested that serum B2M is a predictive and prognostic factor for diffuse aggressive NHL [15]; serum B2M was correlated with all evaluated efficacy outcomes, such as response, duration of remission, and OS. Another study aimed at identifying prognostic factors for localized aggressive NHL; however, serum B2M was not associated with OS in univariate and multivariate analyses in patients with localized aggressive NHL [16]. Because the prognostic value of serum B2M was not the major endpoint in this study, serum B2M data were available in only 67 patients, approximately a half of the study cohort, which might have reduced the statistical power of the study for serum B2M.
The prognostic impact of serum B2M in patients with diffuse large B-cell lymphoma (DLBCL) was previously demonstrated in a large single-center retrospective study conducted in Spain [14]. In this study, which included 382 patients, the clinical and biological characteristics and clinical outcomes were defined according to the primary origin of DLBCL. Although only 58% of the study population was available for measurement of serum B2M level, its prognostic significance for OS was retained in multivariate analysis along with IPI and bulky disease. In patients with aggressive mantle cell lymphoma (MCL), the role of serum B2M was investigated in a phase II trial of rituximab plus hyper-fractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (CVAD) alternating with rituximab plus high-dose methotrexate and cytarabine [17]. In this study, which included 97 untreated aggressive MCL patients, the serum B2M level was predictive of response and failure-free survival, but not of OS. The impact of serum B2M on failure-free survival was significant in patients older than 65 years, whereas no difference in failure-free survival according to serum B2M level was detected in patients younger than 65 years. These results suggest the predictive role of serum B2M and a potential interaction between age and serum B2M level in aggressive MCL.
Go to : 
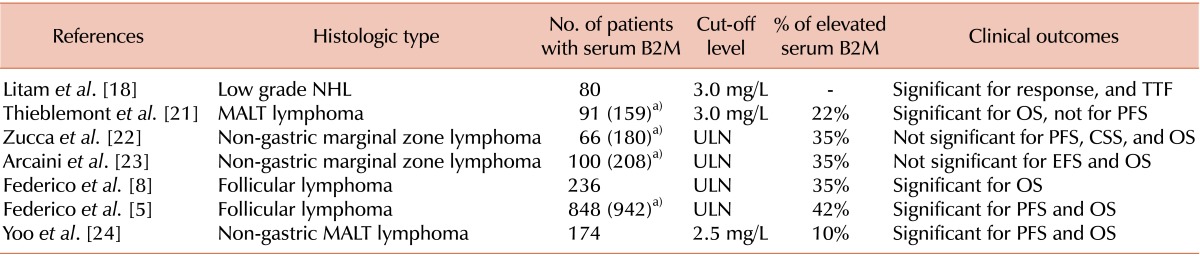
The prognostic relevance of serum B2M in indolent NHL has been relatively well identified from the results of several studies (Table 3). Approximately 12-42% of patients with indolent lymphomas present with elevated baseline serum B2M levels. Litam et al. [18] first reported the prognostic value of serum B2M in low-grade lymphomas and showed that response and time-to-treatment failure were significantly associated with serum B2M. In a cooperative study that resulted in the proposal of the Follicular Lymphoma International Prognostic Index (FLIPI), the serum B2M level was significantly associated with OS in univariate analysis and was recognized as a potential prognostic factor in patients with follicular lymphoma; however, it was not included in multivariate analysis because of a high rate of missing data [19]. The data set of prospective studies performed by the GISL group was later used to examine the relationship between serum B2M and survival outcomes in 236 patients with follicular lymphoma treated with anthracycline-containing regimens [8]. In this study, serum B2M was significantly associated with OS independently of the FLIPI (age, Ann Arbor stage, hemoglobin level, number of nodal area, and serum LDH) with a hazard ratio of 2.9. A follow-up large multi-center retrospective study that included 942 patients was conducted to update the FLIPI and confirmed that serum B2M is a significant prognostic factor for follicular lymphoma [5]. Based on this finding, serum B2M was incorporated into the new prognostic index for follicular lymphoma termed the FLIPI-2, which includes the serum B2M level, longest diameter of the largest involved node longer than 6 cm, bone marrow involvement, hemoglobin level, and age [5]. In a recent phase 