II. Materials and Methods
1. Patients
This study dealt with patients diagnosed with oral cancer and subjected to radical resection of lesion at the Department of Oral and Maxillofacial Surgery, Pusan National University Hospital between March 1998 and March 2008. The inclusion criteria were as follows:
1) Patient confirmed to have oral cancer through biopsy.
2) Patient who did not have metastasis at the first diagnosis.
3) Patient who did not receive any treatment for the primary tumor site in other hospitals.
4) Patient with no history of malignant tumors on any other part.
5) Patients operated by the same surgical team with similar treatment protocol.
6) Patient identified to be in a state of survival after diagnosis
for 5 years.
The patients were interviewed, and their medical records were analyzed. Patients with no more follow-up visits after 5 years were contacted to confirm their survival by calling; when their survival was confirmed, additional clinical and radiological examinations were performed to determine the factors impacting the long-term survival rate.
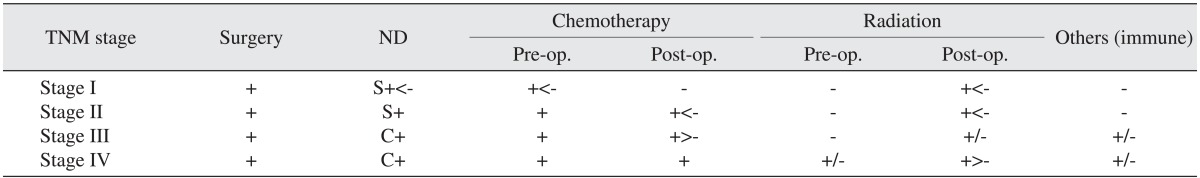
During the observation period, the treatment strategy of our department could be changed for others depending on the general condition of the patient. Note, however,that chemotherapy prior to operation had been done for 2 cycles routinely, followed by radical wide excision and neck dissection as necessary. Likewise, chemotherapy and radiation therapy may be needed depending on the histopathologic results.(
Table 1) Through the analysis of these records, we tried to review the treatment outcome and evaluate the detailed problems of all oral cancer patients. In addition, through long-term follow-up, analysis of the factors impacting the 5-year survival rate of oral cancer patients was performed.
Table 1
Protocol for the treatment of head and neck malignancy in the Department of Oral and Maxillofacial Surgery at Pusan National University Hospital

2. Methods
The final 37 patients were analyzed by gender, age, degree of alcohol drinking, smoking status, primary site, type of carcinoma, histopathologic grade, stage, neck dissection, combination therapy, recurrence, and cervical lymph node metastasis; we then analyzed how these factors impacted the patient's 5-year survival rate for correlation.
The TNM stage was classified according to the TNM classification for the lip and oral cavity of the American Joint Committee on Cancer Guide (7th edition). Each of the impacting factors was compared and reviewed by making individual tables for each factor. Considering the status of patients from the point when follow-up is over, survival was assumed when the patient had been alive for 5 years from diagnosis; if the follow-up was paused, or the patient was discharged due to other reasons, such was treated as censored data.
As a retrospective clinical study, this study was approved by the Pusan National University Dental Hospital Institutional Review Board (IRB No. PNUDH-2013-007).
Go to :

IV. Discussion
Out of the 37 oral cancer patients included in this study, 23 (62.2%) were male and 14 (37.8%) were female. This represents a 1.6 : 1 ratio, showing a comparable result with the United States male oral cancer percentage ratio of 60.2% and Funk et al.
8 and Kim et al.
9 reported percentage ratio of 65.7% males and 34.3% females. In this study, however, gender was not related to the 5-year survival rate.
In this study, 13 (35.1%) patients were heavy smokers and 17 (46.0%) were habitual drinkers, showing lower percentages compared to the previous reports (57-65%)
9-
14. This seemed to be due to the bias of patients in the interview at the time of hospitalization; hence the difficulty in revealing that smoking and alcohol may have significant effects on the survival rate in this report.
