Abstract
Peripheral blood stem cell (PBSC) transplantation following myeloablative therapy is a mainstay of treatment for various types of malignancies. This study aimed to evaluate the differences between the Optia MNC and COBE Spectra MNC systems (Terumo BCT, Japan) according to apheresis procedures and the parameters of apheresis, products, and collection. The clinical data of 74 patients who underwent autologous PBSC collection from July 2012 to July 2015 were reviewed retrospectively. The patients comprised 48 (65%) men and 26 (35%) women with a median age of 56 yr (range, 23–66 yr). Of 216 procedures, 111 (51%) and 105 (49%) were processed by using COBE and Optia MNC, respectively. PBSC collection rates, throughput, numbers of stem cells retrieved, collection efficacy, and platelet loss were compared. There were no significant differences in the median CD34+ cell counts of collected products (0.61×108 vs 0.94×108), CD34 collection efficiency (43.5% vs 42.1%), and loss of platelets (40.1% vs 44.7%). The Spectra Optia MNC apheresis system was comparable to the COBE Spectra system in collecting autologous CD34+ hematopoietic stem cells and retention of platelets.
Peripheral blood stem cell (PBSC) transplantation following myeloablative therapy is a mainstay of treatment for various types of malignancies [12]. Stem cell-rich populations for stem cell transplantation (SCT) are generated by enforced mobilization with granulocyte-colony-stimulating factor (G-CSF), with or without chemotherapy.
Two types of stem cell collection systems are currently utilized: manually modified and automated collection systems. The most commonly used system is the COBE Spectra apheresis system (Terumo BCT, Tokyo, Japan), which requires intermittent optical/manual input from the operator, making it labor-intensive and prone to user-dependent variability [34]. The quality of the cells collected with apheresis systems such as COBE has been reported to depend on the operator and stable blood flow [56].
The Spectra Optia MNC (Terumo BCT) has two programs for mononuclear cell (MNC) collections: the MNC program and the continuous MNC (CMNC) program [7]. We used the MNC program, which has electronic features, including optical sensors for continuous automatic monitoring and stabilization of the interface and monitoring of the collection flow line, as well as a novel tubing system that is specifically designed to reduce platelet (PLT) attrition. The CMNC program has a continuous collection mode, which is similar to the MNC program of COBE Spectra [589].
Although the two systems have been compared [6], we did not find a report of such a comparison study in Korea. Therefore, the aim of this study was to evaluate the differences between the Optia MNC and COBE Spectra MNC systems according to apheresis procedures and the parameters of apheresis, products, and collection.
The study was approved by the Institutional Review Board in National Cancer Center, Korea (No. 2015-0272). The study included 74 patients who underwent autologous PBSC transplantation between July 2012 and July 2015, during which time both the Optia MNC and Spectra systems were used. The 74 patients comprised 48 (65%) men and 26 (35%) women with a median age of 56 yr (range, 23–66 yr). Of the 74 patients, 29 (39%) had B-cell lymphoma, 27 (36%) had multiple myeloma, 16 (22%) had natural killer (NK)/T-cell lymphoma, and 2 (3%) had Hodgkin's disease.
Stem cells were collected according to the institution's mobilization protocol, by using cytotoxic agents plus G-CSF (n=54, 73%) or G-CSF alone (n=20, 27%). Patients mobilized with cytotoxic agents plus G-CSF were injected once daily with 10 µg G-CSF/kg body weight, and PBSCs were collected on the day that the hematopoietic progenitor cell (HPC) count was ≥5/µL or the white blood cell (WBC) count was >3,000/µL [101112]. Patients mobilized with G-CSF alone were injected once daily for four days with the same dose of G-CSF prior to the apheresis procedure. WBC, PLT, MNC, and HPC counts, and Hct levels in the peripheral blood and PBSC products were determined by using an XE-2100 hematology analyzer (Sysmex Corporation, Kobe, Japan), with differential counts determined by manual counting. Peripheral blood parameters on each day of collection are shown in Table 1.
Stem cells were collected according to the manufacturer's recommendations [7], by using a central venous access (allowing for a flow of at least 65 mL/min). Large-volume leukapheresis to process more than 3–5-times the blood volume was performed with both devices for the collection each day. The whole blood-to-anticoagulant (3,000 IU heparin with 500 mL of acid-citratedextrose solution) ratio was 24:1 in both devices.
The collection pump rate set for the COBE procedures was 1.0–1.3 mL/min, and the separation factor remained constant during the apheresis procedure regardless of the actual WBC count. When we used the Optia MNC system, collection preference was determined by the device algorithm. Buffy coat collection from the elutriation chamber was triggered manually when the operator observed a buffy coat overflow earlier than the red blood cell sensor. Patients undergoing stem cell collection were assigned to the COBE or Optia MNC system in a random manner.