3 trial of iodine-131 tositumomab radioimmunotherapy, prognostic models including serum B2M significantly predicted progression-free survival (PFS) and OS of patients with follicular lymphoma, which further validated the prognostic relevance of serum B2M in follicular lymphoma [20]. Furthermore, a statistically significant interaction between the serum B2M level and the investigated regimens suggested the predictive features of serum B2M. Radioimmunotherapy plus cyclophosphamide, doxorubicin, vincristine, and prednisolone (CHOP) was superior to rituximab plus CHOP in terms of PFS in patients with normal serum B2M, whereas no difference in PFS was observed between the two treatment arms in those with elevated serum B2M [20].
In marginal zone lymphoma (MZL), a retrospective analysis conducted to define the prognostic factors in patients with mucosa-associated lymphoid tissue (MALT) lymphoma revealed that serum B2M was significantly associated with OS but not with PFS [21]. However, no association between the serum B2M level and OS was observed in two studies that analyzed non-gastric MZL patients [22, 23], and a relationship with cause-specific survival was found only in the univariate analysis of one study [22]. However, all three studies were limited by the fact that serum B2M data were available for only a half of the study population, which might reduce the statistical power for defining the real association between the serum B2M level and survival outcomes. A recent retrospective study based on prospectively collected data that included 174 patients with non-gastric MALT lymphoma showed that serum B2M was significantly associated with PFS and OS [24]. The impact of serum B2M on survival outcomes was independent of international prognostic index (IPI). The hazard ratio for OS in patients with elevated serum B2M were 26.9; the impact of serum B2M level on OS was much larger than that of IPI (hazard ratio of 2.4). However, a larger sample size is necessary to verify these conflicting results. If the prognostic significance of serum B2M level in MZL is confirmed in future studies, previously established prognostic indices for MZL, such as the IPI or marginal zone lymphoma prognostic index (MZLPI) [25] may need to be updated with the inclusion of serum B2M.
Go to : 
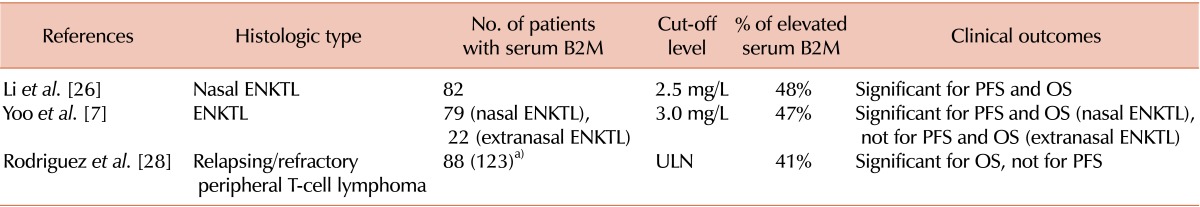
In contrast to the abundance of data regarding serum B2M in B-cell lymphoma subtypes, its prognostic relevance has not been evaluated in extranodal natural killer (NK)/T-cell lymphoma (ENKTL). Two recent studies conducted in Asia evaluated pretreatment serum B2M levels in patients with ENKTL (Table 4) [7, 26]. Although these studies used different cut-off values (2.5 mg/L and 3.0 mg/L), the proportion of patients with elevated serum B2M level was approximately 47% in both studies. A study conducted in China that included patients with nasal ENKTL demonstrated that baseline serum B2M is an independent prognostic factor for PFS and OS, and this remained significant in the multivariate analysis including the IPI [26]. A Korean study that analyzed both nasal and extranasal ENKTL showed that baseline serum B2M level was a significant factor for OS only in nasal ENKTL and not in extranasal ENKTL [7]. The prognostic relevance of serum B2M was verified in multivariate analyses including either the IPI or the Korean Prognostic Index [27], which is another validated prognostic index for ENKTL. Furthermore, the association between the serum B2M level and OS remained significant in a subset analysis for disseminated nasal ENKTL, which has very aggressive clinical features and poor prognosis. The hazard ratios for elevated serum B2M level in terms of OS were 3.3-3.8 in these studies, which is comparable to or higher than those of previously validated prognostic indices (2.2-3.1).
