INTRODUCTION
The equation derived in the Modification of Diet in Renal Disease (MDRD) study was recommended by the 2002 Kidney Disease Outcomes Quality Initiative of the National Kidney Foundation practice guidelines for chronic kidney disease (CKD) to calculate the serum creatinine (Scr)-based estimated glomerular filtration rate (eGFR) in adults [
1]. Following its introduction, many clinical laboratories have calculated the eGFR in adults using the MDRD equation and have reported this value along with the Scr concentration via laboratory information systems (LISs). However, the MDRD equation has a major disadvantage, in that bias is increased at higher GFRs [
2]. To overcome this limitation, the Scr-based Chronic Kidney Disease Epidemiology Collaboration (CKD-EPIcr) equation was developed, and the eGFR equation recommended for adults in the current 2012 Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for CKD has been changed from the MDRD to the CKD-EPIcr equation [
234].
According to the 2012 KDIGO guidelines for CKD, it is acceptable to use an alternative Scr-based eGFR equation instead of the CKD-EPIcr equation; however, the accuracy of the alternative equation should be compared to that of the CKD-EPIcr equation [
4]. New eGFR equations, including the revised Lund–Malmö (revised LM) [
56] and full age spectrum (FAS) equations [
7], were developed for use with Western (mainly Caucasian) populations. Both equations reportedly produce accurate and precise eGFRs compared with those of the CKD-EPIcr equation [
57]. When estimating GFR using the Scr-based eGFR equation, the most significant factor is the Scr concentration, which reflects individual lean body mass that is in turn influenced by sex, age, body mass index (BMI), and race. In addition, laboratory-related factors can affect Scr levels, such as the calibrator used to measure Scr concentration (isotope dilution mass spectrometry [IDMS]-traceable vs non-IDMS-traceable calibrator), type of test reagent (Jaffe vs enzymatic method), and the analytical performance of individual laboratories [
