Abstract
Lichen planus (LP) is a relatively common mucocutaneous disease with autoimmune etiology. Considering its malignancy potential, it is important to define the correct diagnosis, treatment, and clinical follow-up for patients with LP so that the disease is not diagnosed late, thus hindering the chances of curing the disease. This study aims to describe a clinical case of oral squamous cell carcinoma, potentially originated from LP. The patient is undergoing clinical and histopathological follow-up. A 64-year-old Caucasian male patient presented with a proliferative verrucous lesion on the tongue and sought treatment at the School of Dentistry, University of Passo Fundo (UPF), Passo Fundo, Brazil. He claimed the lesion had been present since 1988, and had been initially diagnoses as “oral lichen planus.” The physical exam presented three diagnostic hypotheses: plaque-like oral LP, verrucous carcinoma, and squamous cell carcinoma. After incisional biopsy and histopathological analysis, squamous cell carcinoma was diagnosed, probably originating from oral LP. The case study shows that malignancy from oral LP is possible, which justifies periodic clinical and histopathological follow-up, as well as the elimination of risk factors for carcinoma in patients with oral LP.
Lichen planus (LP) is a chronic mucocutaneous disease characterized by outbreaks and inactivation; it is also an immunological disorder with unknown causal factors1. Stress is the most significant etiological factor for LP. While exacerbation of the lesion by psychological stress and anxiety has not yet been proven, prolonged emotional stress has been shown to enhance the initiation and clinical expression of such a lesion2.
Considering that initial LP and carcinoma lesions are most often asymptomatic, early diagnosis is hindered, and the manifestations can be being overlooked by the patient or health professionals3. Oral biopsy is recommended to confirm the malignancy suspicion of precancerous lesions such as leukoplakia, ulceration, erythroplakia, or LP4.
Oral squamous cell carcinoma is responsible for more than 90% of all oral cancers and is one of the most aggressive diseases worldwide5. The individuals most affected by oral cancer are male, Caucasian, and over 40 years old; the sites most often affected are the tongue and the floor of the mouth6.
The present work aims, through a brief literature review, to present a comprehensive view of squamous cell carcinoma, with the goal of introducing dentists to better diagnostic methods for such lesions. This study also aims to highlight the importance of periodic clinical follow-up of precancerous lesions such as LP by establishing a logical sequence of diagnosis and clinical management of a patient with oral squamous cell carcinoma, probably originating from LP.
A 64-year-old Caucasian male patient with high levels of stress sought dental assistance at the School of Dentistry, University of Passo Fundo, Passo Fundo, Brazil, in 2014. He asked for assessment of a tongue lesion, which had been significantly increasing in size for approximately 6 months.
In the dental visit, he reported pain when swallowing and a lesion in his tongue, which had been diagnosed in 1988 through a histopathological exam as “oral lichen planus.” The patient was not able to recall the place at which the exam was performed; however, he affirmed that the lesion remained the same over the years. Dental follow-up was not performed because the patient did not seek assistance again.
During anamnesis, the patient asserted that he does not consume and never has consumed alcoholic beverages, and that he does not smoke and has never smoked. The first biopsy, performed in 1988, showed bilateral lesions in the jugal mucosa, edge, and dorsum of the tongue. These lesions presented as erythematous depapillated areas associated with leukoplakic areas in the form of grooves that caused burning, with a 3-year evolution. The result of the histopathological exam was “erosive oral lichen planus.”