In peripheral T-cell lymphoma (PTCL), the serum B2M level was evaluated in the salvage setting in patients with the refractory disease who underwent autologous stem cell transplantation [28]. This multi-center retrospective analysis included a relatively large number (N=123) of patients and showed elevated serum B2M levels in 41% of patients at the time of transplantation. Serum B2M was identified as the single variable associated with both PFS and OS in a multivariate analysis that included other prognostic factors for PTCL. The investigators in this study developed a prognostic index that included serum B2M and an adjusted IPI that could successfully stratify the survival outcomes.
Go to : 
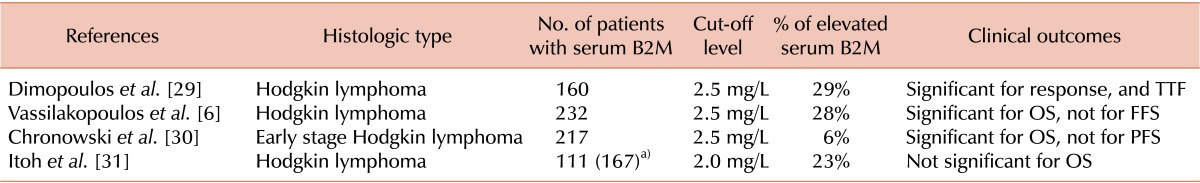
The baseline serum B2M level was elevated in 23-29% of patients with HL in previous studies (Table 5). In contrast to an initial study performed at a single institution that suggested serum B2M as a predictor of treatment outcomes in patients with HL [29], later studies with larger sample sizes failed to demonstrate that serum B2M is predictive of failure-free survival [6, 30]. However, these studies demonstrated that serum B2M is significantly associated with OS [6, 30]. Elevated serum B2M level was the single independent prognostic factor in a study that included only early stage HL patients, whereas in a study including all stages of HL, the hazard ratio of elevated serum B2M level for OS was 3.5. The relevance of serum B2M was recently evaluated in a comprehensive analysis of prognostic factors in HL [31]. In this multi-center retrospective study performed by a Japanese lymphoma study group, serum B2M was not included in the final multivariate model despite its significance in the univariate analysis. The authors suggested that a large proportion of missing data (34% of the overall study population) might account for the inconsistent results. The findings of previous studies indicate that elevated serum B2M level is a poor prognostic factor in terms of OS, but is not predictive of therapeutic outcomes in patients with HL.
Go to : 
The serum B2M level is a powerful prognostic factor in various histologic subtypes of malignant lymphomas as demonstrated in many adequately powered studies. Despite strong evidence of the impact of serum B2M, this marker has not been included in prognostic models for malignant lymphomas except in follicular lymphoma. This is largely due to the substantial proportion of the study population with missing data on serum B2M. Current evidence suggests that there is room for the improvement of index systems and that serum B2M levels could contribute to the provision of enhanced prognostic information. Considering that sensitive prognostic index systems are useful in daily practice and clinical trials for patients with malignant lymphomas, the prognostic relevance of the serum B2M level in previously established prognostic indices should be validated in future studies with large sample sizes, similar to updating the prognostic model for follicular lymphoma. Furthermore, optimal cut-off values of serum B2M for the prediction of clinical outcomes need to be established. B2M as a potential target for cancer therapy is being investigated in the pre-clinical setting, and biologic agents against B2M have shown promising anti-cancer activity [32, 33]. As anti-B2M therapy is expected to be effective in B2M-overexpressing malignancies, the serum B2M level could become a useful biomarker for patients treated with these drugs, particularly in malignant lymphomas, in which its prognostic significance has been well established.
Go to : 
References
1. Peterson PA, Cunningham BA, Berggard I, Edelman GM. β2-microglobulin-a free immunoglobulin domain. Proc Natl Acad Sci U S A. 1972; 69:1697–1701. PMID: 4558655.

