Abstract
Adenoid cystic carcinoma is a rare epithelial tumour, and comprises about 1% of all malignant tumours of the oral and maxillofacial region. It is a malignant tumour which may develop in the trachea, bronchus, lungs or mammary glands, in addition to the head and neck region. Occurrences in the head and neck are mostly detected in the major salivary gland, oral cavity, pharynx and paranasal sinus where it presents as a slow growing firm nodular swelling. The aim of the article is to highlight the unique presentation of adenoid cystic carcinoma as a solitary ulcer on the floor of the mouth.
Go to : 
Adenoid cystic carcinoma was first described by Billroth in 1859 and called "cylindroma" due to its characteristic histologic appearance1. In 1953, Foote and Frazell2 renamed the lesion as adenoid cystic carcinoma. Adenoid cystic carcinoma is a malignant salivary gland tumour characterized by a deceptive histologic pattern, indolent, locally invasive growth with high propensity for perineural invasion, local recurrence and distant metastasis. These uncommon neoplasms account for fewer than 1% of all head and neck malignancies and fewer than 10% of all salivary neoplasms. They make up 15%-30% of submandibular gland tumours, 30% of minor salivary gland tumours, and 2%-15% of parotid gland tumors1. It is defined by the World Health Organization as "a basaloid tumour consisting of epithelial and myoepithelial cells in various morphological configurations, including tubular, cribriform and solid patterns. It has a relentless clinical course and usually a fatal outcome"3. The present case report is unique as it shows presentation of adenoid cystic carcinoma as a solitary ulcer on the floor of the mouth rather than the classical nodular swelling.
Go to : 
A 56-year-old male patient presented with a chief complaint of an ulcer in the left floor of the mouth for one week. He gave a history of pain of respect on the left side of the jaw which was continuous, dull, throbbing, and radiating to the ear of the same side. The patient had under gone extraction for tooth #18 but the pain still persisted. Thereafter, he noticed an ulcer on the left floor of the mouth which was initially small and increased to the present size. There was no relevant history of injury, traumatic surgery or biopsy. There was no history of bleeding or discharge, but he did have difficulty eating and speaking. There was no history of smoking, quid chewing or alcohol consumption.
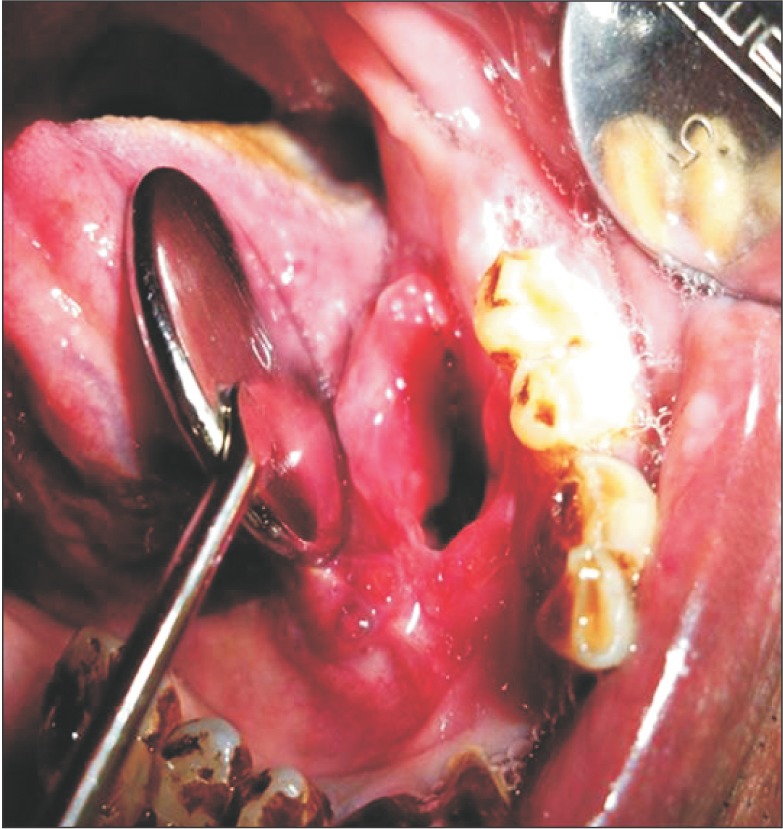
Intraoral examination of the soft tissues, the buccal mucosa, labial mucosa, tongue, and palate showed no abnormalities, but there was a solitary ulcer on the left floor of the mouth. Examination of gingival status revealed his oral hygiene status to be poor, with severe stains and calculus deposits. On hard tissue examination, healing socket was present in respect to tooth #19. On local examination of the lesion, inspectory findings revealed a solitary ulcer presenting as a mucosal tear involving the mucosa and connective tissue extending mesially from left side of the lingual frenum and distally to an area corresponding to the extracted tooth #19 region. The ulcer was seen in close approximation to the left alveolar ridge. It was oval in shape, 3×2 cm at its widest point, and 3 cm deep. The margins were everted, the edges of the ulcer were sloping, and the floor was not evident. The colour was the same as that of the adjacent mucosa, but erythematous at the periphery of the ulcer. No bleeding or discharge was evident. Photographs were taken with due consent from the patient.