8].
The recently introduced FAS and revised LM equations were developed from the Scr results for Caucasians obtained by using an enzymatic method with an IDMS-traceable calibrator [
57]. Thus, it was necessary to evaluate whether these equations could be applied to Koreans. Ji et al [
9] compared the revised LM equation with the MDRD and CKD-EPI equations for use in Koreans; however, the accuracy of the revised LM equation was not evaluated, because eGFR was not compared with measured GFR (mGFR) in that study. In addition, studies on the performance evaluation of the Asian CKD-EPI and FAS equations for Koreans are limited. Therefore, the purpose of this study was to assess the analytical and clinical performance of the eGFR equations, including the revised LM and FAS equations, in Koreans.
DISCUSSION
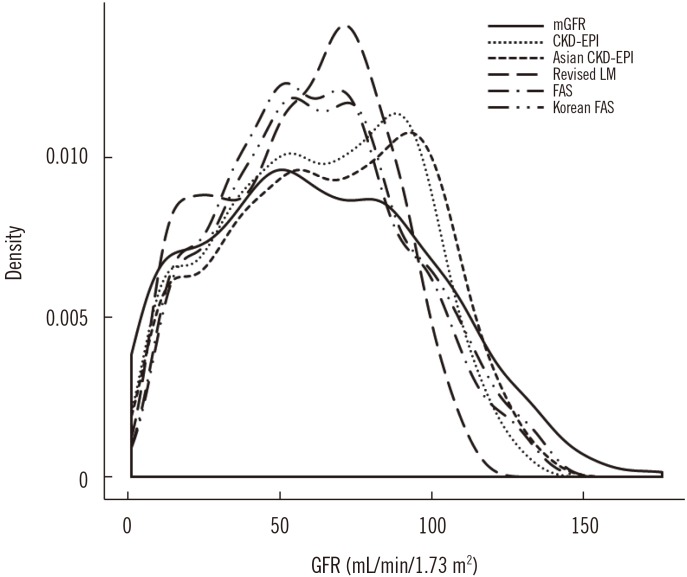
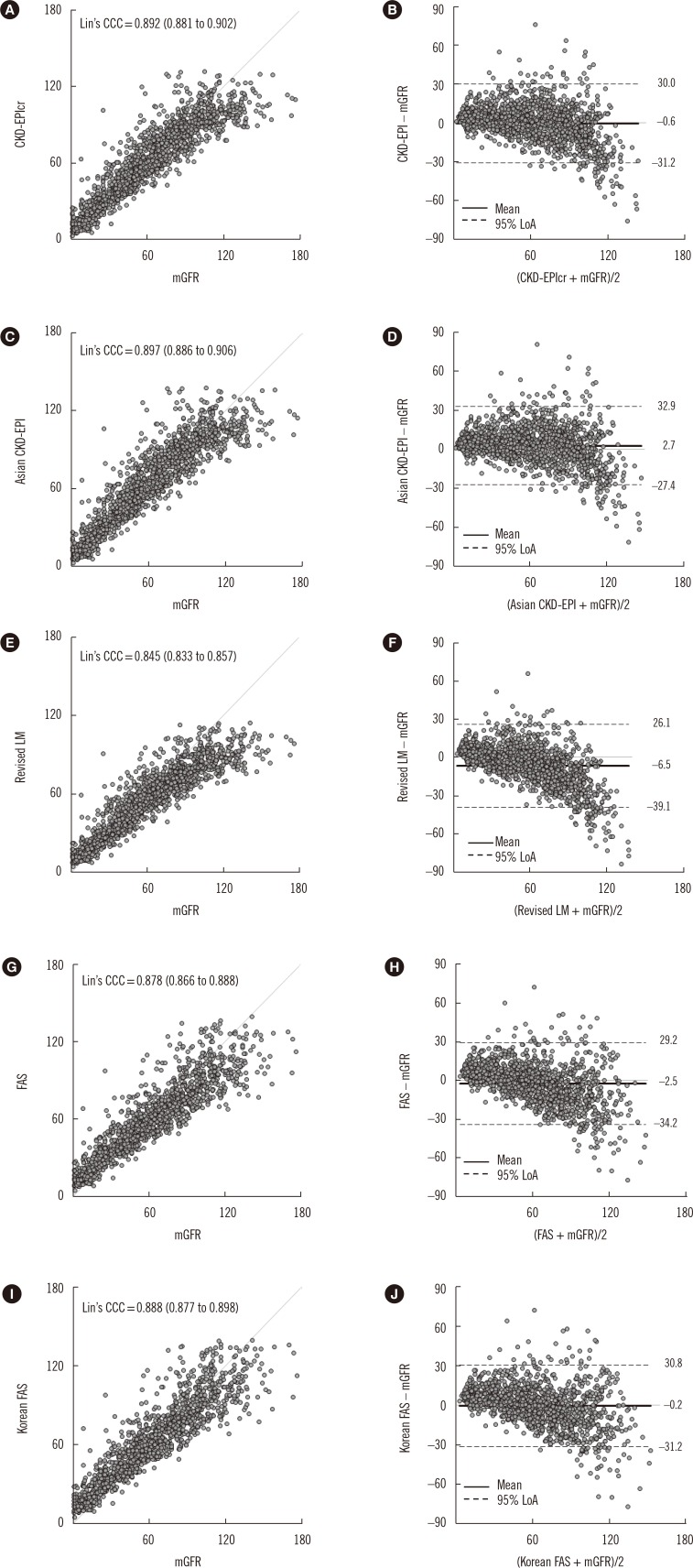
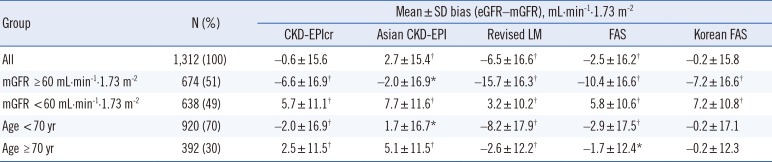
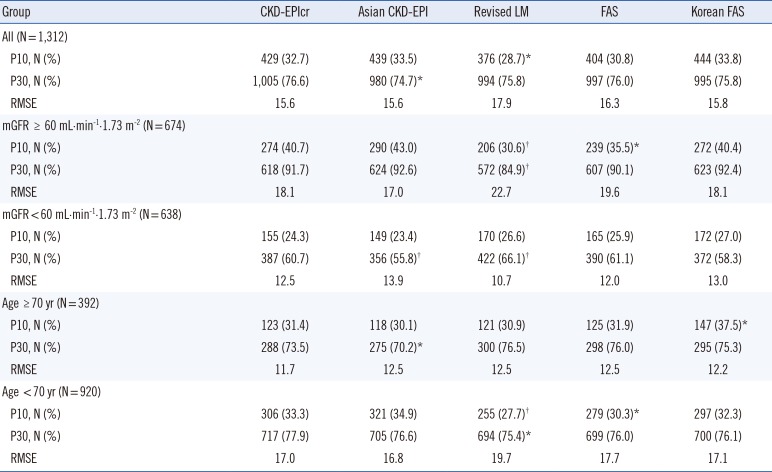
Although the bias of each eGFR equation varied depending on sex, mGFR, and age group, overall, the mean bias of the CKD-EPIcr and Korean FAS equations was not significantly different from zero. Moreover, among the five eGFR equations, the mean bias of the Korean FAS equation was nearest to zero and was smaller than that of the CKD-EPIcr equation (−0.2 vs −0.6). The P30 values of the five eGFR equations ranged from 74.7% to 76.6%, with the highest value obtained for CKD-EPIcr, and the remaining four equations showing clinically irrelevant differences. Both the CKD-EPIcr and Asian CKD-EPI equations showed the lowest RMSE value of 15.6; the RMSE values for the revised LM, FAS, and Korean FAS equations were 17.9, 16.3, and 15.8, respectively.