2. Grey HM, Kubo RT, Colon SM, et al. The small subunit of HL-A antigens is beta 2-microglobulin. J Exp Med. 1973; 138:1608–1612. PMID: 4128442.
3. Shi C, Zhu Y, Su Y, Chung LW, Cheng T. Beta2-microglobulin: emerging as a promising cancer therapeutic target. Drug Discov Today. 2009; 14:25–30. PMID: 19056512.
4. Munshi NC, Anderson KC, Bergsagel PL, et al. Consensus recommendations for risk stratification in multiple myeloma: report of the International Myeloma Workshop Consensus Panel 2. Blood. 2011; 117:4696–4700. PMID: 21292777.

5. Federico M, Bellei M, Marcheselli L, et al. Follicular lymphoma international prognostic index 2: a new prognostic index for follicular lymphoma developed by the international follicular lymphoma prognostic factor project. J Clin Oncol. 2009; 27:4555–4562. PMID: 19652063.

6. Vassilakopoulos TP, Nadali G, Angelopoulou MK, et al. The prognostic significance of beta(2)-microglobulin in patients with Hodgkin's lymphoma. Haematologica. 2002; 87:701–708. PMID: 12091120.
7. Yoo C, Yoon DH, Jo JC, et al. Prognostic impact of beta-2 microglobulin in patients with extranodal natural killer/T cell lymphoma. Ann Hematol. 2014; 93:995–1000. PMID: 24441948.

8. Federico M, Guglielmi C, Luminari S, et al. Prognostic relevance of serum beta2 microglobulin in patients with follicular lymphoma treated with anthracycline-containing regimens. A GISL study. Haematologica. 2007; 92:1482–1488. PMID: 18024396.
9. Josson S, Nomura T, Lin JT, et al. β2-microglobulin induces epithelial to mesenchymal transition and confers cancer lethality and bone metastasis in human cancer cells. Cancer Res. 2011; 71:2600–2610. PMID: 21427356.

10. Chen CH, Su CY, Chien CY, et al. Overexpression of beta2-microglobulin is associated with poor survival in patients with oral cavity squamous cell carcinoma and contributes to oral cancer cell migration and invasion. Br J Cancer. 2008; 99:1453–1461. PMID: 18841160.
11. Hagberg H, Killander A, Simonsson B. Serum beta 2-microglobulin in malignant lymphoma. Cancer. 1983; 51:2220–2225. PMID: 6189572.
12. Anderson H, Scarffe JH, Swindell R, Crowther D. Serum beta 2-microglobulin in patients with non-Hodgkin's lymphoma. Eur J Cancer Clin Oncol. 1983; 19:327–331. PMID: 6190656.
13. Hahn JS, Lee S, Chong SY, Min YH, Ko YW. Eight-year experience of malignant lymphoma-survival and prognostic factors. Yonsei Med J. 1997; 38:270–284. PMID: 9409190.
14. Lopez-Guillermo A, Colomo L, Jimenez M, et al. Diffuse large B-cell lymphoma: clinical and biological characterization and outcome according to the nodal or extranodal primary origin. J Clin Oncol. 2005; 23:2797–2804. PMID: 15728226.
15. Johnson PW, Whelan J, Longhurst S, et al. Beta-2 microglobulin: a prognostic factor in diffuse aggressive non-Hodgkin's lymphomas. Br J Cancer. 1993; 67:792–797. PMID: 8471438.
16. Alici S, Bavbek SE, Kaytan E, Eralp Y, Onat H. Prognostic factors in localized aggressive non-Hodgkin's lymphoma. Am J Clin Oncol. 2003; 26:1–5. PMID: 12576915.

17. Romaguera JE, Fayad L, Rodriguez MA, et al. High rate of durable remissions after treatment of newly diagnosed aggressive mantle-cell lymphoma with rituximab plus hyper-CVAD alternating with rituximab plus high-dose methotrexate and cytarabine. J Clin Oncol. 2005; 23:7013–7023. PMID: 16145068.