(Fig. 1) On palpation of the site, the size and the extent of the ulcer were confirmed. The ulcer was mildly tender on palpation, with sloping edges. The margins and the base of the ulcer were mildly indurated in a posterior aspect. The ulcer rested on a muscular base.
Based on the history given by the patient and the clinical features, a provisional diagnosis of carcinoma of the floor of the mouth T4aN0M0, chronic generalized periodontitis, partially edentulous in respect to tooth #7, #5, #3, #13, #14, #15, #24, #19, #25, and #31 was made. Lesions presenting as solitary ulcers on the floor of the mouth were considered in the differential diagnosis. Traumatic ulcers are most common on the tongue, lips, mucobuccal fold, gingiva and palate. They present as solitary ulcers with raised reddish borders and white necrotic floors with associated history of trauma. On the floor of the mouth, they are usually due to calculus or sharp denture margins. Major recurrent apthous ulcers are mostly solitary, deep, and painful, with smooth margins and a reddish halo and may persist for months with history of reoccurrences.
Habit associated lesions seen in quid chewers may also form ulcerations at the region of quid placement. Odontogenic infections may be associated with an ulcer that may serve as a cloacal opening of a sinus draining a chronic alveolar abscess. They are mostly seen in the palate and sublingual or vestibular areas. Pus exudation with regionally involved teeth would help in differentiation. Other conditions such as acute necrotic ulcerative gingivostomatitis or gangrenous stomatitis usually present as necrotic sloughing ulcerative lesions diffusely involving the gingiva. Solitary ulcers are also seen in nonodontogenic systemic diseases such as uncontrolled diabetes mellitus, blood dyscrasias (leukemia, sickle cell anemia), gastrointestinal and immunocompromised individuals and autoimmune conditions (pemphigus, pemphigoid, erythema multiforme, epidermolysis bullosa). The ulcers in such conditions are well demarcated, painful, and shallow with an erythematous halo and a grey necrotic floor, usually in the marginal and interdental gingiva. Salivary gland disorders such as adenoid cystic carcinoma, mucoepidermoid carcinoma, mucous adenocarcinoma, Warthin tumor and necrotising sialometaplasia are seen as solitary palatal ulcers.
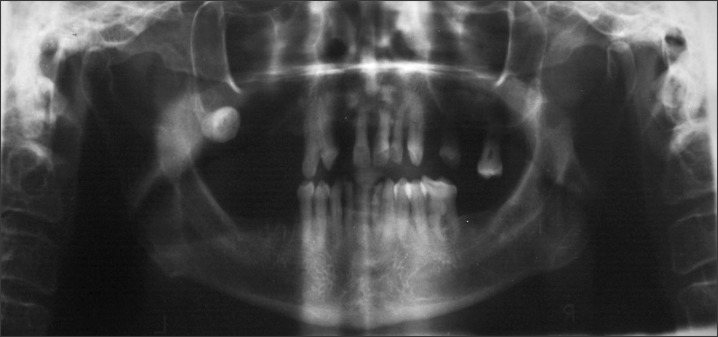
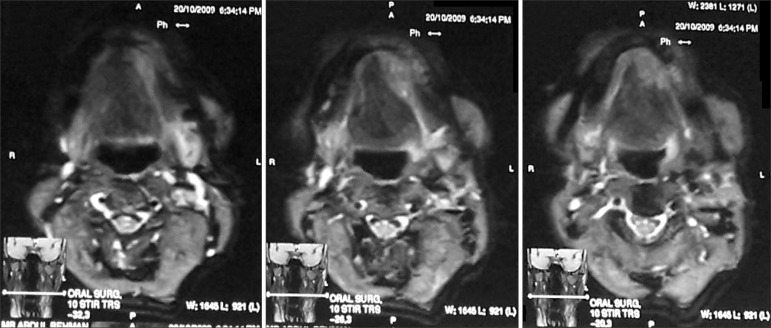
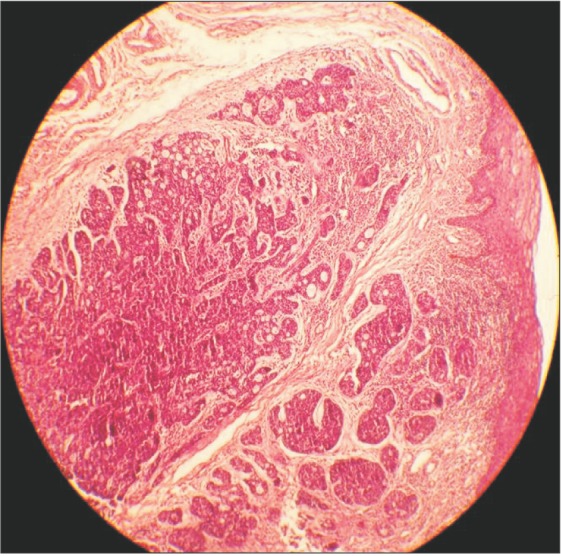