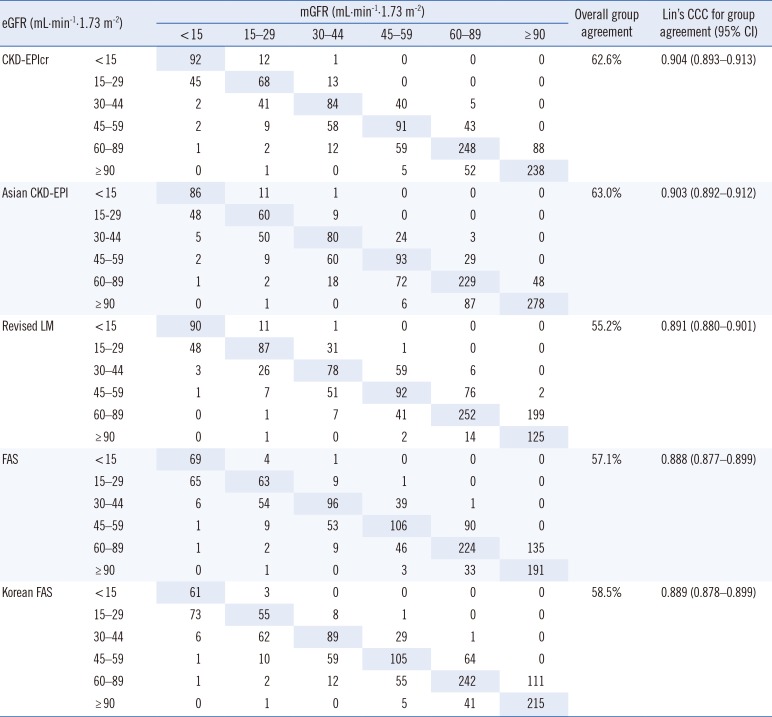
Comparison of the numeric values between mGFR and eGFR showed that although the Lin's CCC values of the five eGFR equations were all <0.90 (range 0.845–0.897), since both the mGFR and eGFR do not involve analytical chemistry measurement procedures, we considered that a Lin's CCC range of 0.85 to 0.90 is quite acceptable. Consequently, these results suggest that the Korean FAS equation has equivalent analytical and clinical performance to the CKD-EPIcr equation in Korean adults compared with the Asian CKD-EPI, revised LM, and FAS equations. Overall, both the CKD-EPIcr and Korean FAS were able to provide precise and accurate GFR estimates, but the other equations were also advantageous in the sub-group analysis. For example, the Asian CKD-EPI and revised LM equations showed the lowest bias for the mGFR ≥ 60 mL·min-1·1.73 m-2 group and for the mGFR<60 mL·min-1·1.73 m-2 group, respectively.
The performance of the eGFR equation could be improved by using coefficients specific for local ethnic groups [
812]. The Asian CKD-EPI equation, as one of the four-level race (White and other, Black, Asian, Native American and Hispanic) CKD-EPI equations, was modified from the CKD-EPIcr equation [
12]. In a previous study, the bias (eGFR–mGFR, mL·min
-1·1.73 m
-2) of the Asian CKD-EPI equation was found to be significantly improved compared with the CKD-EPIcr equation (−0.8 vs −2.1,
P<0.005) [
12]. In the present study, however, the mean bias of the Asian CKD-EPI equation was significantly higher than that of the CKD-EPIcr equation (2.7 vs −0.6 mL·min
-1·1.73 m
-2,
P<0.001). In addition, the P30 of the Asian CKD-EPI equation was significantly lower than that of the CKD-EPIcr equation (74.7% vs 76.6%,
P<0.05). These conflicting results may be explained by the following reasons. First, the Asian CKD-EPI equation may be less representative because it was developed from a relatively small group of participants (n=100) compared with the White and other (n=5,216), Black (n=2,585), or Native American and Hispanic (n=353) equations. Second, even the same Asian individual may have different clinical characteristics depending on their local region of origin or residence. Third, several factors related to the above-mentioned Scr assay can have an influence on the results. In a previous study, we reported that the bias of the Korean version of the CKD-EPI equation was significantly smaller than that of the CKD-EPIcr equation (−2.3 vs −3.0 mL·min
-1·1.73 m
-2,
P<0.001) [
11]. Therefore, the bias of the eGFR equation can vary depending on which equation is applied to which study group [
1214].
