I. Introduction
Dental implants have revolutionized the treatment of patients suffering from tooth loss. The introduction of osseo-integrated dental implants gave these patients a functional, esthetic solution to partial or total edentulism. Osseointegrated threaded titanium screw-type implants rarely lose integration after the first year of clinical function
1, and dental implants can be successful on a long-term basis at very high rates
2. Nevertheless, various complications have been observed over the years. As one of the major types of late failure, implant failure can occur for many reasons. In particular, the 2-stage external hex screw-type implant systems have been reported to exhibit unacceptably high rates of mechanical failure.
While implants rarely fracture, this complication still merits consideration for patients and clinicians alike. Many authors report very low rates of fracture. Given the increasing popularity of dental implants, the number of failures due to late implant fracture is also expected to increase. In 1992, Tolman and Laney
3 reported 3 implant fractures in a study of 1,778 implants placed (0.17%). Balshi
4 reported that 8 out of 4,045 implants fractured (0.2%). All fractures were associated with marginal bone loss. Majority of these latter cases (6 of 8) involved supporting posterior prostheses, with all patients experiencing loosening or fracture of prosthetic gold screws or abutment screws prior to implant fracture. Similarly, Rangert et al.
5 found 39 patients with fractured implants among 10,000 implants placed. They reported fracture rates of 0-6% in the maxilla but only 0-3% in the mandible. An early study by Adell et al.
2 recorded an implant fracture rate of 3.5%, with most of these fractures occurring after 5 years of clinical function; note, however, that this relatively high rate may have been due to the inclusion of implants inserted while the technique was still being developed and the longer maximum follow-up period of 15 years
2. According to Balshi
4, implant fractures may result from (1) defects in implant design and manufacturing, (2) non-passive fit of the prosthetic framework, and (3) physiological or biomechanical overload.
Fracture of the implant fixture presents several clinical challenges
6. First, the fractured fragment must be removed atraumatically to minimize bone loss. Second, an implant site of adequate length and diameter must be re-established. Finally, osseointegration of the replacement fixture must be achieved before initiating restorative replacement.
This scanning electron microscopic (SEM) study aimed at examining fractured implants for the presence of fatigue striations, dimpled surfaces, porosities, or defects of the titanium and manufacturing defects
7. According to Morgan et al.
8, the pathognomonic mark of fractures results from fatigue failure.
Targeting 6 patients, this study sought to evaluate the clinical and SEM findings of fractured implants to analyze the causes of implant fracture. We would also like to discuss the possible mechanisms by which the putative underlying factors contribute to implant fracture as well as how these cases are managed with reference to the reviewed articles on the subject.
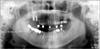
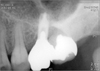
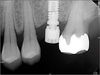
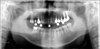
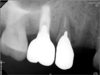
Eight implant fractures occurring between 1996 and 2010 were documented.(
Table 1) A total of 8 implant were analyzed out of an estimated 3,500 implants placed. The computer records of 6 patients treated at Wonkwang Dental Hospital were examined to gather the following data: patient's age and sex and the location of the fractured implant; the date of implant fracture; the type, length, and diameter of the implant and its position in the dental arch; the type of prosthetic rehabilitation involved, the numbers of abutments and pontics; the presence or absence of distal extensions or cantilevers; the loading time before the fracture, and; the presence of parafunctional activity. Radiographs and clinical photographs were evaluated to confirm the location of the fractured implant, presence or absence of cantilevers in the prosthesis, occlusal material, and number of teeth replaced by the prosthesis. Finally, the therapeutic solutions offered in each case were analyzed. Two fractured implants were analyzed by SEM. All of the fractures involved 4.0 mm implants, and no fracture of any 5.0 mm implant was reported.
In a series of 3,500 implants documented during the study period, we recorded 8 implant fractures or a 0.25% rate. These 8 fractured implants consist of 5 3i Osseotite implants (Biomet 3i, Palm Beach Gardens, FL, USA), 1 LC Restore implant (Lifecore Biomedical Inc., Chaska, MN, USA), and 2 Osstem US implants (Osstem Inc., Seoul, Korea). The lengths of the fractured implants ranged from 11.5 to 13.0 mm, whereas the diameter was 4.0 mm in all cases (n=8). There were more male patients than females (5 : 1), and the mean patient age was 56.7 years (48-70). Most of the fractures (n=6) involved implant-supported fixed prostheses, whereas only 2 fractured implants were supporting overdentures. A great majority of the fractured implants (n=6, 75%) were located in the molar or premolar regions (5 in the molar regions, 1 in a premolar region), whereas the other 2 were in the canine regions. More of the implant fractures were located in the upper jaw (n=6) than in the lower jaw (n=2). Most of the implants had fractured within 3-4 years of loading. Most of the patients with implant fractures (83%) exhibited bruxism and clenching. With regard to the management approaches, 6 fractured implants were removed entirely with help from explantation trephines. No further implant placement proved necessary in 1 case (implant-supported denture), whereas additional implants were placed during the same surgical intervention in the 7 other cases. No case was managed by the removal of the coronal portion of the fractured implant with posterior rectification of the apical fragment.