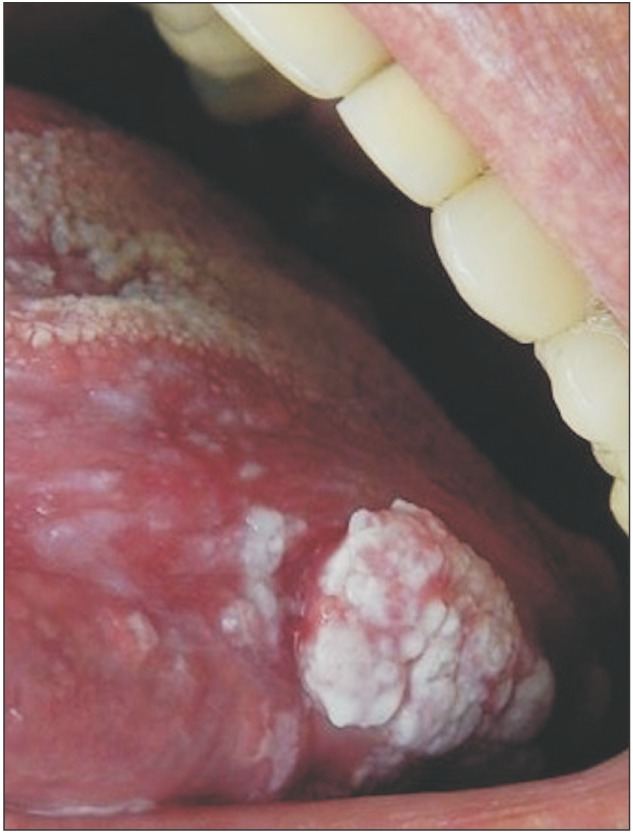
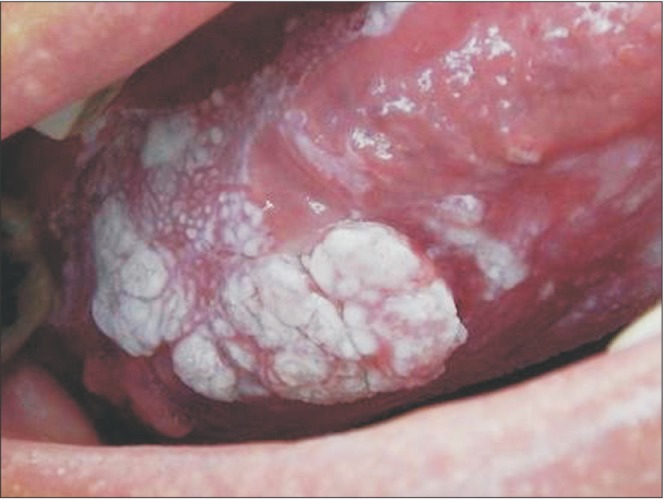
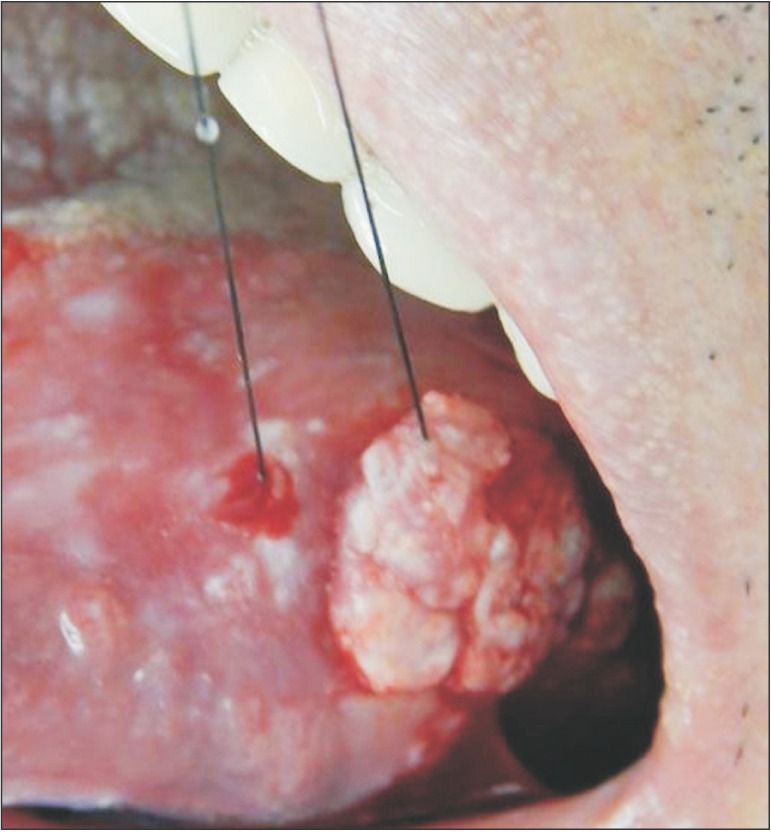
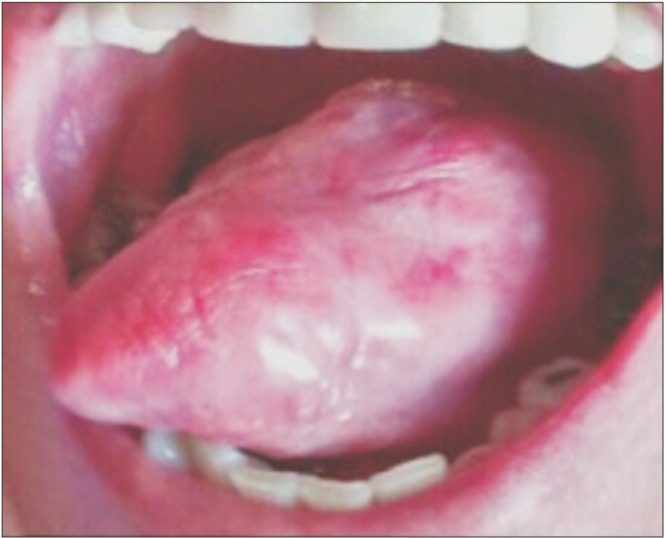
The physical exam in 2014 revealed a lesion on the left edge and in the posterior area of the dorsum of the tongue, extending to the floor of the mouth. The lesion was approximately 10 cm along its largest axis and showed aspects of ulcerated vegetation. It also presented a leukoplakic and erythematous surface and firm consistency, which are clinically suggestive