This article has been
cited by other articles in ScienceCentral.
Dear Editor,
Ceftolozane-tazobactam (C/T) and ceftazidime-avibactam (CZA) were recently approved for the treatment of complicated intra-abdominal infections and complicated urinary tract infections (
http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm427534.htm,
http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm435629.htm, both accessed February 24, 2016). To date, only one study has simultaneously evaluated the activities of C/T and CZA
in vitro against
Pseudomonas aeruginosa, and few studies have evaluated the effects of these antibiotics on multi-drug resistant (MDR) gram-negative bacteria [
123]. This study aimed to examine the activities of C/T and CZA against β-lactam-resistant
Enterobacteriaceae and
P. aeruginosa clinical isolates.
The isolates were recovered from clinical specimens at Barnes-Jewish Hospital (St. Louis, MO, USA) from September to December 2014. Specimen sources included respiratory, blood, urine, and wound samples. Isolates of
Enterobacteriaceae were included if they tested non-susceptible to cefepime and/or had an extended-spectrum β-lactamase (ESBL)-producing phenotype.
P. aeruginosa isolates were included if they tested non-susceptible to meropenem. We included carbapenem-resistant
Enterobacteriaceae isolates recovered from August 2012 to December 2014; these strains were tested for the
blaKPC and
blaNDM genes by real-time PCR [
45]. All isolates were negative for the
blaNDM gene.
Frozen stocks of all isolates were subcultured twice consecutively on 5% sheep's blood agar (Hardy Diagnostics, Santa Maria, CA, USA) prior to antimicrobial susceptibility testing (AST). Species-level identification was confirmed by using the VITEK MS system (IVD v2.3.3, bioMérieux, Durham, NC, USA) [
67]. AST was performed by using gradient diffusion (Etest, bioMérieux). In brief, a 0.5 McFarland standard suspension of each isolate was inoculated onto Mueller-Hinton agar (Hardy Diagnostics), and Etest strips were applied. Plates were incubated overnight at 35℃ in ambient air. Each day, QC strains (
Escherichia coli ATCC 25922 and ATCC 35218, and
P. aeruginosa ATCC 27853) were tested. C/T and CZA results and QC were interpreted by using Food and Drug Administration breakpoints. The categorical interpretation and QC ranges for all other antibiotics (BD BBL and Co., Sparks, MD, USA) were based on standardized disk diffusion criteria [
8].
We evaluated 120 clinical isolates comprising 45
P. aeruginosa strains and 75
Enterobacteriaceae strains.
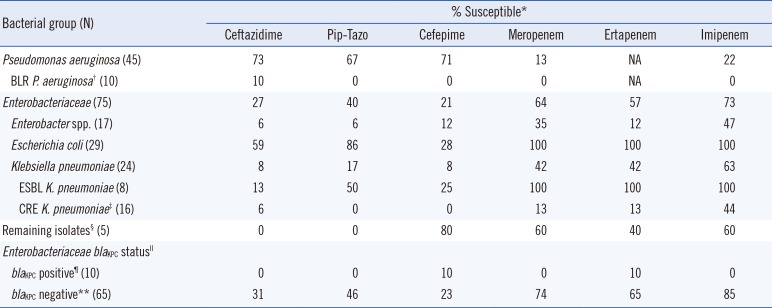
Table 1 shows the overall β-lactam susceptibility profile. A subset of
P. aeruginosa isolates (n=10, 22%), termed “β-lactam-resistant (BLR)”, were resistant to piperacillin/tazobactam, cefepime, meropenem, and imipenem.
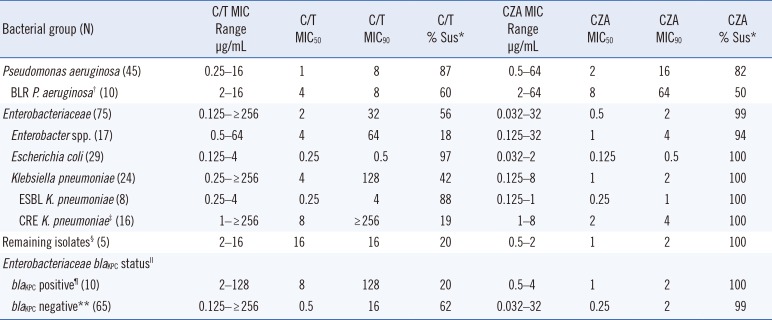
The 50% minimum inhibitory concentration (MIC
50, 1 µg/mL) and 90% minimum inhibitory concentration (MIC
90, 8 µg/mL) of C/T were lower for the
P. aeruginosa isolates than for the
Enterobacteriaceae isolates (2 µg/mL and 32 µg/mL, respectively;
Table 2). Furthermore, 87% (n=39) of all
P. aeruginosa isolates and 60% (n=6) of the BLR
P. aeruginosa isolates were C/T-susceptible (
Table 2).