According to the research of Krolls and Hoffman
15, oral cancer frequently strikes those in their 40s-70s. According to Kim et al.'s report
9, males in their 70s and females in their 60s were affected mostly by oral cancer. In this study, out of all the age distributions of oral cancer patients, patients in their 50s (11 patients in their 50s with the average)--with average age of 55.5 years (±13.9)--were most affected. There were 11 (29.7%) patients under 40 years and 26 (70.3%) patients over 50 years, resulting in a higher number of older patients. Note, however, that 26- and 28-year-old patients with tongue cancer were observed in these cases as well. It is the form of oral cancer that can affect even a healthier and younger age group as well. Out of the 37 patients, the oldest patient was an 81-year-old male who did not show any recurrence of the primary site or cervical lymph node metastasis after 1 year since operation but expired due to general weakness and respiratory disease; thus, this case was processed as censored data. There were no significant changes in the survival rate of oral cancer patients by age group; as factors that can have an effect on the 5-year survival rate among the elderly and patients who had systemic diseases, however, limitations in choosing the treatment method due to general condition, age, patient-related complications, and deaths caused by associated diseases may be considered
16.
Based on the histopathologic examination of specimen after operation, SCC accounted for more than 80% of the results, showing similar ratio of generally known oral tumors
17. In this study, the survival rate of patients with cancer occurring on the FOM was lowest at 50.0% for 5 years. This seems to be due to the difficulty in terms of surgical approach and cervical lymph metastasis tendency on both sides; considering these facts, the primary lesion's location can be regarded as a perilous factor that can affect survival.
According to Shah and Patel
17, oral cancer's TNM patient stage ratio was 37% (stage I), 36% (stage II), 18% (stage III), and 9% (stage IV). In domestic research, Kim et al.
9 reported that, out of 180 oral cancer patients, 31 (17.2%) cases belonged to stage I, 24 (13%), to stage II, 14 (7.8%), to stage III, and 111 (61.7%), to stage IV. The difference in prevalence between types of stage was deemed attributable to the patients' residence, financial status, and cultural differences; hence the need for additional epidemiological research on the patient's economic activity or education level and oral health policy by location. In inferring the reason for the high prevalence level of patients in early stages (stage I, II) and terminal stage (stage IV) in this study, patients diagnosed with oral cancer in the earlier stages were mostly transferred to our hospital because such can be found out by chance when they visit the local clinic due to different chief complaints, and others, by visiting the hospital doctors after suffering from edema and pain since the cancer has already progressed.
The concordance of cTNM and pTNM was 83.8%, relating to the reliability of the magnetic resonance imaging (MRI), computed tomography (CT), and positron emission tomography-CT (PET-CT) examinations before the operation to evaluate the primary site, cervical metastasis, and distant metastasis. In this study, PET-CT examination was mainly used for the evaluation of cervical lymph node metastasis or follow-up check before operation.
According to the reports of Goshen et al.
18, PET-CT examination has 88% accuracy, 100% sensitivity, and 77% specificity. When it came to negative predictive value from patients who showed metastasis, it was found to be 100%. In Nahmias et al.'s research
19, 192 out of 1,678 lymph nodes had histopathologic metastasis, with sensitivity and specificity to N0 reported to be 79% and 82%, respectively; those to N+ were 95% and 25%, respectively. Thus, sensitivity and specificity for cervical metastasis were concluded to be 48% and 99%, respectively. In addition, PET-CT ensures patients' comfort when setting the irradiation site for radiation therapy
20.
In this study, there were 3 patients who showed signs of occult neck nodal metastasis, with 4 patients testing positive (cN+) in the clinical examination but pathologically negative (pN0) for cervical lymph node. Moreover, the sensitivity of the cervical lymph node examination before operation was 76.9%, and its specificity was 83.3%. From cN0, the potential diagnostic methods for cervical lymph node metastasis (palpation, X-ray, fine needle aspiration cytology) are affected by the number of lymph nodes removed and histological techniques for the examination of lymph nodes
21. Therefore, when we perform surgery and treatment for the oral cancer patient, we should consider PET-CT or other examinations, cognizant of the need for neck dissection for occult lymph node metastasis as one of the complications of surgery. Likewise, clinical follow-up and careful review of the clinical findings and diagnostic examination at an appropriate time after the operation are very important.
Cho and Kim
22 reported a 54% 5-year survival rate among oral cancer patients during the period 1991 to 1996. Kim et al.
9 found the 5-year survival rate of oral cancer patients from 1999 to 2006 to be 57.7%. From the report of Lee et al.