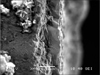
Scanning electron microscopy revealed striations on the fractured surfaces of the clinical specimens similar to those seen on the laboratory-fatigued specimens and in contrast to the dimpled surfaces of the overloaded specimens.(
Figs. 1-
3) There were no porosities within the titanium, and the fracture lines were situated in different planes. The fractured surfaces exhibited a dimpled aspect characteristic of tensile fracture.
Discussion
The long-term maintenance of osseointegration seems to be a reasonable expectation, but this neither ensures the ongoing survival of the dental restoration supported by the implant nor guarantees trouble-free prosthetic service. Osseointegrated thread titanium screw-type implants rarely lose integration after the first year of clinical function. Note, however, that implant failure can occur for other reasons, and it is one of the major causes of delayed failure
3-
5,
8. Many clinicians and patients have regarded such osseointegrated implants as successful for some time before delayed failure occurs. There are 2 main causes of a delayed implant fracture: (1) loss of supporting tissue caused by infection or peri-implantitis, and; (2) mechanical problems including fractures
3,
9.
Since osseointegrated implants have no periodontal ligament, occlusal traumatism cannot occur. Adverse forces generated by occlusal activity may instead result in the mechanical complications of implant components, i.e., screw loosening, screw fracture, or fixture fracture
5,
10. As an infrequent complication, implant fracture affects only 0.16-3.8% of implants
3,
4,
6,
7,
9. Eckert et al.
11 reported an implant fracture rate of 0.6% in both the maxilla and the mandible. According to Jemt and Leckholm
12, 1 implant fractured out of the 259 implants loaded. A multicenter retrospective analysis of 174 ITI implants used for single-tooth replacements (86.6% placed in posterior sites) reported no fixture fractures
13. The frequency of implant fracture in our series was 0.24%.
There are many causes of implant fractures, including but not limited to location, implant diameter, mechanical problems, and bruxism
3-
8. The main risk factors for implant fracture seem to be (1) bending overload
5, (2) manufacturing imperfections
4, (3) restoration design
14, (4) accuracy of restoration fit
2, (5) number, dimensions, and positioning of implants
5,
14, (6) marginal bone loss
2, (7) occlusal and parafunctional habits
2,
5,
7, and (8) chemical factors (galvanic implant corrosion)
15,
16. Regarding the effect of the location of the implant, Rangert et al.
5 reported that 90% of fractured implants are located in the regions of the molars and premolars. This agrees with our finding, i.e., majority of fractures are in the regions of molars and premolars, although the exact proportion is slightly greater than that of our cases. We reported that more of the implant fractures were in the upper than in the lower jaw. Another observer, Balshi
4, found that implant fractures occurring in the regions of premolars and molars were evenly distributed between the maxilla and mandible. Our series showed a significant difference between the numbers of fractures in each jaw.
Another important factor is implant diameter. All 8 of our fractured implants had diameter of 4 mm. Eckert et al.
11 and Balshi
4 found that all fractured implants had diameter of 3.75 mm. Implant diameters of 4.0 mm and 3.75 mm are similar in the clinic. Implants with small diameters tend to be fractured more easily than those with large diameters, especially when placed in a posterior location. According to Siddiqui and Caudill
15, an implant with diameter of 5.0 mm is 3 times stronger than one with diameter of 3.75 mm, whereas a 6.0 mm implant is 6 times stronger than a 3.75 mm implant. Another advantage of larger implants is that they are more biomechanically appropriate for replacing large posterior teeth.
Many retrospective clinical studies have reported high rates of screw loosening and/or fracture associated with 2-stage external hex implant systems. We also found an association of implant fracture with 2-stage external hex implant systems having hybrid-type surface implants. The implant fractures in our study cases all involved hybrid-type implant fixtures. The crest modules of machined hybrid-design implants are most often designed less for load bearing than for minimizing plaque accumulation and acting as a bridge to the load-bearing structure of the implant body in submerged implants
12. Still, the machined surface of a hybrid design does not effectively distribute the occlusal force. Since a smooth, machined surface in the coronal region is more readily debrided of biofilm, a hybrid design was assumed to ensure better mucosal health and lower the risk of peri-implant diseases of osseointegrated implants
17. A recent study found a positive correlation between the amount of bone loss after 6 months and the length of the machined surface for various implant systems; thus relating bone loss to the level of the "second thread." Jung et al.