CD34+ cells were counted in pre- and post-apheresis peripheral blood samples and in a product sample by flow cytometry (FACS Canto II, Becton, Dickinson and Company, San Jose, CA, USA), using a single-platform assay (BD stem cell enumeration kit, Becton Dickinson and Company) [1314].
The CD34+ cell dose per kg body weight was determined. The apheresis target dose for one autologous SCT was ≥3×106 CD34+ cells/kg body weight before freezing. The collection efficiency was calculated by using the widely accepted ‘CE2’ model [1516] as follows: (Total CD34+ cells obtained from apheresis ×100)/[Peripheral CD34+ cells/µL×Apheresis volume processed (µL)].
PLT loss during apheresis was calculated by using the formula: % PLT loss=[(PLT per µL before apheresis-PLT per µL after apheresis)/PLT per µL before apheresis]×100 [1617].
All statistical analyses were performed by using Stata Statistical Software Release 12 (StataCorp LP, College Station, TX, USA). Descriptive data are presented as median with range. Comparisons were made by using Fisher's exact test, Mann-Whitney U test, as appropriate; P<0.05 was considered statistically significant.
The 74 patients underwent a total of 216 autologous apheresis procedures. There were no differences in sex, age, body weight, diagnosis, mobilization regimens, and duration of collection between the COBE and Optia MNC groups (Table 1).
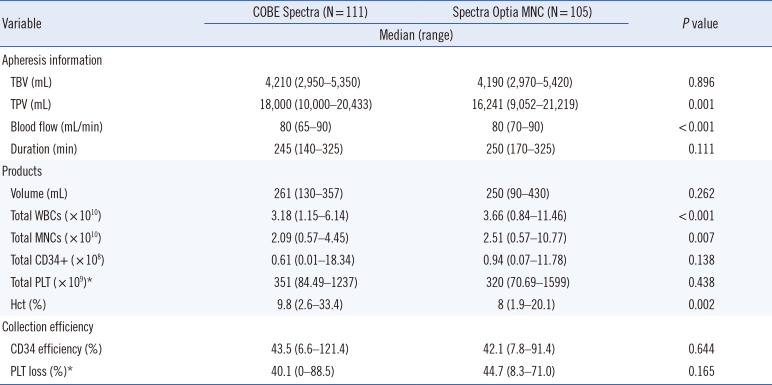
All procedures were well tolerated and no signs of clinical adverse events such as citrate toxicity were observed for any of the apheresis procedures. The details of performance of the two apheresis systems are summarized in Table 2. The total processed volume (TPV) was higher in the COBE group (P=0.001), whereas the flow rate was lower in the COBE group than in the Optia MNC group (77 mL/min vs 81 mL/min, P<0.001). However, the total procedure time was similar in the two groups (P=0.111). The parameters for stem cell preparations are summarized in Table 2. The volume of the stem cell preparations was similar in the two systems (P=0.262). Although WBC (P<0.001) and MNC (P=0.007) counts were significantly higher and Hct (P=0.002) was significantly lower in preparations collected by Optia MNC than by COBE, there were no significant differences in total CD34+ (P=0.138) and PLT (P=0.438) counts. The median CD34+ collection efficiency by the COBE and Optia MNC systems were similar (43.5% vs 42.1%, P=0.644), as were PLT losses after apheresis (40.1% vs 44.7%, P=0.165). Patients who received PLT transfusions before stem cell collection were excluded from the calculation of PLT loss (Table 2).
The present results showed that the performance of two apheresis systems, COBE and Optia MNC, was comparable for autologous PBSC collection. Although the TPV was higher and the blood flow rate was lower with the COBE system than with the Optia MNC system, the procedure did not differ significantly. The higher flow rate observed with the Optia MNC system was likely due to the ability of this instrument to maintain a high flow rate in the allowable range. However, a previous study reported that the collection time for the same TPV was longer with the Optia MNC system than with the COBE system owing to discontinuous collection [18]. Moreover, the TPV was smaller using the Optia MNC system, which induces early termination of apheresis when the target dose is reached, even if a full apheresis cycle has not been completed [8].