An orthopantomogram and a mandibular crossectional occlusal radiograph were taken and showed no abnormality in the region of interest.(Fig. 2) A magnetic resonance image was taken, and the axial sections showed a T1-weighted image of a mass of medium intensity extending from the midline of the mandible to the premolar region on the left side.(Fig. 3) An incisional biopsy was performed and the histopathologic report showed the presence of uniform basaloid cells arranged in the form of solid islands, along with a cribriform pattern at some places. The tumour cells were found to be infiltrating the adjacent tissues. Normal salivary gland tissue was found adjacent to the connective tissue.
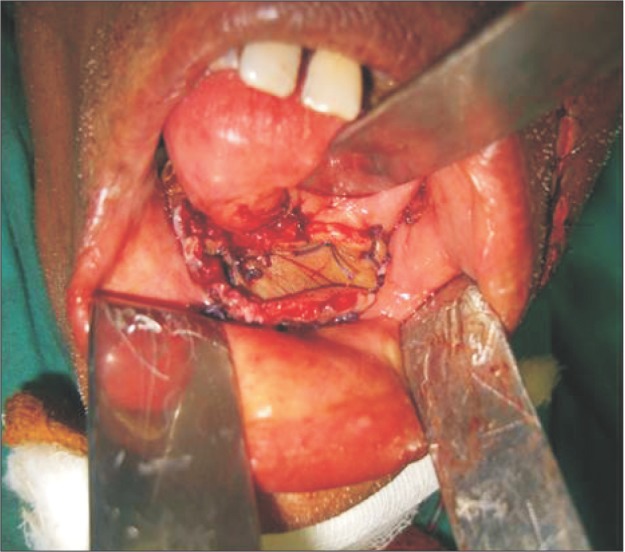
Based on the clinical features and the histopathologic report, a final diagnosis of adenoid cystic carcinoma T4aN0M0 was made.(Fig. 4) Wide excision of the lesion with hemimandibulectomy was completed.(Fig. 5) The patient was then referred for further radiotherapy.
Go to : 
Adenoid cystic carcinoma is a malignant tumour with a deceptively benign histological appearance, characterized by indolent locally invasive growth with a high propensity for local recurrence and distant metastasis. According to the literature, adenoid cystic carcinoma mostly occurs in the fifth or sixth decade of life. Gender predilection is an inconsistent feature in the literature; however, it is slightly more common in women than in men4,5. Although the palate is the most commonly involved intraoral site, other commonly involved areas are the major and minor salivary gland regions, the floor of the mouth, the tongue and the gingiva (in decreasing order). Adenoid cystic carcinoma usually presents as a slowly growing, firm, unilobular mass in the glands. However, in our case it presented as a solitary ulcer on the floor of the mouth. The clinical course is characterized by an initial period of slow and indolent growth that is usually asymptomatic, although bone invasion or perineural spread can cause pain or hypoesthesia5. The tumour usually spreads through a haematogenous route with distant metastasis. Cervical node metastasis is rare6.
Adenoid cystic carcinoma of the salivary glands is a malignant epithelial tumour with bidirectional differentiation towards luminal (ductal) and abluminal (myoepithelial and basal) cells. The tumour is composed of basaloid cells with small, angulated, and hyper chromatic nuclei and scant cytoplasm arranged into three prognostically significant patterns: cribriform, tubular, and solid. The combination of surgery and postoperative radiation therapy has improved locoregional control of the disease. Despite this achievement, late local recurrence and distant metastasis rates remain high and may occur decades after initial diagnosis5.
In its most frequently seen histological pattern, the majority of the cells are small and darkly stained with scant cytoplasm. The cells are arranged in nests or sheets that are fenestrated by round or oval spaces, creating the characteristic "cribriform" design. Occasionally, the tumours have a predominantly solid cellular growth with a basaloid or anaplastic appearance that has few, if any, fenestrations. The solid variant often demonstrates small areas of necrosis. Tubular structures with minimal stratification of the lining epithelium are often mixed with the classic cribriform and solid areas6. Several authors have suggested that a solid histological pattern indicates a more serious prognosis than a cystic pattern. Stewart7 first noted the increased aggression suggested by the solid variant, although he credits the initial observation to Patey and Thakray8.