Within the
Enterobacteriaceae, the C/T data showed group-dependent differences. For example, the
E. coli isolates had a low MIC
90 (0.5 µg/mL), whereas the
Enterobacter spp. had a higher MIC
90 (64 µg/mL). Overall, 56% (n=42) of all
Enterobacteriaceae isolates were C/T-susceptible (
Table 2).
In contrast to the C/T results, the
Enterobacteriaceae had lower MIC
50 (0.5 µg/mL) and MIC
90 (2 µg/mL) values for CZA compared to the
P. aeruginosa isolates (2 µg/mL and 16 µg/mL, respectively;
Table 2). Notably, 82% (n=37) of all
P. aeruginosa isolates were CZA-susceptible (
Table 2), whereas 99% (n=74) of
Enterobacteriaceae isolates were CZA-susceptible. An
Enterobacter spp. isolate was CZA-resistant (MIC of 32 µg/mL), but was found to be negative for the
blaKPC and
blaNDM genes by real-time PCR.
Comparison of the P. aeruginosa C/T and CZA results showed 87% (n=39) concordance with 35 isolates testing susceptible and four isolates testing non-susceptible to both antibiotics. For the Enterobacteriaceae, there was only a 57% concordance between the C/T and CZA results; 42 isolates tested susceptible and one isolate tested non-susceptible to both antibiotics. All remaining isolates were only CZA-susceptible.
The availability of new agents with anti-gram-negative activity holds promise for treating MDR organisms. CZA provides an alternative treatment for
blaKPC-positive organisms, which are otherwise treated by agents with less desirable safety or efficacy profiles. Ceftolozane is a new cephalosporin with activity against
P. aeruginosa, and in combination with tazobactam shows activity against ESBL-producing
Enterobacteriaceae [
9]. Notably, although two of the
blaKPC-positive isolates were C/T-susceptible, we would not expect C/T to be clinically effective.
In summary, we report the activities of C/T and CZA against a collection of BLR Enterobacteriaceae and P. aeruginosa isolates. Our results suggest that C/T and CZA are active against and represent possible therapeutic options for infections with BLR gram-negative bacteria.
Acknowledgments
Cubist supplied the C/T Etest strips, and Actavis supplied the CZA Etest strips. There was no funding for this study.
References
1. Estabrook M, Bussell B, Clugston SL, Bush K.
In vitro activity of ceftolozane-tazobactam as determined by broth dilution and agar diffusion assays against recent U.S.
Escherichia coli isolates from 2010 to 2011 carrying CTX-M-type extended-spectrum β-lactamases. J Clin Microbiol. 2014; 52:4049–4052. PMID:
25143578.
2. Sader HS, Castanheira M, Mendes RE, Flamm RK, Farrell DJ, Jones RN. Ceftazidime-avibactam activity against multidrug-resistant
Pseudomonas aeruginosa isolated in U.S. medical centers in 2012 and 2013. Antimicrob Agents Chemother. 2015; 59:3656–3659. PMID:
25845861.
3. Buehrle DJ, Shields RK, Chen L, Hao B, Press EG, Alkrouk A, et al. Evaluation of the
in vitro activity of ceftazidime-avibactam and ceftolozane-tazobactam against meropenem-resistant
Pseudomonas aeruginosa isolates. Antimicrob Agents Chemother. 2016; 60:3227–3231. PMID:
26976862.
4. Doern CD, Dunne WM Jr, Burnham CA. Detection of
Klebsiella pneumoniae carbapenemase (KPC) production in non-
Klebsiella pneumoniae Enterobacteriaceae isolates by use of the Phoenix, Vitek 2, and disk diffusion methods. J Clin Microbiol. 2011; 49:1143–1147. PMID:
21209164.
6. Manji R, Bythrow M, Branda JA, Burnham CA, Ferraro MJ, Garner OB, et al. Multi-center evaluation of the Vitek® MS system for mass spectrometric identification of non-
Enterobacteriaceae Gram-negative bacilli. Eur J Clin Microbiol Infect Dis. 2014; 33:337–346. PMID:
24019163.