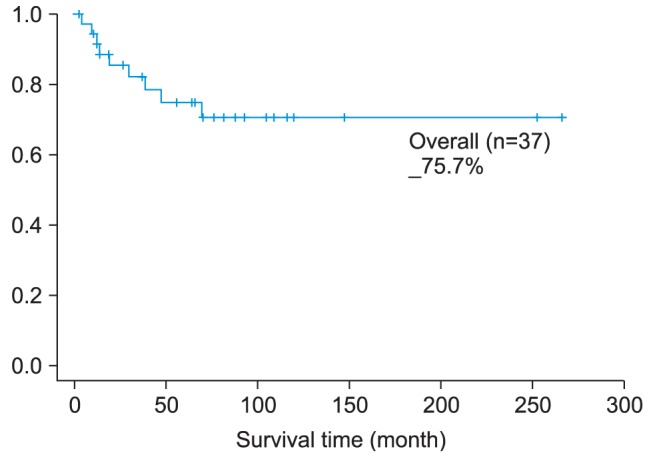
23, however, the 5-year survival rate of oral cancer patients was 63.2%. This study took the records of patients who underwent operations during the period 1998 to 2008, and the 5-year survival rate of these patients was 75.7%, which was considerably higher than that of previous research studies.
Comparing the ratio of patients in stage IV, Lee et al.
23 reported 57.1%, Cho and Kim
22, 61.3%, and Kim et al.
9, 61.7%. Note, however, that the ratio of this study was lower (35.2%). The etiology seems to be based on the other research studies for cases in stage IV and who did not undergo surgery but received palliative therapy. The same cannot be said for this study, though.
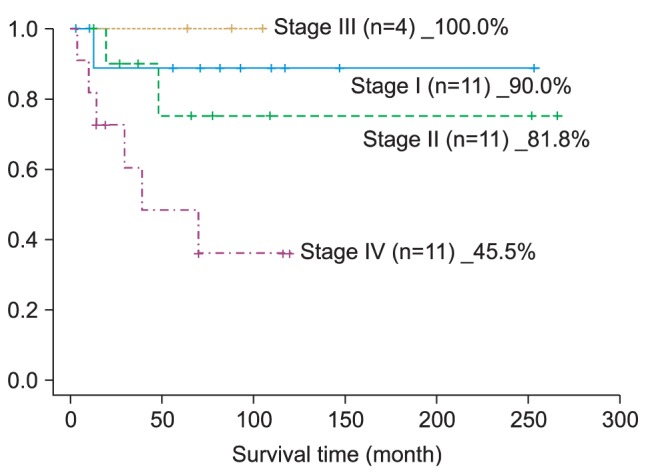
Our unusual stage III result (100%) for this study can be attributed to the small number of patients in stage III. The small number of individuals in the stage III results can also be seen in other research studies
9,
22-
24. To guess the reason, the situation wherein the primary carcinoma (T3) does not cause cervical lymph node metastasis and the situation wherein carcinoma less than 4cm (T1, T2) causes cervical lymph node metastasis are unlikely to occur. Moreover, if there were T1 and T2 patients who did not show clinical cervical metastasis clinically, they can have cervical lymph node metastasis; if patients in other stages actually belonged to stage III, then such can explain the low ratio of stage III.
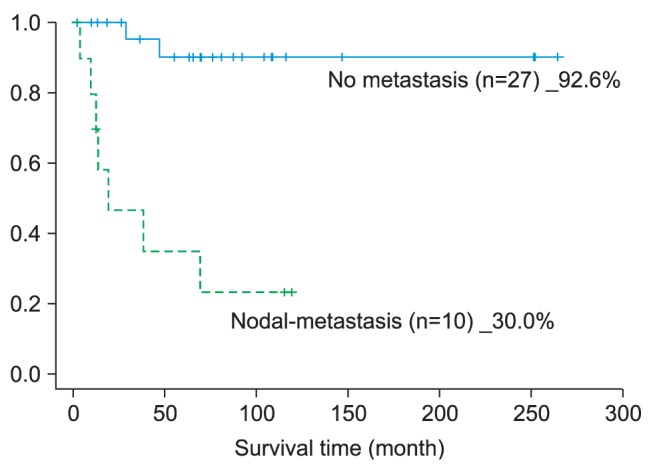
According to the 5-year survival rate in the case of cervical lymph node metastasis, in this study, a significant difference of 62.6% was noted between the two groups; in the research by Koo et al.
25, similar results of 43% and 79% were reported. Note, however, that the treatment of clinically negative cervical lymph node is still controversial. Nonetheless, it is important to consider the potential for cervical metastasis before proceeding with surgery. According to Zbären et al.'s study
26, 20% of patients with cervical lymph node metastasis--which is pN0 in the oral cancer and pathological examination--have potential for metastasis or recurrence. On the other hand, O'Brien et al.
27 stated that there may be a 30% possibility. Similarly, Keski-Säntti et al.