of a malignant lesion of the epithelial lining whose origin is related to oral LP.(Fig. 1, 2) According to the clinical classification of oral and oropharyngeal cancer stages described by the American Cancer Society (2015), the patient was stage III, T3, M0, N0.
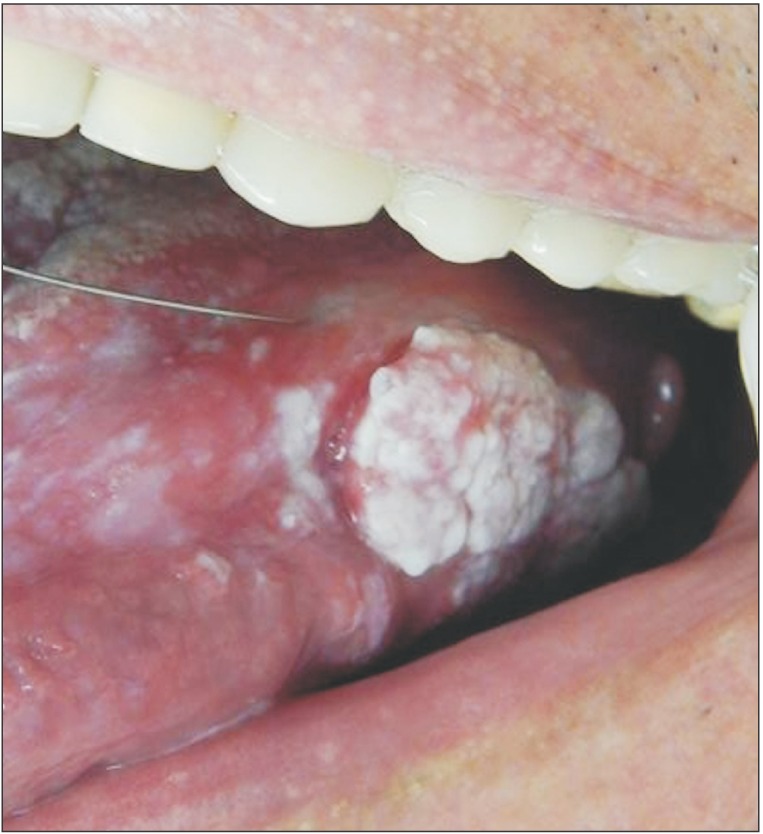
The possible diagnosed were erosive oral LP, squamous cell carcinoma, and verrucous carcinoma. Considering the potential for a malignant lesion, we chose to perform an incisional biopsy of the lesion at the edge of the tongue.