Little information exists on cytogenetic abnormalities in salivary gland neoplasms, but in adenoid cystic carcinoma, anomalies in the terminal part of the 6q and 9p chromosomes have been reported. Recent studies have demonstrated a high incidence of loss of heterozygosity at chromosome 6q23-359. The differential diagnosis of adenoid cystic carcinoma in cludes tumours that also exhibit tubular and cribriform structures, such as polymorphous low-grade adenocarcinoma, tumours with basaloid cellular morphology, such as basal cell adenoma, and basal cell adenocarcinoma and tumours with a dual population of ductal and myoepithelial cells, such as pleo morphic adenoma8.
Possible treatments of adenoid cystic carcinoma include four different modalities: surgical therapy, radiotherapy, chemotherapy and combined therapy. Surgical excision with wide margins is the treatment of choice10. However, there is still controversy regarding the adjuvant treatment of this tumour. Avery et al.11 recommend postoperative radiation since radiation often produces tumour regression and relieves symptoms. Postoperative radiotherapy is also indicated when the tumour location is close to the base of the cranium with the presence of neck lymph node metastasis and perineural invasion, with a solid histological subtype, and for recurrent tumours11. Adenoid cystic carcinoma shows a limited response to chemotherapy, and it is believed that this lack of a response is due to its slow growth rate. However, a study by Alcedo et al.12 opened up a new possibility in the pharmacological treatment of this tumour by demonstrating its response to imatinib mesylate, a potent inhibitor of KIT gene tyrosine kinase, which is an enzyme involved in the pathogenesis of the tumour. However, more studies are needed to confirm its effectiveness. Distant metastasis can occur even decades after the primary tumour has been treated and after having achieved adequate locoregional control. This leads to low long-term survival rates. Some authors even consider this tumour to be an incurable disease12.
Furthermore in the setting of inoperable, incompletely resected or recurrent tumours, outcomes after conventional therapy are dismal and remain a therapeutic challenge. Some authors have applied other forms of radiotherapy, particularly neutron irradiation, aiming to improve treatment results13. However, adenoid cystic carcinoma remains incurable because further improvements in local-regional control are not likely to impact survival due to the high number of distant failures.
The present case differs from usual cases of adenoid cystic carcnioma with respect to site and presentation, making it a rare entity. Thus, the aim of this article is to emphasise that though the literature states that adenoid cystic carcinoma ulcerates the superficial lesional mucosa, it can also present as an ulcerative lesion.
Go to : 
References
2. Foote FW Jr, Frazell EL. Tumors of the major salivary glands. Cancer. 1953; 6:1065–1133. PMID: 13106826.