28 reported a low 5-year survival rate of 33% for patients confirmed to have cervical metastasis during observation and who underwent salvage surgery.
Thus, the 5-year survival rate of oral cancer patients with cervical lymph node metastasis is a very important factor in prognosis; bearing in mind the potential for cervical lymph node metastasis, and even if it is a case of cN0, planning ipsilateral or bilateral (tip of tongue, FOM, >T3, over the midline primary tumor) neck dissection must be considered
25,
29-
31.
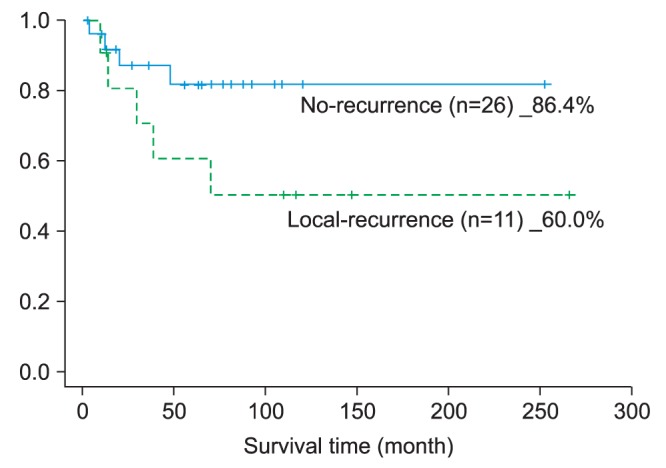
In this study, there were 15 (40.5%) patients with primary tumor recurrence or cervical lymph node metastasis, 7 of whom survived the second operation. The 5-year survival rate in case of recurrence was 46.7%; looking at cervical lymph node metastasis cases separately, however, the 5-year survival rate was lower than 30%. After operation, if the recurring tumor grows deeper in the primary site or in the cervical region, it usually positions itself in a place that is difficult to excise; hence the difficulty of completing resection due to the surrounding anatomical structures. As a result, even with the salvage surgery option as mentioned previously, the survival rate is assumed to become even lower
25,
28,
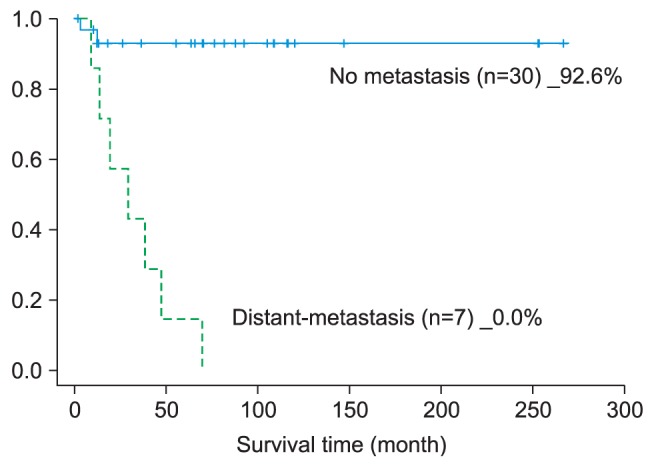
29. Moreover, cervical lymph node metastasis itself implies the possibility of distant metastasis, apparently causing the lower 5-year survival rate.
As found in this study, too, oral cancer has high possibility of recurrence, in which case the prognosis is poor; hence the importance of continuous follow-up. The follow-up conducted in this study was monthly for up to 6 months after the operation and once every 3 months from 6 months to 1 year after operation; from 1 year to 5 years, only when there was recall and if required by the patient was the PET-CT examination used to check closely for recurrence, cervical lymph node metastasis, and distant metastasis. According to the National Comprehensive Cancer Network guideline related to carcinoma of the head and neck, during follow-up, there is a need to consider the recurrence, risk of secondary primary tumor, treatment methods, and complications
30,
32. We need to examine carefully the clinical symptoms and findings of the patient and, if necessary, perform radiology imaging examination. Likewise, the guideline suggests that CT, MRI, and PET-CT examinations--which use radiology and contrast--be performed each year after operation for high-risk patients (1) with two or more lymph node metastasis, (2) wherein the affected size of a single lymph node is larger than 3 cm, (3) with extravasation spread of nodal lesion, and (4) with history of previous recurrence
30.