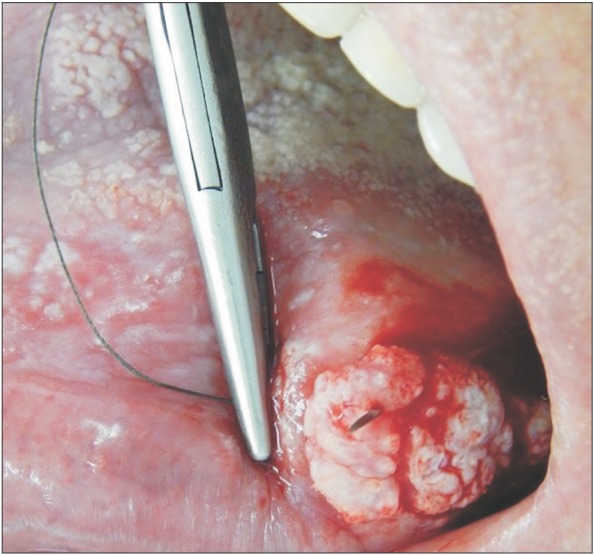
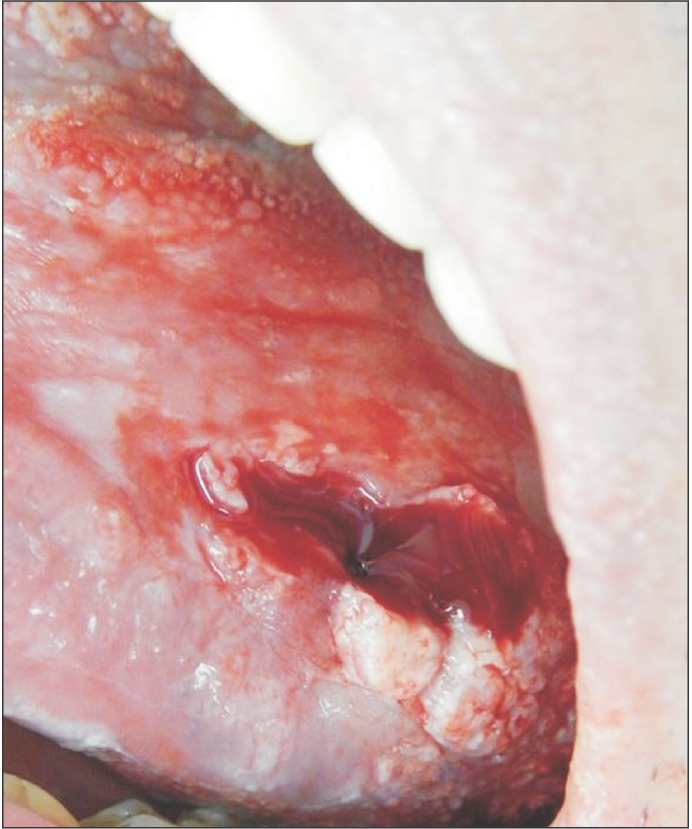
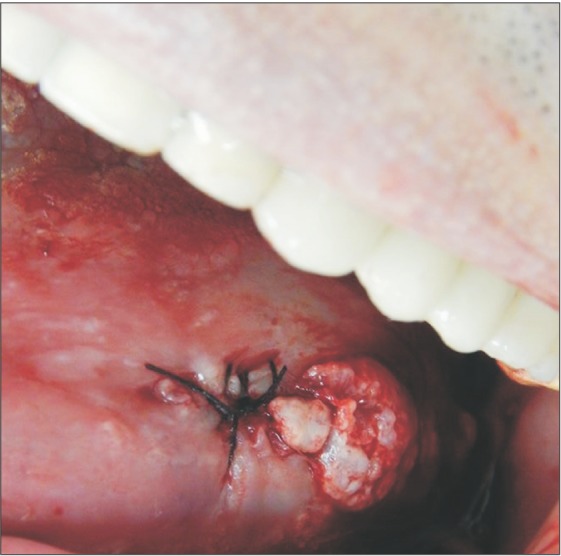
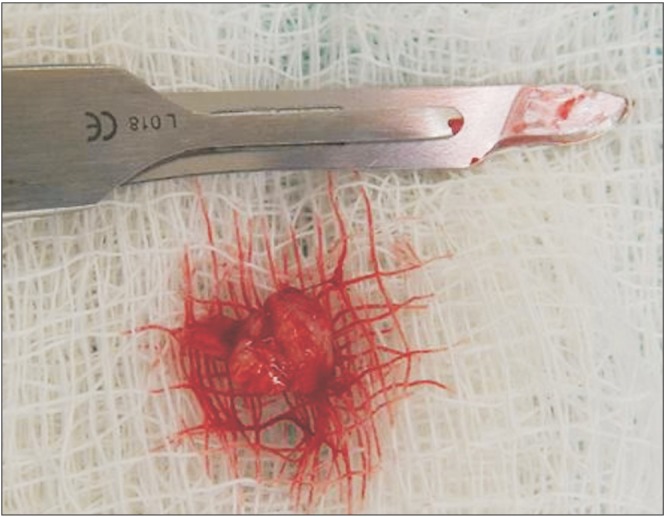
The surgical procedure was performed under local anesthesia around the lesion with mepivacaine and 1:100,000 adrenaline (DFL, Rio de Janeiro, Brazil).(Fig. 3) A simple interrupted suture was initiated at the upper region of the lesion for traction (Fig. 4, 5), favoring a circular incision around the lesion.(Fig. 6) Lastly, suturing was performed.(Fig. 7)
Immediately after the operation, an analgesic was prescribed for pain. In addition, the patient was required to use topical 0.12% chlorhexidine digluconate mouthwash three times a day for seven days after his oral hygiene routine.
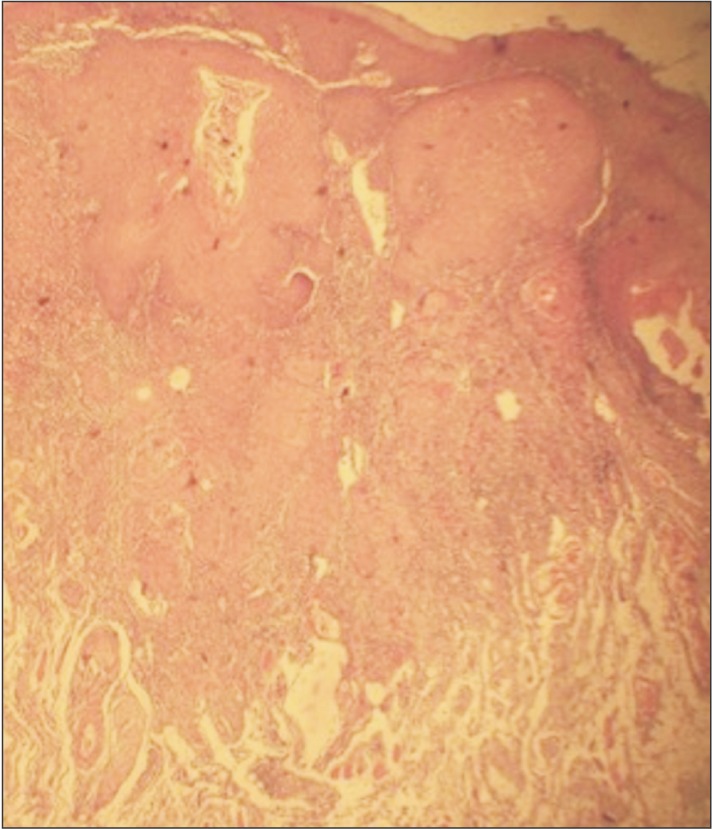
The histopathological exam of the surgical piece showed a squamous cell carcinoma lesion grade I (well differentiated). Upon diagnosis, the patient was referred to an oncological team including a head and neck surgeon, an oral and maxillofacial surgeon, an oncologist, and a speech therapist. The surgery to remove the lesion has not yet been performed because the patient is under chemotherapy and radiotherapy to reduce the lesion size. The patient is also under clinical follow-up with no signs of recurrence. Moreover, after approximately 6 chemotherapy sessions and 35 radiotherapy sessions, significant improvement can be observed, with characteristics of proper healing and absence of papillae on the left lateral edge of the tongue.(Fig. 8, 9, 10) Thus, due to the significant lesion regression caused by radiotherapy and chemotherapy, the medical team did not recommend complete excision of the lesion.