3. El-Naggar AK, Huvos AG. Adenoid cystic carcinoma. In : Barnes L, Eveson JW, Reichart P, Sidransky D, editors. Pathology and genetics of head and neck tumors. World Health Organization classification of tumors. Lyon: IARC Press;2005.
4. Bianchi B, Copelli C, Cocchi R, Ferrari S, Pederneschi N, Sesenna E. Adenoid cystic carcinoma of intraoral minor salivary glands. Oral Oncol. 2008; 44:1026–1031. PMID: 18396442.

5. Jaso J, Malhotra R. Adenoid cystic carcinoma. Arch Pathol Lab Med. 2011; 135:511–515. PMID: 21466371.

6. Spiro RH. Distant metastasis in adenoid cystic carcinoma of salivary origin. Am J Surg. 1997; 174:495–498. PMID: 9374223.
7. Stewart J. Carcinoma of salivary glands showing the cylindroma pattern. Br J Surg. 1961; 49:241–245. PMID: 13917137.

8. Patey DH, Thackray AC. The treatment of parotid tumours in the light of a pathological study of parotidectomy material. Br J Surg. 1958; 45:477–487. PMID: 13536351.

9. Rapidis AD, Givalos N, Gakiopoulou H, Faratzis G, Stavrianos SD, Vilos GA, et al. Adenoid cystic carcinoma of the head and neck. Clinicopathological analysis of 23 patients and review of the literature. Oral Oncol. 2005; 41:328–335. PMID: 15743696.
10. Norberg-Spaak L, Dardick I, Ledin T. Adenoid cystic carcinoma: use of cell proliferation, BCL-2 expression, histologic grade, and clinical stage as predictors of clinical outcome. Head Neck. 2000; 22:489–497. PMID: 10897109.

11. Avery CM, Moody AB, McKinna FE, Taylor J, Henk JM, Langdon JD. Combined treatment of adenoid cystic carcinoma of the salivary glands. Int J Oral Maxillofac Surg. 2000; 29:277–279. PMID: 11030399.

12. Alcedo JC, Fábrega JM, Arosemena JR, Urrutia A. Imatinib mesylate as treatment for adenoid cystic carcinoma of the salivary glands: report of two successfully treated cases. Head Neck. 2004; 26:829–831. PMID: 15350030.

13. Huber PE, Debus J, Latz D, Zierhut D, Bischof M, Wannenmacher M, et al. Radiotherapy for advanced adenoid cystic carcinoma: neutrons, photons or mixed beam? Radiother Oncol. 2001; 59:161–167. PMID: 11325445.

Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download