18. Litam P, Swan F, Cabanillas F, et al. Prognostic value of serum beta-2 microglobulin in low-grade lymphoma. Ann Intern Med. 1991; 114:855–860. PMID: 2014946.
19. Solal-Celigny P, Roy P, Colombat P, et al. Follicular lymphoma international prognostic index. Blood. 2004; 104:1258–1265. PMID: 15126323.

20. Press OW, Unger JM, Rimsza LM, et al. A comparative analysis of prognostic factor models for follicular lymphoma based on a phase III trial of CHOP-rituximab versus CHOP + 131iodinetositumomab. Clin Cancer Res. 2013; 19:6624–6632. PMID: 24130072.

21. Thieblemont C, Berger F, Dumontet C, et al. Mucosa-associated lymphoid tissue lymphoma is a disseminated disease in one third of 158 patients analyzed. Blood. 2000; 95:802–806. PMID: 10648389.

22. Zucca E, Conconi A, Pedrinis E, et al. Nongastric marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue. Blood. 2003; 101:2489–2495. PMID: 12456507.

23. Arcaini L, Burcheri S, Rossi A, et al. Nongastric marginal-zone B-cell MALT lymphoma: prognostic value of disease dissemination. Oncologist. 2006; 11:285–291. PMID: 16549813.

24. Yoo C, Yoon DH, Yoon S, et al. Prognostic impact of β2-microglobulin in patients with non-gastric mucosa-associated lymphoid tissue lymphoma. Leuk Lymphoma. 2014; 1–6. PMID: 24913511.

25. Oh SY, Kwon HC, Kim WS, et al. Nongastric marginal zone B-cell lymphoma: a prognostic model from a retrospective multicenter study. Cancer Lett. 2007; 258:90–97. PMID: 17936499.

26. Li ZM, Zhu YJ, Sun J, et al. Serum beta2-microglobin is a predictor of prognosis in patients with upper aerodigestive tract NK/T-cell lymphoma. Ann Hematol. 2012; 91:1265–1270. PMID: 22373550.

27. Lee J, Suh C, Park YH, et al. Extranodal natural killer T-cell lymphoma, nasal-type: a prognostic model from a retrospective multicenter study. J Clin Oncol. 2006; 24:612–618. PMID: 16380410.

28. Rodriguez J, Conde E, Gutierrez A, et al. The adjusted International Prognostic Index and beta-2-microglobulin predict the outcome after autologous stem cell transplantation in relapsing/refractory peripheral T-cell lymphoma. Haematologica. 2007; 92:1067–1074. PMID: 17640855.
29. Dimopoulos MA, Cabanillas F, Lee JJ, et al. Prognostic role of serum beta 2-microglobulin in Hodgkin's disease. J Clin Oncol. 1993; 11:1108–1111. PMID: 8501496.

30. Chronowski GM, Wilder RB, Tucker SL, et al. An elevated serum beta-2-microglobulin level is an adverse prognostic factor for overall survival in patients with early-stage Hodgkin disease. Cancer. 2002; 95:2534–2538. PMID: 12467067.

31. Itoh K, Kinoshita T, Watanabe T, et al. Prognostic analysis and a new risk model for Hodgkin lymphoma in Japan. Int J Hematol. 2010; 91:446–455. PMID: 20198461.

32. Yang J, Zhang X, Wang J, et al. Anti beta2-microglobulin monoclonal antibodies induce apoptosis in myeloma cells by recruiting MHC class I to and excluding growth and survival cytokine receptors from lipid rafts. Blood. 2007; 110:3028–3035. PMID: 17644731.
33. Yang J, Cao Y, Hong S, et al. Human-like mouse models for testing the efficacy and safety of anti-beta2-microglobulin monoclonal antibodies to treat myeloma. Clin Cancer Res. 2009; 15:951–959. PMID: 19188166.
Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download