7. Richter SS, Sercia L, Branda JA, Burnham CA, Bythrow M, Ferraro MJ, et al. Identification of
Enterobacteriaceae by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry using the Vitek MS system. Eur J Clin Microbiol Infect Dis. 2013; 32:1571–1578. PMID:
23818163.
8. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. 25th Informational supplement, M100-S25. Wayne, PA: Clinical and Laboratory Standards Institute;2015.
9. Sucher AJ, Chahine EB, Cogan P, Fete M. Ceftolozane/tazobactam: A new cephalosporin and β-lactamase inhibitor combination. Ann Pharmacother. 2015; 49:1046–1056. PMID:
26160970.
Table 1
β-Lactam susceptibility profiles of the study isolates (N=120)
|
Bacterial group (N) |
% Susceptible*
|
|
Ceftazidime |
Pip-Tazo |
Cefepime |
Meropenem |
Ertapenem |
Imipenem |
|
Pseudomonas aeruginosa (45) |
73 |
67 |
71 |
13 |
NA |
22 |
|
BLR P. aeruginosa† (10) |
10 |
0 |
0 |
0 |
NA |
0 |
|
Enterobacteriaceae (75) |
27 |
40 |
21 |
64 |
57 |
73 |
|
Enterobacter spp. (17) |
6 |
6 |
12 |
35 |
12 |
47 |
|
Escherichia coli (29) |
59 |
86 |
28 |
100 |
100 |
100 |
|
Klebsiella pneumoniae (24) |
8 |
17 |
8 |
42 |
42 |
63 |
|
ESBL K. pneumoniae (8) |
13 |
50 |
25 |
100 |
100 |
100 |
|
CRE K. pneumoniae‡ (16) |
6 |
0 |
0 |
13 |
13 |
44 |
|
Remaining isolates§ (5) |
0 |
0 |
80 |
60 |
40 |
60 |
|
Enterobacteriaceae blaKPC status∥
|
|
|
|
|
|
|
|
blaKPC positive¶ (10) |
0 |
0 |
10 |
0 |
10 |
0 |
|
blaKPC negative** (65) |
31 |
46 |
23 |
74 |
65 |
85 |
Table 2
Ceftolozane-tazobactam (C/T) and ceftazidime-avibactam (CZA) activity against BLR gram-negative bacteria
|
Bacterial group (N) |
C/T MIC Range µg/mL |
C/T MIC50
|
C/T MIC90
|
C/T % Sus*
|
CZA MIC Range µg/mL |
CZA MIC50
|
CZA MIC90
|
CZA % Sus*
|
|
Pseudomonas aeruginosa (45) |
0.25–16 |
1 |
8 |
87 |
0.5–64 |
2 |
16 |
82 |
|
BLR P. aeruginosa† (10) |
2–16 |
4 |
8 |
60 |
2–64 |
8 |
64 |
50 |
|
Enterobacteriaceae (75) |
0.125– ≥ 256 |
2 |
32 |
56 |
0.032–32 |
0.5 |
2 |
99 |
|
Enterobacter spp. (17) |
0.5–64 |
4 |
64 |
18 |
0.125–32 |
1 |
4 |
94 |
|
Escherichia coli (29) |
0.125–4 |
0.25 |
0.5 |
97 |
0.032–2 |
0.125 |
0.5 |
100 |
|
Klebsiella pneumoniae (24) |
0.25– ≥ 256 |
4 |
128 |
42 |
0.125–8 |
1 |
2 |
100 |
|
ESBL K. pneumoniae (8) |
0.25–4 |
0.25 |
4 |
88 |
0.125–1 |
0.25 |
1 |
100 |
|
CRE K. pneumoniae‡ (16) |
1– ≥ 256 |
8 |
≥ 256 |
19 |
1–8 |
2 |
4 |
100 |
|
Remaining isolates§ (5) |
2–16 |
16 |
16 |
20 |
0.5–2 |
1 |
2 |
100 |
|
Enterobacteriaceae blaKPC status∥
|
|
|
|
|
|
|
|
|
|
blaKPC positive¶ (10) |
2–128 |
8 |
128 |
20 |
0.5–4 |
1 |
2 |
100 |
|
blaKPC negative** (65) |
0.125– ≥ 256 |
0.5 |
16 |
62 |
0.032–32 |
0.25 |
2 |
99 |






 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download