The revised LM equation showed a positive bias at lower GFR levels and a negative bias at higher GFR levels [
51516]. Our results are in accordance with these findings: the mean bias of the revised LM equation was lower than that of the CKD-EPIcr equation (3.2 vs 5.7), and the P30 value of the revised LM equation was significantly higher (66.1% vs 60.7%,
P<0.001) than that of the CKD-EPIcr equation at mGFR<60 mL·min
-1·1.73 m
-2. In contrast, the mean bias of the revised LM equation was higher at mGFR ≥60 mL·min
-1·1.73 m
-2 than that of the CKD-EPIcr equation (−15.1 vs −6.6), and the P30 of the revised LM equation was lower (84.9% vs 91.7%,
P<0.001). In other words, the revised LM equation predicted GFR more precisely and accurately than the CKD-EPI equation at GFR<60 mL·min
-1·1.73 m
-2, and the opposite was true for GFR ≥60 mL·min
-1·1.73 m
-2.
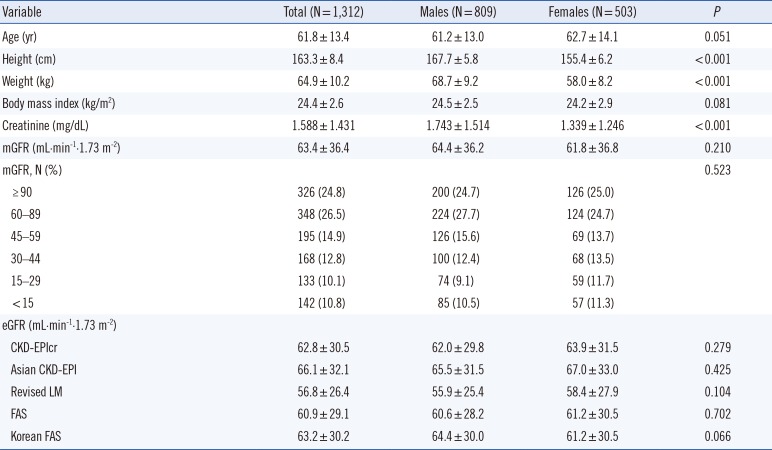
In general, the performance of the eGFR equation reflects the characteristics of the subjects involved in the development of the corresponding eGFR equation. The revised LM equation was intended to improve performance at a lower GFR level, and the median GFR of the study group was 55 mL·min
-1·1.73 m
-2 [
6]. By contrast, the CKD-EPIcr equation was developed to improve the performance at a higher GFR level, and the mean GFR of the study group was 68 mL·min
-1·1.73 m
-2 [
2]. Therefore, the characteristics of these research groups are reflected in the performance evaluation conducted in the present study. In addition to the characteristics of the study participants, performance evaluation of the eGFR equations may be influenced by the Scr concentration and GFR measurement methods [
8]. For example, the revised LM equation used iohexol to measure GFR, whereas the CKD-EPIcr equation used iothalamate. For measurement of the Scr concentration, the revised LM equation used the enzymatic and modified Jaffe methods according to age, and the CKD-EPIcr equation used the enzymatic method. These differences in measurement methods could have affected the analytical performance of the eGFR equations.
The FAS equation has a simple structure compared with that of the MDRD, CKD-EPIcr, and revised LM equations. It is very important that the Q-value is set appropriately for application to the FAS equation. Because the Q-value for the FAS equation was derived from a Belgian population, we determined the Korean version of Q-values from a Korean population and compared the performance of the FAS equation with that of the Korean FAS equation. Our results showed that the Q-value of Korean males was higher than that of Belgian males (85 vs 80 µmol/L), while the values for females were the same in both populations (62 µmol/L) [
7]. This difference may be explained by the characteristics of the population (Belgian vs Korean) or the analytical method used for Scr measurement (Jaffe vs enzymatic method). In this study, the overall mean bias of the Korean FAS equation was significantly lower than that of the FAS equation (−0.2 vs −2.5,
P<0.0001), according to the concept of population-based Scr. These results are consistent with the implications of the FAS equation. Although we did not evaluate children, the FAS equation has the advantage of being able to apply the same equation to the age group of two years and older, unlike the CKD-EPIcr equation.
In conclusion, both the CKD-EPIcr and Korean FAS equations showed equivalent analytical and clinical performances, and thus may be the preferred choice for laboratories to report eGFR along with Scr in the Korean adult population.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


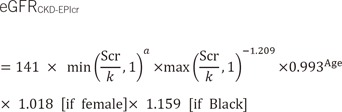

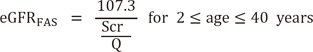
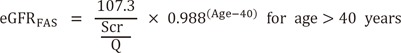
 XML Download
XML Download