18 reported significant marginal bone loss more than 3 mm long in machined coronal regions. Such bone loss may be attributable to the lack of effective mechanical loading between the machined coronal region of the implant and the surrounding bone. According to Piao et al.
17, the machined surfaces of hybrid-design implants were associated with more bone loss than those of rough-surface implants. Apparently the most important indicator of the start of an implant fracture, such marginal bone reabsorption may often extend beyond the actual fracture line. We believe that the surfaces of hybrid-design implants have different bone-implant-contact ratios or affinities for osseointegration and consequently different bending and tensile strengths in their upper and lower thirds; thus making them less able to resist occlusal forces and parafunctional activity. This seems to be the mechanism of implant fracture in these cases.
The specific bone loss pattern seen in implant fracture cases has been described as a primary cause and a mechanism of fracture
5-
7. Coronal bone resorption increases the bending stress of the implant because of the loss of supporting bone
8. In addition, this type of bone resorption usually extends to the level corresponding to the end of the abutment screw, reducing the resistance to bending of this region. Both clinical and experimental animal studies
4,
5,
7 have shown that implant overload induces resorption of the marginal bone. When such reabsorption extends apically beyond the 3rd implant thread, it reaches a structurally weak zone coinciding with the end of the prosthetic screw and the border of the hybrid surface. This contributes to fatigue at a point of low resistance to torque. Rangert et al.
5 have shown using a combination of in vitro studies and bending testing in the laboratory that metal fatigue can cause fractures. The warning signs of such fatigue include loosening, torsion, or fracture of the post screws and ceramic fractures of the prosthesis. These signs indicate metal fatigue that will ultimately lead to fracture if not corrected on time.
Galvanic implant corrosion can also contribute to fractures
17,
18. Corrosion of the metal crown may be caused or accele rated by differences in the electric potentials of the implant (made of pure titanium) and the crown (made of nickel-chromium-molybdenum alloy). Contrary to common belief, titanium is a highly reactive metal. Cathodic to most metals, it may accelerate galvanic attack on less noble metals when they are coupled together. The parameters that affect the magnitude of this galvanic corrosion include the difference in electrolytic potential between the metals and the contact area of the interface. In particular, the electrolytic potential of a given metal is constant. The difference can be minimized by fabricating restorations from a metal alloy with an electrolytic potential similar or close to that of titanium. Alternatively, the contact area between the implant and the restoration may be isolated with cement (such as zinc phosphate cement)
19.Reducing the actual surface area of contact between the 2 metals is not recommended, as doing so will reduce the retention of restoration
15. Therefore, the possibility of galva nic corrosion must always be considered when choosing a metallic alloy for the crown. Note, however, that there is currently little additional evidence to support the role for this phenomenon as a major contributor to implant fractures. We generally manage fractured implants by complete extraction using a trephine burr to pull out the fractured implant fixture. When the percentage of contact with bone is high, and the fracture is located not too far from the apical area, however, restoration of the connection between the post and the implant may be a valid option. Therefore, confirming the condition of the fracture radiologically is essential before determining whether to extract the fragment.
This investigation demonstrated that the fractures of the fixture components of these dental implants occurred due to fatigue under physiological loads, with marginal alveolar bone loss around the fixture. It is important to avoid mechanical problems and excessive bone reabsorption to prevent implant fracture. We would like to suggest that clinicians consider adopting the following measures:
1) Avoid placing hybrid-type implant fixtures.
2) Carefully control occlusal forces: eliminate all posterior contacts in mandibular eccentric movements.
3) Perform staggered placement of implants: avoid a straight-line configuration.
4) Avoid or minimize posterior cantilevers and buccolingual offsets particularly in partially edentulous patients.
5) When chronic loosening of gold or abutment screws or fracture of components other than the implants occurs, critically reassess the prosthesis, re-tighten the abutment, use 1 gold screw, and check the fit.
6) Pronounced bruxers or clenchers who have experienced multiple implant fractures should be managed by the placement of additional implants.
7) Ensure perfect fit: resolder and assess passive fit.
Periodic and careful periodontal and prosthodontic evaluation and analysis should be performed before and after implant restoration. Perfect passive fit and adaptation according to the biomechanical principles must also be considered when planning the placement of implants in partially and completely edentulous ridges. It is important to know and apply such measures since they will reduce the risk of implant fractures. If necessary, prosthodontic measures should be taken to optimize occlusion to achieve adequate occlusal contact and avoid undesired forces.




 PDF
PDF ePub
ePub Citation
Citation Print
Print







 XML Download
XML Download