Previous studies have reported conflicting results about product volumes with these two systems. One study found that the product volume and TPV were lower with the Optia MNC system than with the COBE system [16]. Another study, which compared three different apheresis systems (COM.TEC [Terumo BCT], COBE, and Optia MNC) found that the product volume was lowest with the COBE system [18]. In contrast, our comparison of the Optia MNC and COBE systems showed no significant differences in product volume. Although sex, age, body weight, diagnosis, mobilization regimens, and collection duration were similar in the two groups, the peripheral blood WBC count was significantly higher in the Optia MNC group. Stem cell preparations obtained with the Optia MNC system may therefore have higher WBC and MNC counts. This result is in contrast to that of a previous study [19]. Although the peripheral blood PLT count was higher in the Optia MNC group, there were no significant differences in PLT counts in blood products and in PLT loss between the two groups. The Hct level in blood cell preparations was significantly lower in the Optia MNC group, consistent with previous findings [20]. This may reflect the chamber system in Optia MNC, which reduces non-target cells in the final product. Moreover, a similar CD34+ collection efficiency was observed in the two groups, consistent with previous studies. Although this study was conducted at a single center, the results demonstrate that the collection efficiencies of Spectra and Optia MNC are comparable.
Acknowledgments
This work was supported in part by the grant from the National Cancer Center, Korea (NCC-1210101) and by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (No. 2014R1A2A2A01002553).
References
1. Barlogie B, Shaughnessy J, Tricot G, Jacobson J, Zangari M, Anaissie E, et al. Treatment of multiple myeloma. Blood. 2004; 103:20–32. PMID: 12969978.
2. Li DQ, Hou YF, Wu J, Chen Y, Lu JS, Di GH, et al. Gene expression profile analysis of an isogenic tumour metastasis model reveals a functional role for oncogene AF1Q in breast cancer metastasis. Eur J Cancer. 2006; 42:3274–3286. PMID: 16979889.
3. Ikeda K, Ohto H, Nemoto K, Yamamoto G, Kato K, Ogata T, et al. Collection of MNCs and progenitor cells by two separators for PBPC transplantation: a randomized crossover trial. Transfusion. 2003; 43:814–819. PMID: 12757534.
4. Heuft HG, Dubiel M, Rick O, Kingreen D, Serke S, Schwella N. Inverse relationship between patient peripheral blood CD34+ cell counts and collection efficiency for CD34+ cells in two automated leukapheresis systems. Transfusion. 2001; 41:1008–1013. PMID: 11493732.
5. Karafin MS, Graminske S, Erickson P, Walters MC, Scott EP, Carter S, et al. Evaluation of the Spectra Optia apheresis system for mononuclear cell (MNC) collection in G-CSF mobilized and nonmobilized healthy donors: results of a multicenter study. J Clin Apher. 2014; 29:273–280. PMID: 24677347.
6. Brauninger S, Bialleck H, Thorausch K, Felt T, Seifried E, Bonig H. Allogeneic donor peripheral blood “stem cell” apheresis: prospective comparison of two apheresis systems. Transfusion. 2012; 52:1137–1145. PMID: 22044384.
7. TerumoBCT. Spectra Optia® Apheresis System. Last accessed on Dec 1, 2015. http://www.terumobct.com/location/north-america/products-and-services/Pages/spectra-optiaapheresis-system.aspx.
8. Brauninger S, Bialleck H, Thorausch K, Seifried E, Bonig H. Mobilized allogeneic peripheral stem/progenitor cell apheresis with Spectra Optia v.5.0, a novel, automatic interface-controlled apheresis system: results from the first feasibility trial. Vox Sang. 2011; 101:237–246. PMID: 21517897.
9. TerumoBCT. COBE® Spectra Apheresis Syetem. Last accessed on Dec 1, 2015. https://www.terumobct.com/location/north-america/products-and-services/Pages/COBE-spectra-apheresis-system.aspx.
10. Suh C, Kim S, Kim SH, Kim EK, Lee JL, Park KU, et al. Initiation of peripheral blood progenitor cell harvest based on peripheral blood hematopoietic progenitor cell counts enumerated by the Sysmex SE9000. Transfusion. 2004; 44:1762–1768. PMID: 15584992.
11. Kim MK, Kim S, Jang G, Lee SS, Sym SJ, Lee DH, et al. A randomized comparison of peripheral blood hematopoietic progenitor cell level of 5/mm3 versus 50/mm3 as a surrogate marker to initiate efficient autologous blood stem cell collection. J Clin Apher. 2007; 22:277–282. PMID: 17880017.
12. Lefrère F, Zohar S, Beaudier S, Audat F, Ribeil JA, Ghez D, et al. Evaluation of an algorithm based on peripheral blood hematopoietic progenitor cell and CD34+ cell concentrations to optimize peripheral blood progenitor cell collection by apheresis. Transfusion. 2007; 47:1851–1857. PMID: 17880611.
13. Keeney M, Chin-Yee I, Weir K, Popma J, Nayar R, Sutherland DR. Single platform flow cytometric absolute CD34+ cell counts based on the ISHAGE guidelines. International Society of Hematotherapy and Graft Engineering. Cytometry. 1998; 34:61–70. PMID: 9579602.