The focus of the present study was to highlight, through a clinical case, the importance of the potential origin of oral squamous cell carcinoma in an LP lesion789. In such a lesion, the epithelium covering the LP becomes more susceptible to oncogenic factors.
The rate of transformation of LP in squamous cell carcinoma in individual studies has ranged from 0% to 3.5%. There might be a higher frequency of malignancy in some sites, such as the tongue, followed by the oral and gingival mucosa7. These findings correspond with those of the present study, considering that the lesion studied was located on the edge of the tongue. The patient described here was a 64-year-old Caucasian man10, consistent with the observation that squamous cell carcinoma is more prevalent in Caucasian men over 50 years. However, the present study disagrees with the conclusion that LP lesions are more common in women11.
In the present case, the lesion initially presented (in 1988) as plaques and white patches, clinical aspects that are characteristic of LP lesions. Later (2014), the lesion presented as a vegetation with leukoplakic and erythematous areas on the edge of the tongue, suggestive of a malignant lesion. This was confirmed by the histopathological exam. Such lesion evolution indirectly confirms the evolution of the initial LP diagnosis for squamous cell carcinoma, which potentially presented as vegetation on the edges of the tongue12.
The lesion described in the present work involved the edge of the tongue, whereas squamous cell carcinoma and LP largely affect the tongue itself131415.
In 2014, the clinical aspect suggesting malignant lesion led to the performance of an incisional biopsy, whose tissue sample was later forwarded for histopathological exam. Definitive diagnosis of squamous cell carcinoma was performed by biopsy, followed by an anatomopathological exam16.
The main etiological factors of oral squamous cell carcinoma are tobacco, alcohol, and constant stress17. Out of these factors, the patient described here only presented stress, which possibly influenced the occurrence of LP and the later occurrence of squamous cell carcinoma. Stress can act as a triggering agent2.
For the patient described here, the squamous cell carcinoma was treated through surgery, radiotherapy, and chemotherapy18. It is worth noting that, in the present study, the patient quit dental follow-up for 26 years. If the patient had continued dental follow-up via periodic visits, followed by biopsies and routine histopathological exams, the lesion might not have progressed to squamous cell carcinoma. Early detection of premalignant lesions and oral cancer is essential for achieving favorable prognoses19.
In conclusion, considering that oral LP is a relatively common disease with cancerization potential, the present study highlights to the scientific community the need for early treatment and follow-up of precancerous oral lesions. Moreover, this work reports the main clinical and histopathological characteristics of oral squamous cell carcinoma, a lesion with high rates of morbidity and mortality.
References
1. Kouhsoltani M, Aghbali A, Shokoohi B, Ahmadzadeh R. Molecular targeting of Her-2/neu protein is not recommended as an adjuvant therapy in oral squamous cell carcinoma and oral lichen planus. Adv Pharm Bull. 2015; 5(Suppl 1):649–652. PMID: 26793611.

2. Krupaa RJ, Sankari SL, Masthan KM, Rajesh E. Oral lichen planus: an overview. J Pharm Bioallied Sci. 2015; 7(Suppl 1):S158–S161. PMID: 26015696.

3. Santos LCO, Batista OM, Cangussu MCT. Characterization of oral cancer diagnostic delay in the state of Alagoas. Braz J Otorhinolaryngol. 2010; 76:416–422. PMID: 20835525.