An advantage of this study is that it controlled variables by selecting oral cancer patients who received treatment from the same surgical team from a single lab. For its limitations, however, only a small number of target group were considered for the epidemiological studies. In the future, there is a need to collect more patient data to determine the survival rate of each patient by treatment method and analyze how, given the same primary site and cervical lymph node metastasis status, the combined treatment method--such as chemotherapy and radiotherapy performed with primary tumor resection--affects the patient's survival rate.
Go to :

V. Conclusion
A total of 37 oral cancer patients were analyzed based on gender, age, drinking status, smoking status, primary site, type of carcinoma, TNM stage, neck dissection, combination treatment and affected area, and cervical lymph node metastasis, including how these factors impacted the 5-year survival rate of each of the patients.
1. The 5-year survival rate of oral cancer patients was 75.68%; the pathological TNM stage-related, 5-year survival rate was as follows: 90.0% in stage I, 81.8% in stage II, 100% in stage III, and 45.5% in stage IV. The observed difference in survival rate by stage was statistically significant.
2. In the case of cervical lymph node metastasis after operation, the 5-year survival rate was 30%; the patients who did not have such had a 92.6% 5-year survival rate, showing significant difference as well as the greatest impact on the survival rate.
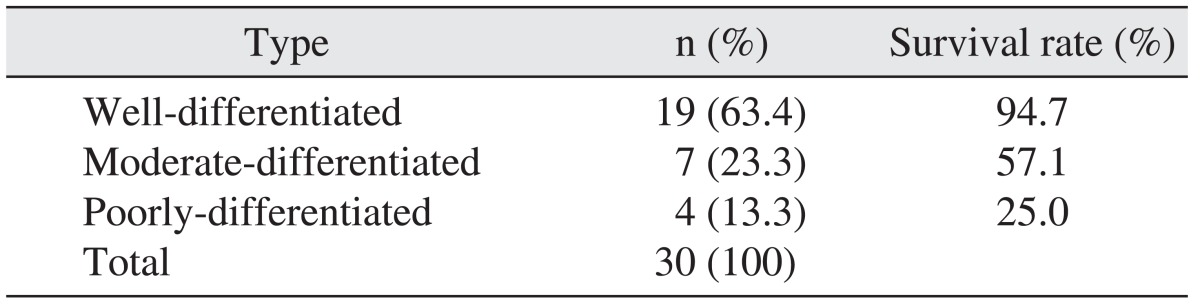
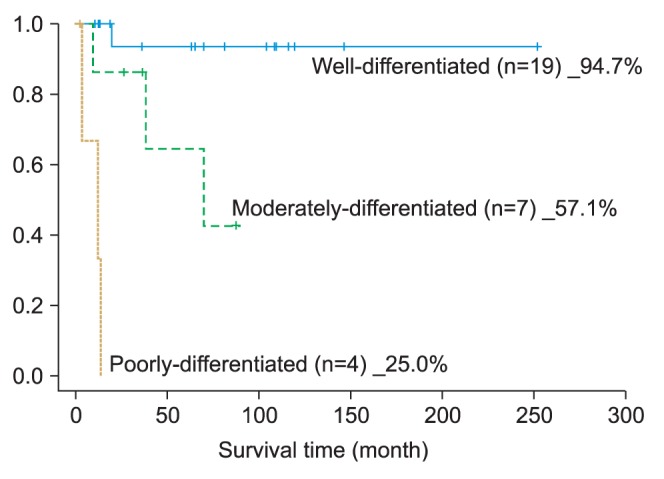
3. Based on the histopathologic examination, the well-differentiated type accounted for the largest portion of SCC's histological differentiation classification. As for the 5-year survival rate, the well-differentiated type recorded 94.7%, moderately differentiated type, 57.1%, and poorly differentiated type, 25.0%; thus showing a large difference in the survival rate depending on the differentiation classification.
4. Two factors--postoperative cervical lymph node metastasis and distant metastasis--had positive moderate coefficient correlation.
5. During the follow-up period, there were 15 (40.5%) patients with confirmed recurrence; the 5-year survival rate of these patients was 46.7%, which was lower than the total survival rate.
In summary, the factor wielding the biggest impact on long-term survival rate after operation was founded to be cervical lymph node metastasis after operation. Clinical and pathological TNM stage and local recurrence were found to be another strong factor impacting the 5-year survival rate. Therefore, we can confirm the primary tumor and pathological findings of cervical lymph node after operation and potentially improve the long-term survival rate of the patients through close follow-up observation to detect any and all postoperative findings such as cervical metastasis.
Go to :





 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download