14. Sutherland DR, Nayyar R, Acton E, Giftakis A, Dean S, Mosiman VL. Comparison of two single-platform ISHAGE-based CD34 enumeration protocols on BD FACSCalibur and FACSCanto flow cytometers. Cytotherapy. 2009; 11:595–605. PMID: 19513900.
15. Ford CD, Lehman C, Strupp A, Kelley L. Comparison of CD34+ cell collection efficiency on the COBE Spectra and Fenwal CS-3000 Plus. J Clin Apher. 2002; 17:17–20. PMID: 11948701.
16. Reinhardt P, Brauninger S, Bialleck H, Thorausch K, Smith R, Schrezenmeier H, et al. Automatic interface-controlled apheresis collection of stem/progenitor cells: results from an autologous donor validation trial of a novel stem cell apheresis device. Transfusion. 2011; 51:1321–1330. PMID: 21155834.
17. Long G, Waller EK, Gregurek S, Tricot G, Marschner S, Bill J. Evaluation of the spectra Optia® mononuclear cell collection procedure in multiple myeloma patients. J Clin Apher. 2015; 30:1–7. PMID: 24941931.
18. Flommersfeld S, Bakchoul T, Bein G, Wachtel A, Loechelt C, Sachs UJ. A single center comparison between three different apheresis systems for autologous and allogeneic stem cell collections. Transfus Apher Sci. 2013; 49:428–433. PMID: 23827328.
19. Lee JS, Cho D, Shin MG, Ryang DW, Lee JJ, Kim HJ, et al. Comparison of Amicus and CS-3000 Plus for the collection of peripheral blood stem cells. Korean J Blood Transfus. 2006; 17:39–47.
20. Cherqaoui B, Rouel N, Auvrignon A, Defachelles AS, Deméocq F, Kanold J, et al. Peripheral blood stem cell collection in low-weight children: retrospective comparison of two apheresis devices. Transfusion. 2014; 54:1371–1378. PMID: 24117598.
Table 1
Characteristics of patients who underwent autologous blood stem cell collection
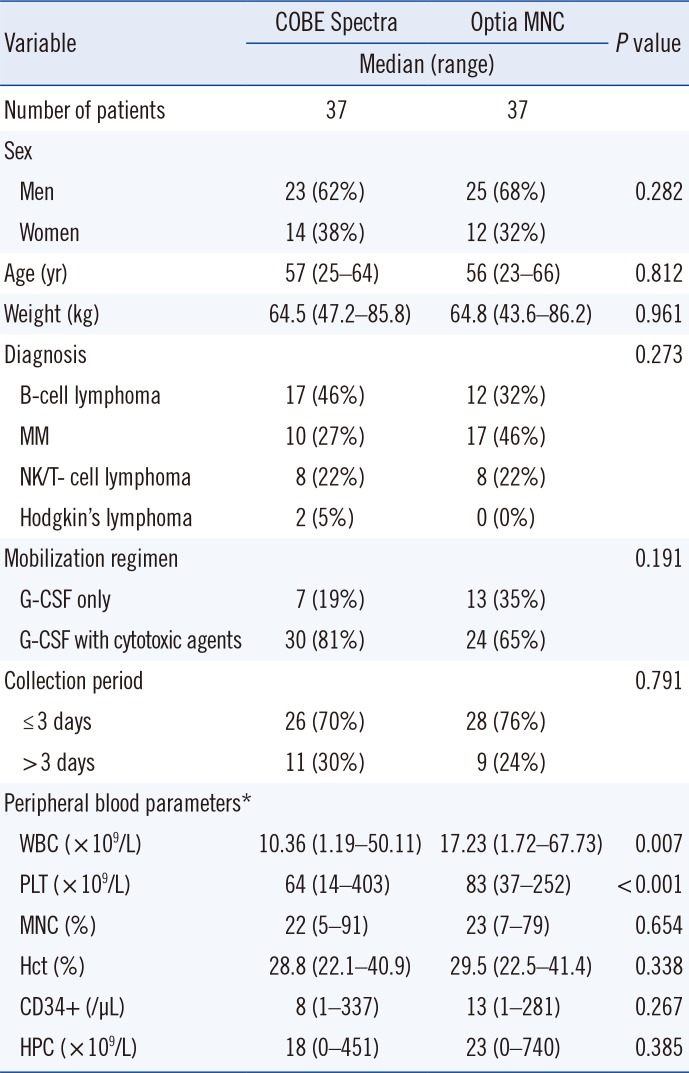
Table 2
Parameters of apheresis, products, and collection




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download