4. Seoane JM, González-Mosquera A, Velo-Noya J. Oral biopsy in the context of oral cancer and precancer. Av Odontoestomatol. 2008; 24:89–96.
5. Bharanidharan R, Dineshkumar T, Raghavendhar K, Kumar AR. Squamous cell carcinoma of the gingiva: a diagnostic enigma. J Oral Maxillofac Pathol. 2015; 19:267.

6. de Oliveira JMB, Pinto LO, Lima NGM, de Almeida GCM. Oral cancer: assessment of academic dentistry and nursing knowledge as for the risk factors and diagnostic procedures. Rev Bras Cancerol. 2013; 59:211–218.
7. Garcia-Pola MJ, Llorente-Pendás S, González-Garcia M, García-Martín JM. The development of proliferative verrucous leukoplakia in oral lichen planus. A preliminary study. Med Oral Patol Oral Cir Bucal. 2016; 21:e328–e334. PMID: 27031060.

8. Budimir V, Richter I, Andabak-Rogulj A, Vučićević-Boras V, Budimir J, Brailo V. Oral lichen planus: retrospective study of 563 Croatian patients. Med Oral Patol Oral Cir Bucal. 2014; 19:e255–e260. PMID: 24608217.
9. Shi W, Yang J, Li S, Shan X, Liu X, Hua H, et al. Potential involvement of miR-375 in the premalignant progression of oral squamous cell carcinoma mediated via transcription factor KLF5. Oncotarget. 2015; 6:40172–40185. PMID: 26474386.

10. Cabello BT, Sazo BN, Salgado FA, Martínez RB. Squamous cell carcinoma of the lip survival rate. Rev Med Chil. 2015; 143:847–855. PMID: 26361020.
11. Mankapure PK, Humbe JG, Mandale MS, Bhavthankar JD. Clinical profile of 108 cases of oral lichen planus. J Oral Sci. 2016; 58:43–47. PMID: 27021539.

12. Pedron IG, dos Santos ESR, Aburad A, Tortamano IP, Adde CA. Squamous cell carcinoma: diagnosis and first behavior. J Health Sci Inst. 2006; 24:237–241.
13. Selvamani M, Yamunadevi A, Basandi PS, Madhushankari GS. Prevalence of oral squamous cell carcinoma of tongue in and around Davangere, Karnataka, India: a retrospective study over 13 years. J Pharm Bioallied Sci. 2015; 7(Suppl 2):S491–S494. PMID: 26538904.

14. Agha-Hosseini F, Mirzaii-Dizgah I. Serum and saliva collagenase-3 (MMP-13) in patients with oral lichen planus and oral squamous cell carcinoma. Med J Islam Repub Iran. 2015; 29:218. PMID: 26478876.
15. Oh MS, Kang SH, Kim HJ, Zhenglin Z, Ryu JI, Nam W, et al. Overall five-year survival rate in squamous cell carcinoma of oral cavity. J Korean Assoc Oral Maxillofac Surg. 2009; 35:83–88.
16. De Carli JP, Trentin MS, Linden MSS, Bós ÂJG, Pedro REL, Silva SO. Oral squamous cell carcinoma of great extent: protocol diagnosis. Odonto. 2010; 18:67–71.
17. Hande AH, Mohite DP, Chaudhary MS, Patel M, Agarwal P, Bohra S. Evidence based demonstration of the concept of ‘field cancerization’ by p53 expression in mirror image biopsies of patients with oral squamous cell carcinoma: an immunohistochemical study. Rom J Morphol Embryol. 2015; 56:1027–1033. PMID: 26662135.
18. Brener S, Jeunon FA, Barbosa AA, Grandinetti HAM. Carcinoma de células escamosas bucal: uma revisão de literatura entre o perfil do paciente, estadiamento clínico e tratamento proposto. Rev Bras Cancerol. 2007; 53:63–69.

19. Yardimci G, Kutlubay Z, Engin B, Tuzun Y. Precancerous lesions of oral mucosa. World J Clin Cases. 2014; 2:866–872. PMID: 25516862.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download